Heat behaves in predictable ways: a hot cup of coffee cools, a laptop warms your hands, the sun heats Earth. But at scales thousands of times smaller than a human hair, those rules begin to break down, and scientists are learning how to take advantage of that.
A new study, published in Nature from researchers at Carnegie Mellon University, in collaboration with Stanford University and Purdue University, shows that heat can be manipulated far more powerfully than previously demonstrated using carefully engineered metamaterials. The work offers one of the clearest experimental confirmations yet that heat transfer can be actively designed and enhanced.
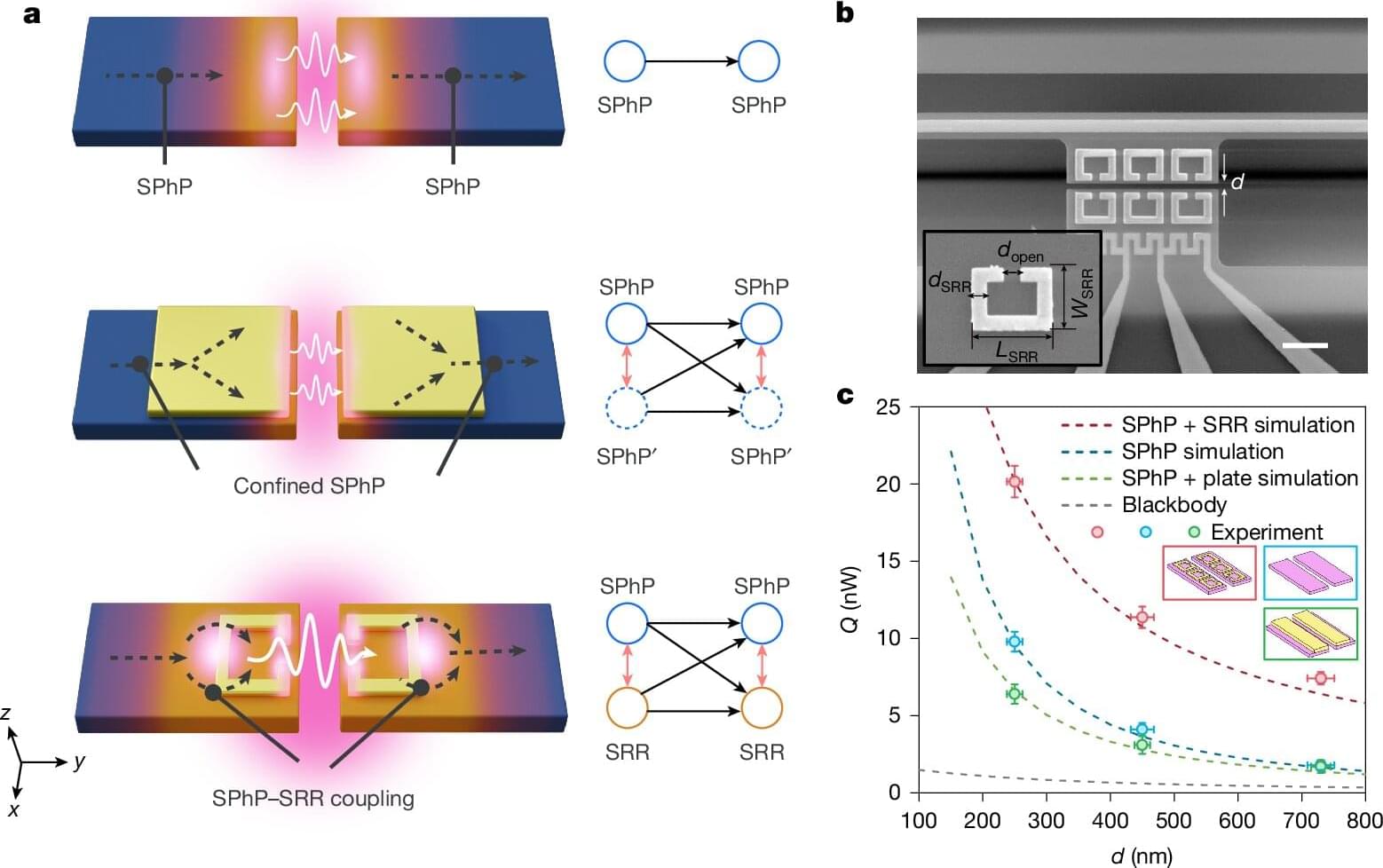
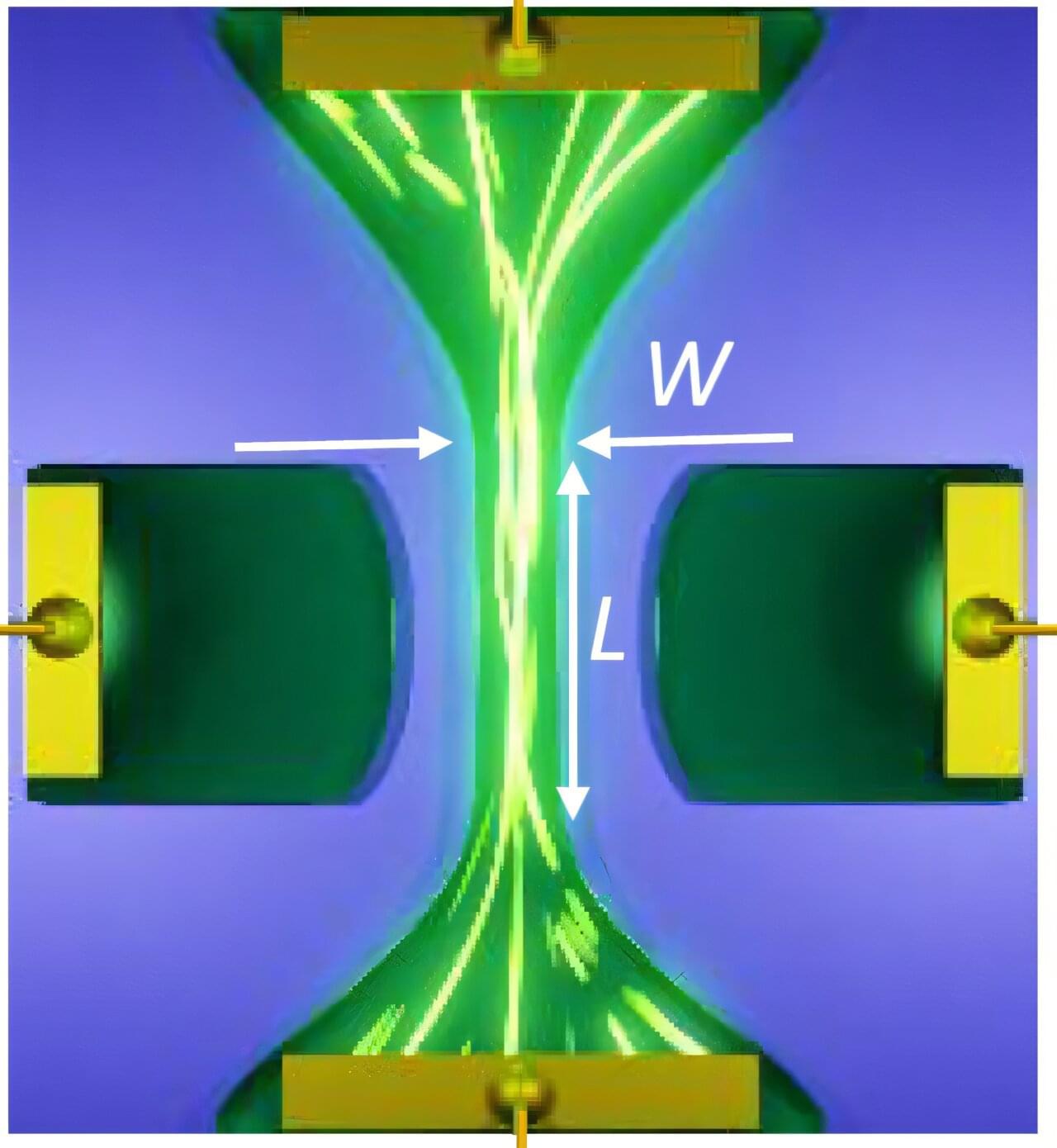
At the core of the discovery is a phenomenon called near-field radiative heat transfer. When two objects are brought extremely close together—just a few hundred nanometers apart—heat doesn’t simply radiate away in the usual sense. Instead, it can tunnel across the gap through electromagnetic waves, dramatically increasing how much energy flows between them.