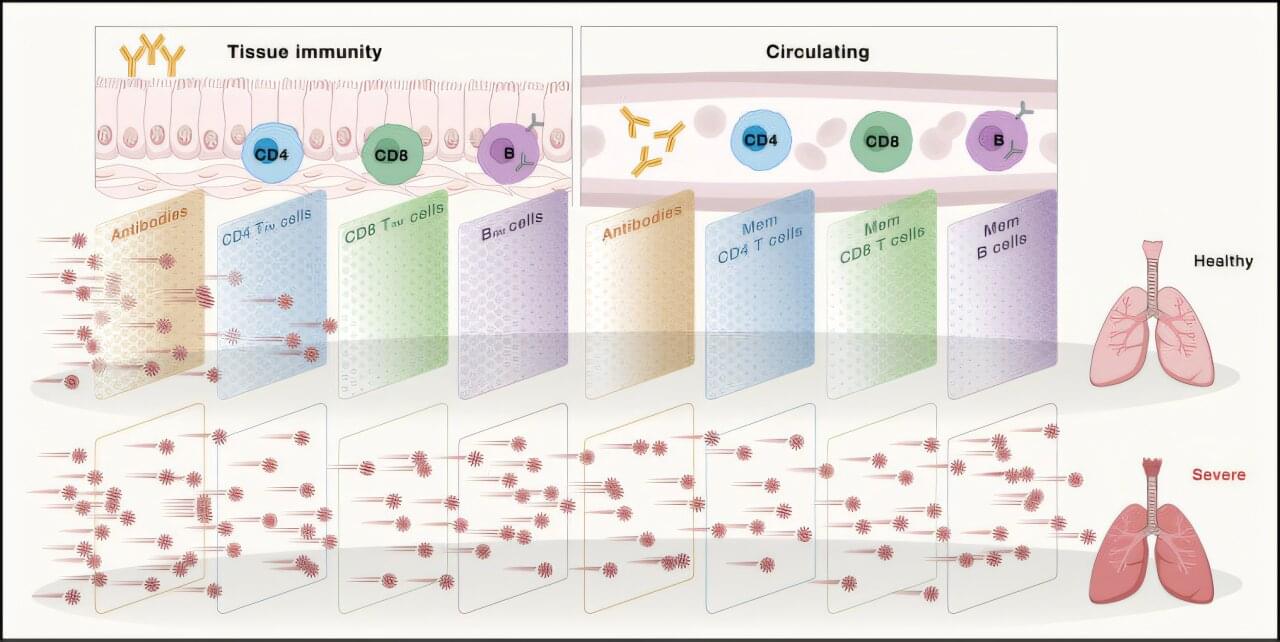
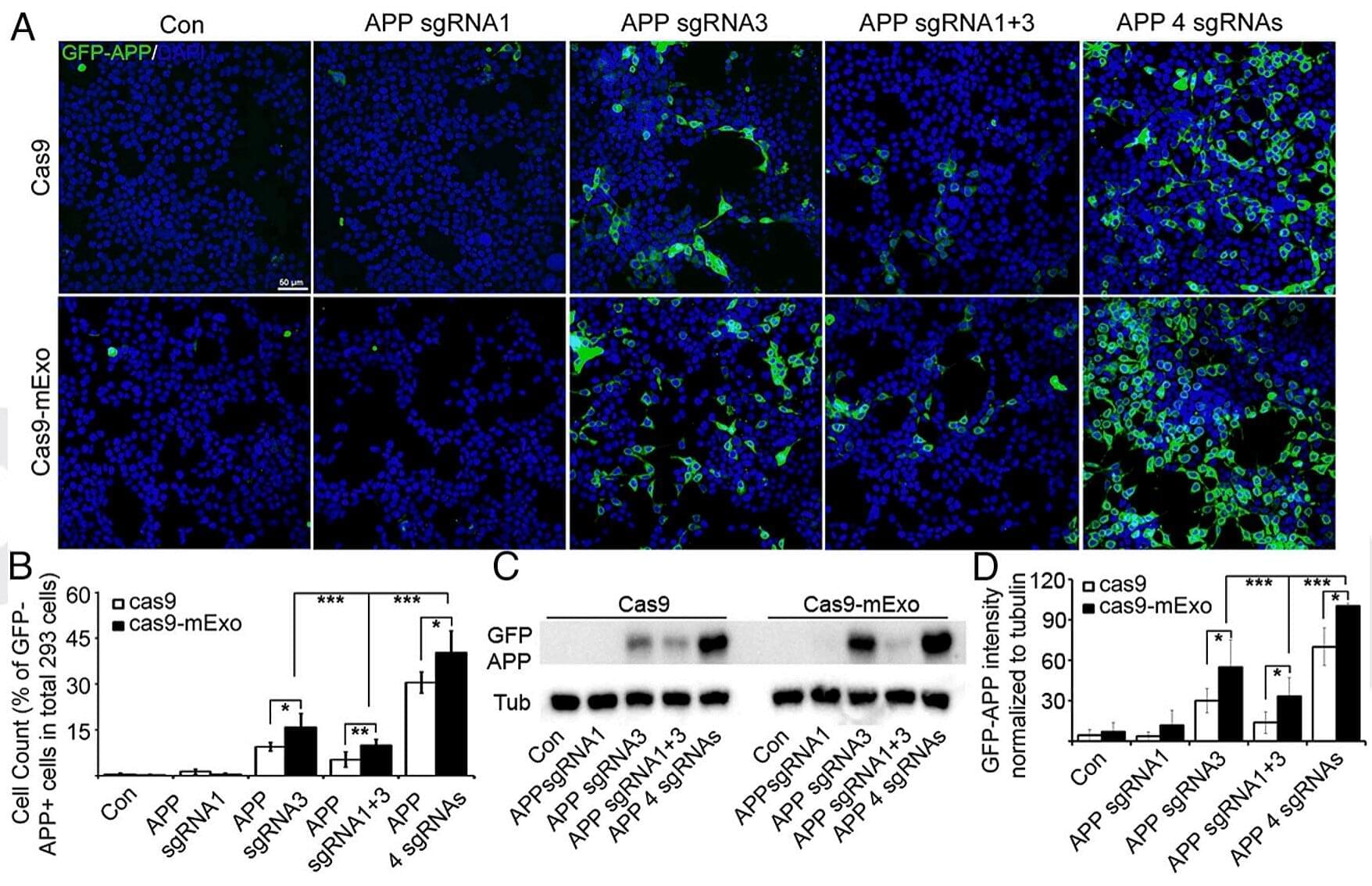
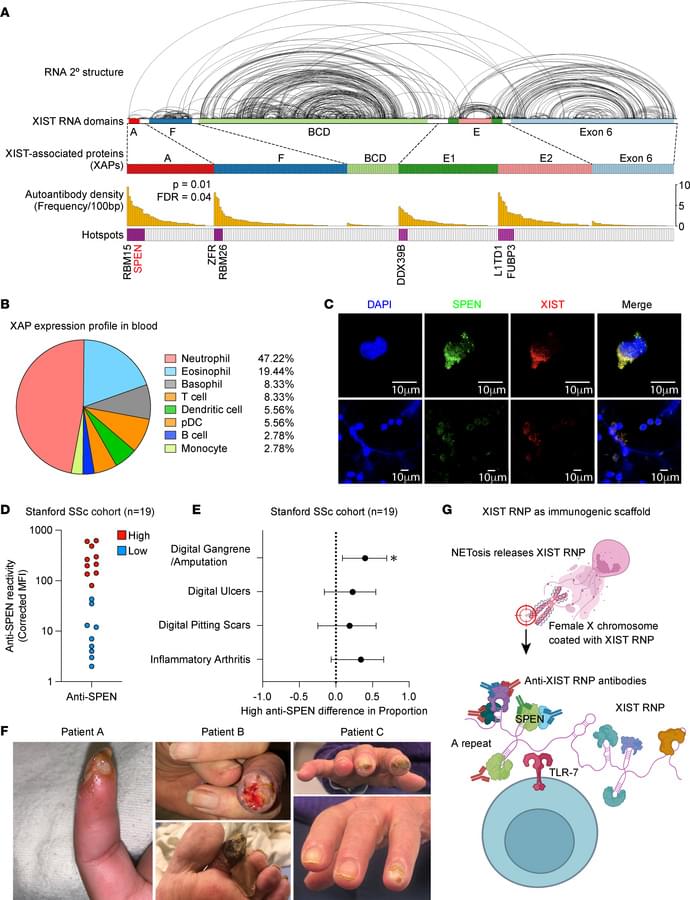
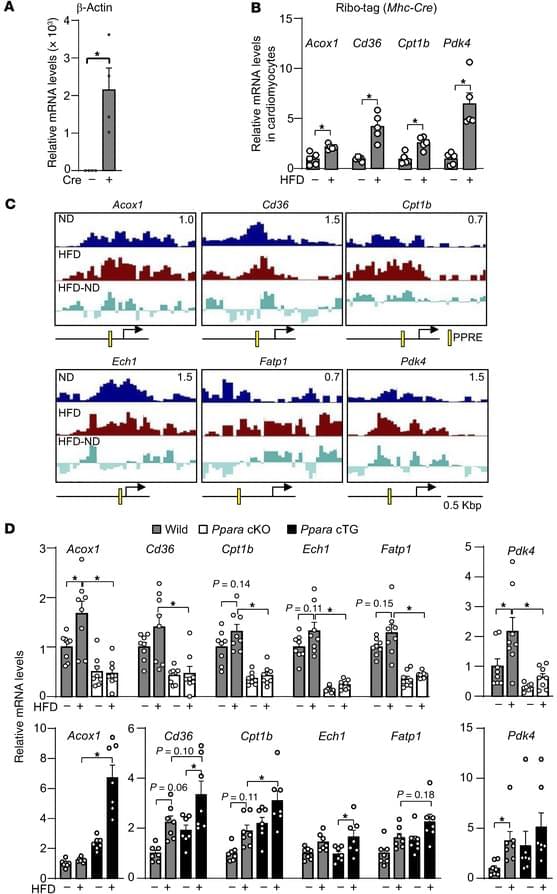
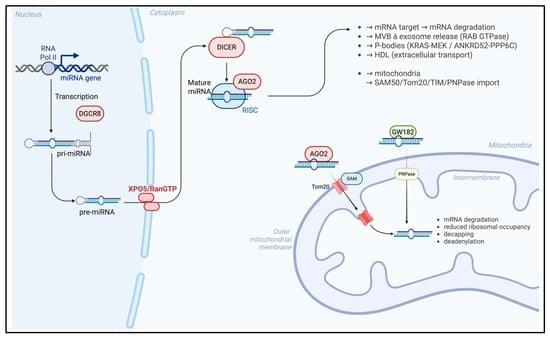
The average human has about 1.8 trillion immune cells. These cells patrol the body for bacteria, viruses, cancers, and other threats. Vaccines enhance this security system by teaching our immune cells to target specific pathogens. According to the World Health Organization, vaccine-induced immunity saves about six lives every minute. But how long does this protective immune “memory” last?
According to Shane Crotty, Ph.D., Professor and Chief Scientific Officer at La Jolla Institute for Immunology (LJI), we still have much to learn about immune memory.
“There are actually not many studies of human immune memory due to vaccines,” says Crotty. “Scientists traditionally don’t track immune memory past one year after vaccination—or even six months after vaccination—and that’s a bit of a problem.”