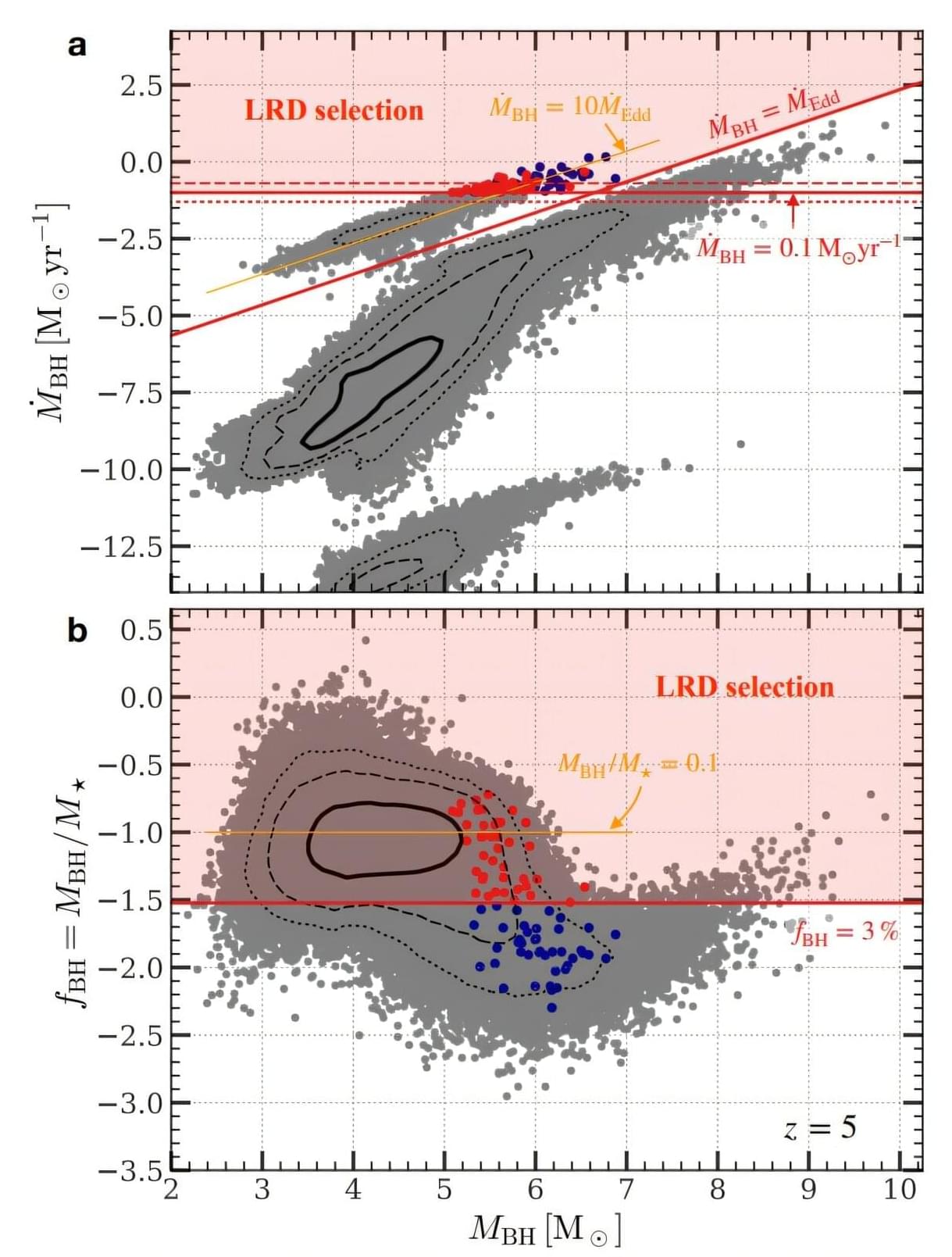
A new theoretical study may have cracked one of the most puzzling discoveries of the James Webb Space Telescope (JWST): Little Red Dots, spotted across the early universe. The paper, posted to the arXiv preprint server on May 29, argues that these objects could be black holes caught in rare, violent bursts of feeding at a rate exceeding theoretical limits.
Since JWST began its survey of the deep universe, astronomers have been puzzled by a class of tiny, faint objects appearing in the early universe in far greater numbers than expected. They have a distinctive V-shaped spectrum—bright in both ultraviolet and optical light, but with a dip in between—along with broad emission lines hinting at active black holes. They also show an absence of X-ray, radio and infrared emission.
They don’t look like ordinary galaxies, and they don’t completely look like quasars, either. What they are has been an open question. Some researchers argue that Little Red Dots may need some outside-the-box physics to explain their origin and nature.