Quantum mechanics, unlike classical physics, allows objects to exist in more than one state at the same time. This idea is often illustrated by Schrödinger’s cat, imagined as being both alive and dead until it is observed. In the laboratory, physicists can create less dramatic but very real versions of this effect by placing atoms, light or motion into two distinct quantum states at once. Creating and controlling these superpositions is essential for applications ranging from quantum computing to precision timekeeping.
A simple example is a quantum bit, or qubit, in a superposition of both 0 and 1. But quantum systems are not limited to just two states. In a quantum harmonic oscillator, which can occupy many different energy levels, there is a much richer set of possibilities. Quantum harmonic oscillators describe many physical systems, including light, vibrations and the motion of trapped particles, and have been used to create a wide variety of quantum superpositions. One well-known example is a “cat state,” in which an oscillator is placed in a superposition of two wave packets displaced in opposite directions. These wave packets, known as coherent states, resemble classical motion as closely as quantum mechanics allows.
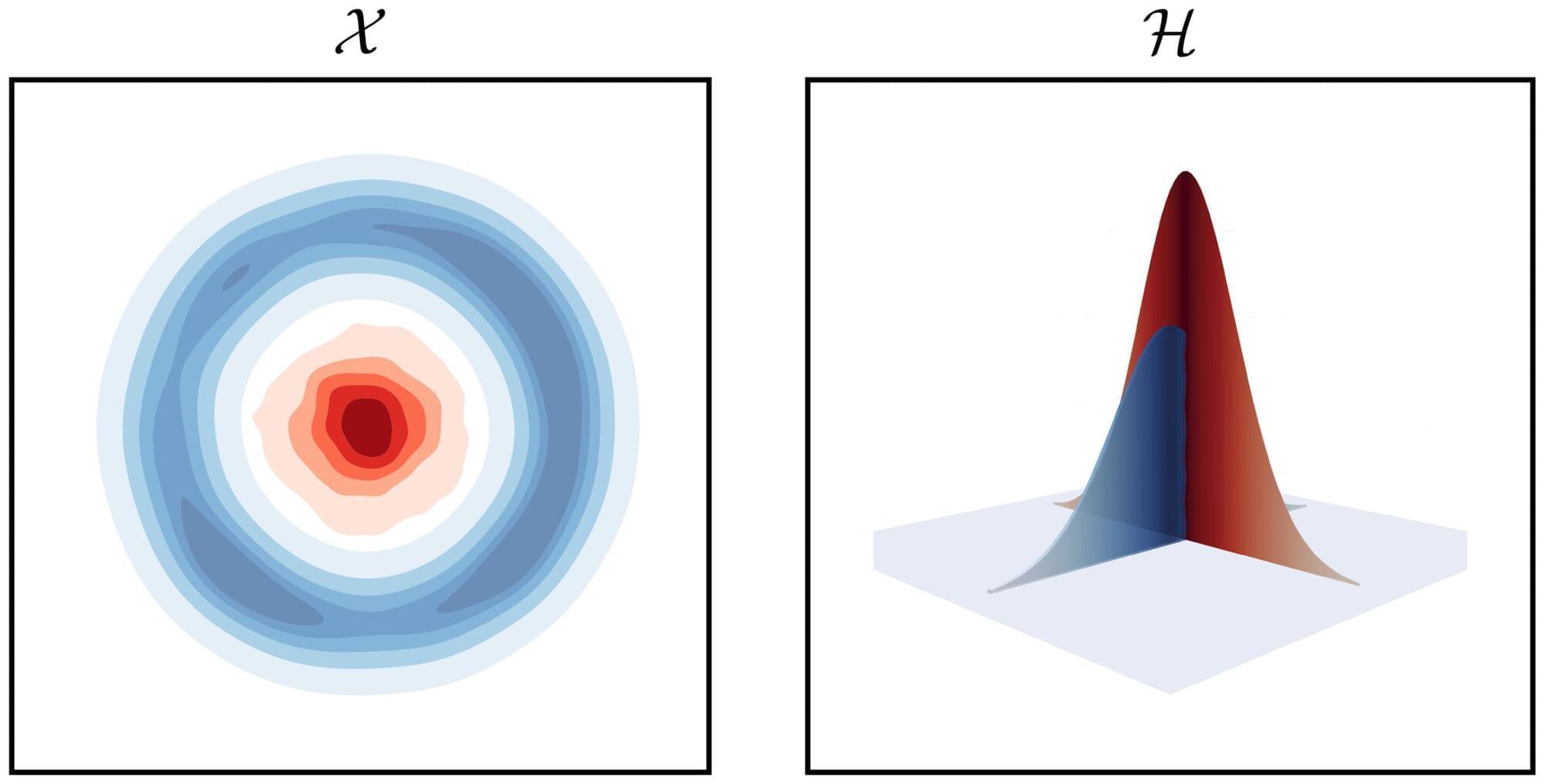
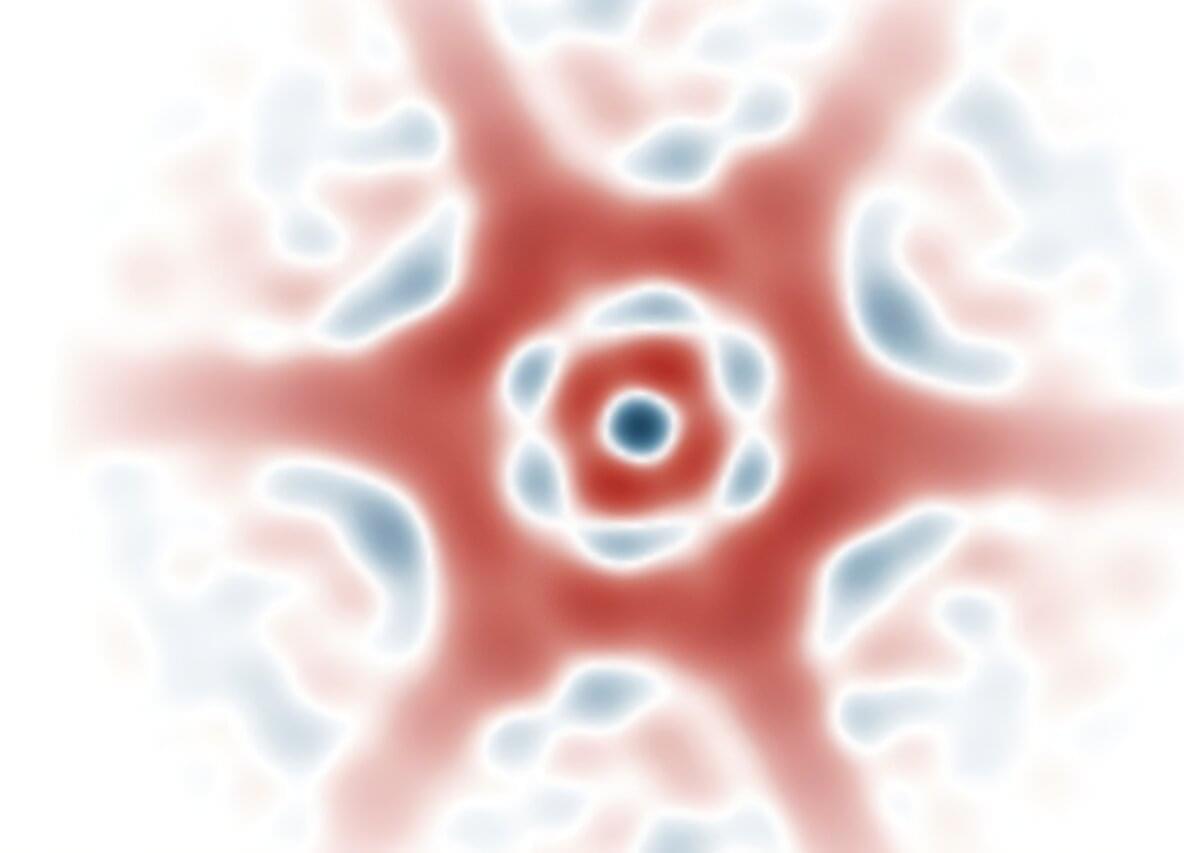
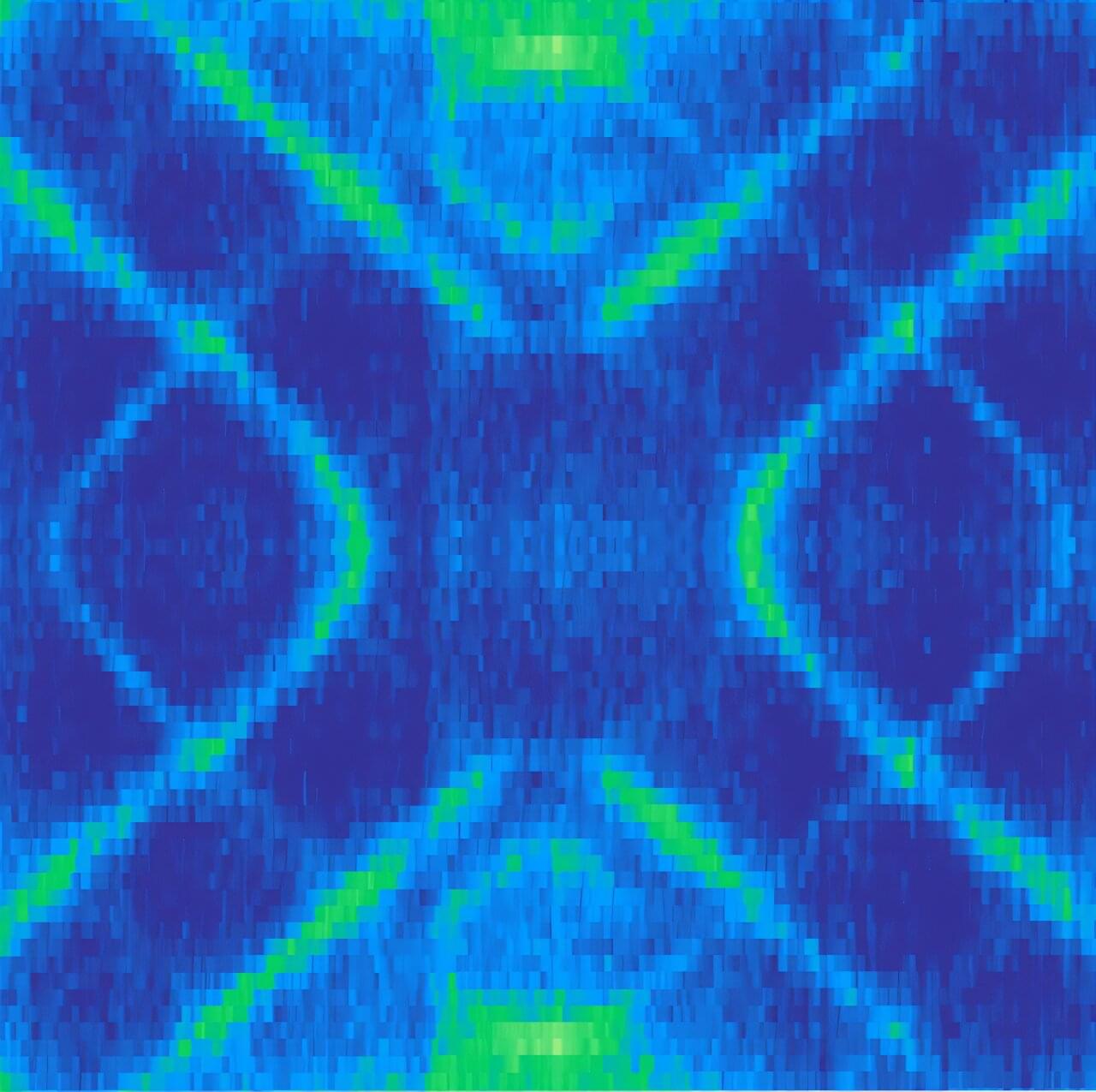
Researchers at the University of Oxford have now demonstrated a new family of quantum superpositions. Instead of building catlike states from coherent-state wave packets, they developed a method for creating superpositions from a broad range of components that are themselves highly nonclassical. In examples such as squeezed-state superpositions, quantum uncertainty is redistributed differently in each part of the state. The research is published in the journal Physical Review X.