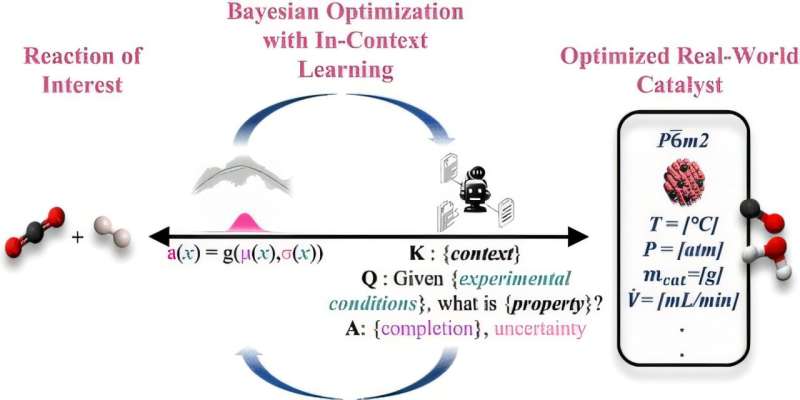
Advances in artificial intelligence promise to help chemical engineers discover complex new materials. These materials could be used for reactions such as turning carbon dioxide into fuel, but technical barriers have limited catalysis adoption so far. Researchers at the University of Rochester are now harnessing the benefits of large language models (LLMs) similar to ChatGPT, Claude, or Gemini to empower more researchers to use AI to discover new materials and accelerate experiment workflows.
In a study published in ACS Central Science, a team led by Marc Porosoff, an associate professor in the Department of Chemical and Sustainability Engineering, and Andrew White, visiting associate professor and the cofounder and chief technology officer of Edison Scientific, describes an AI based–method they developed that allows users to input natural language prompts about the materials they want to create and suggest optimal procedures for experiments to produce them. As the users run the experiments, they input the results back into the AI model and continue iterating until they reach their goal.
“We’re able to leverage the pre-trained knowledge of large language models and well-established statistical methods for materials discovery to help us as researchers navigate large experimental design spaces more efficiently,” says Porosoff.