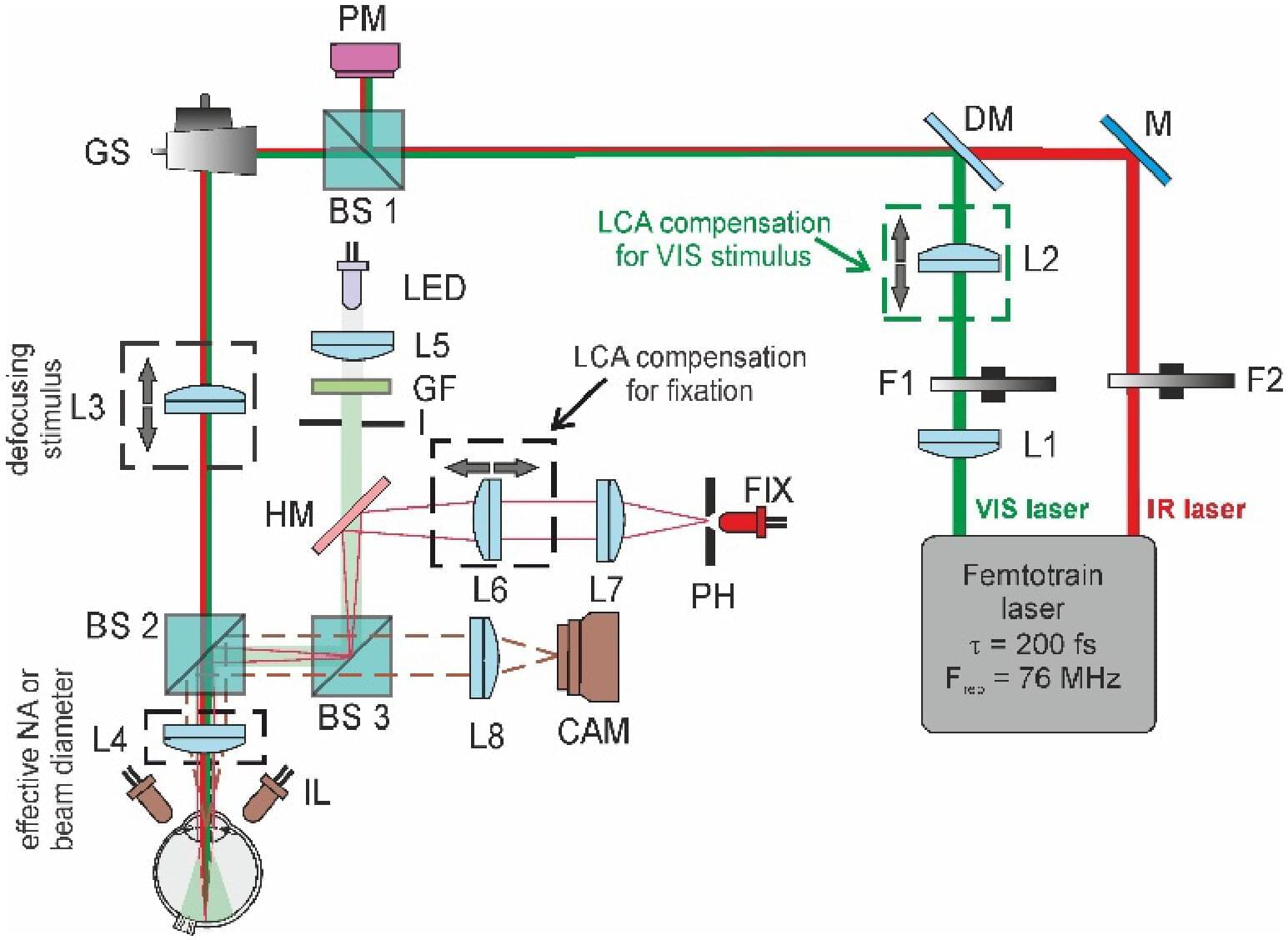
Near-infrared light is invisible to humans. And yet, under the right conditions, the human eye can perceive it. Researchers from Poland’s International Center for Translational Eye Research (ICTER) have now shown that the efficiency of this phenomenon depends not only on the laser pulse itself, but also on two highly specific factors: the beam diameter and the precise focusing of light on the retina. The research is published in the journal Optics Letters.
In everyday life, we see visible light—wavelengths detected by the photoreceptors of the retina. Near-infrared light lies outside this range, which is why it normally remains invisible to us. However, for several years, scientists have known of an exception.
This exception is known as two-photon vision. In this phenomenon, a photopigment in the retina absorbs two infrared photons almost simultaneously. Each photon individually carries too little energy to trigger visual perception, but together they can initiate the process of vision. This is why, under certain conditions, humans can “see” radiation that theoretically should remain invisible.