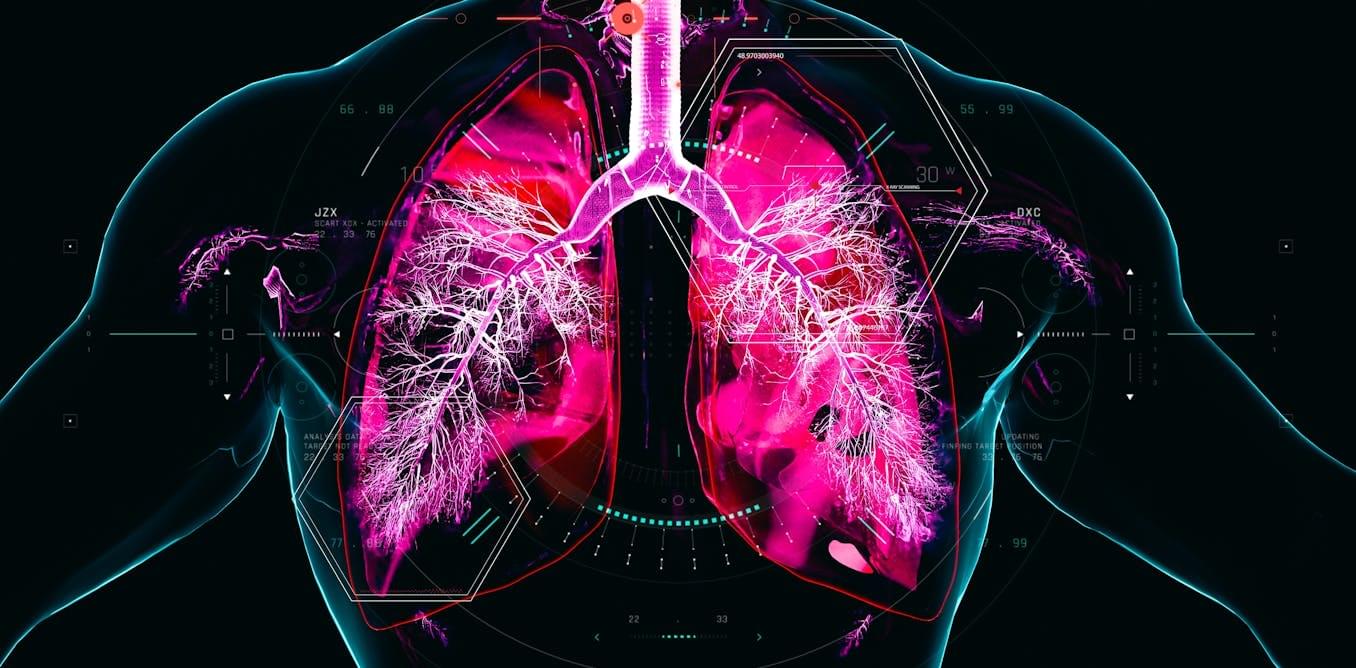
Although cigarette smoking remains the main driver of COPD, e-cigarettes are also raising concerns. Vaping aerosols can contain nicotine, ultrafine particles and flavouring chemicals that may irritate the lungs and contribute to inflammation. The long-term effects are still unclear because these products are relatively new.
That matters particularly for younger people. In Great Britain, recent survey data suggest that 7% of 11-to 17-year-olds currently vape. While that does not mean they will go on to develop COPD, it does mean more young lungs are being exposed to substances whose long-term effects are not yet fully understood.
COPD is often diagnosed only after major lung damage has already occurred. Because it develops so gradually, people may dismiss early breathlessness, coughing or mucus production as a consequence of getting older, being unfit or smoking. Respiratory organisations warn that symptoms such as cough, phlegm and shortness of breath should not be treated as a normal part of ageing, while studies show that COPD remains widely underdiagnosed, including among people with respiratory symptoms.