NCI-CONNECT Rare Brain and Spine Tumor Network connects patients, providers, and advocates to help people with rare brain and spine tumors get better care.



Florida State University chemists have synthesized new molecules derived from bacteria found in a Pacific Ocean sea sponge, a breakthrough for the future of drug development, particularly for rare forms of cancer.
“Around 50% of approved drugs are either natural products or derivatives of natural products,” said Zackary Firestone, a fourth-year doctoral student in FSU’s Department of Chemistry and Biochemistry, and the study’s lead author. “Synthetic access to these molecules is important because it allows for easier procurement for biological testing as well as the making of new derivatives.”
The research team is the first to successfully synthesize two new marine natural products: tetradehydrohalicyclamine B and epi-tetradehydrohalicyclamine B. Both were isolated from bacteria that lives in symbiosis with Acanthostrongylophora ingens, a Pacific-dwelling sea sponge.

A team at the University of Vienna, led by chemist Nuno Maulide, has developed a new method for controlling chemical reactions in a more targeted and efficient manner. At the heart of this is the concept of “cation sampling”: specially selected groups (ketones), in a sense, function as molecular signposts for randomly migrating positive charges, enabling reactions to take place at sites on a molecule that were previously difficult to access. The method allows carbon-hydrogen bonds (C–H bonds) to be specifically modified. The study was published in the Journal of the American Chemical Society.
Organic molecules form the basis of almost all biological processes. They consist mainly of carbon and hydrogen—and hydrogen atoms in particular are very common in such molecules. “If you want to alter the properties of a molecule, you often have to specifically replace individual hydrogen atoms,” explains Philipp Spieß, a former Ph.D. student in the Maulide group and one of the study’s lead authors.
The precise modification of C–H bonds is therefore considered one of the key challenges of modern synthetic chemistry. It plays an important role in the development of new drugs, functional materials and more efficient chemical processes.
The scale of chemistry simulations with quantum computing has increased dramatically in just the last few months. In the latest milestone for the field, researchers from Cleveland Clinic, RIKEN, and IBM used a quantum-centric supercomputing (QCSC) framework to calculate the electronic structure of a pair of large protein-ligand complexes, reaching a scale of 12,635 atoms in the largest simulation.
The molecules were T4-Lysozyme, a protein from a family of proteins involved in the immune system degradation of peptidoglycans in bacterial membranes, and Trypsin, produced in the pancreas and used in digestion. The team simulated these proteins binding to molecules they interact with in nature and immersed in a liquid water solution, at scales of 11,608 atoms and 12,635 atoms respectively. Bringing together an international team of researchers from across the United States and Japan made it possible to develop the necessary algorithm and workflow enhancements to reach this milestone.
The researchers achieved this scale just four months after modeling the 303-atom miniprotein Trp-cage using quantum computing for the first time. Today’s new result not only demonstrates a 40-fold increase in system size compared to the Trp-cage result, it represents a 210-times improvement in accuracy from previous state-of-the-art QCSC approaches in a specific step of the workflow.
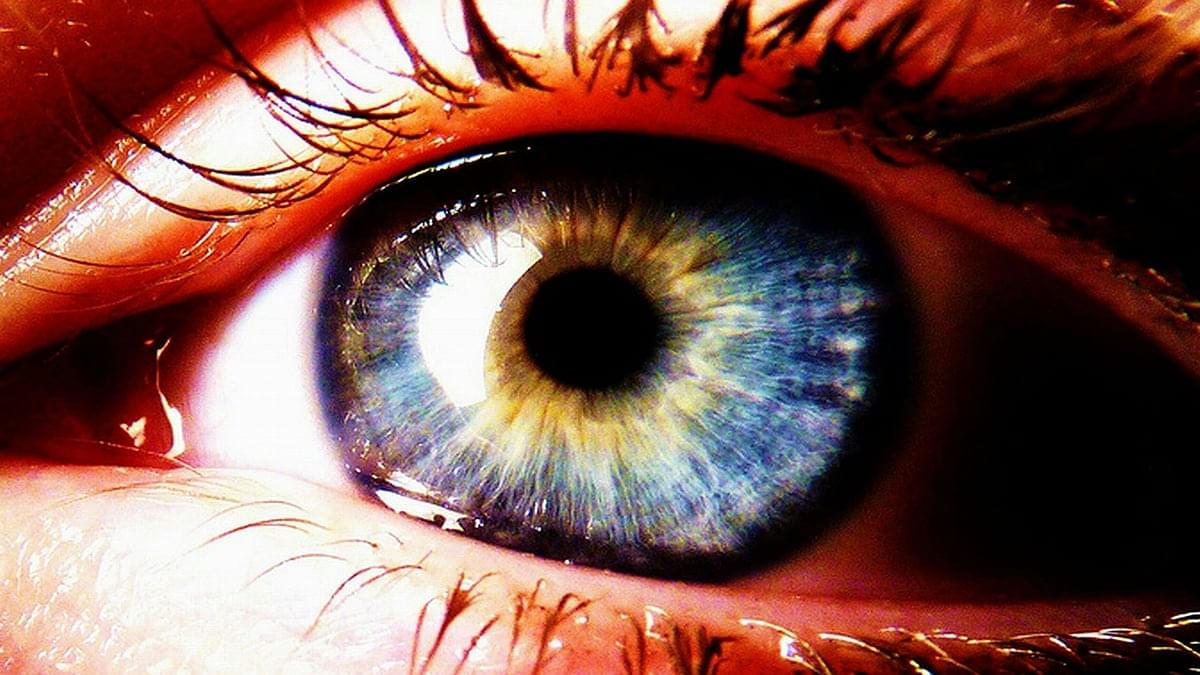
The eyes are a window into our deeper health.
As the only outward extension of the central nervous system, these sensory organs may reflect not only the state of our brain and blood vessels, but also our very bones.
Population studies in Singapore and the UK have now revealed that a person’s risk of osteoporosis may be associated with how quickly their eyes are aging.

Scientists in South Korea have developed experimental contact lenses designed to send electrical signals through the retina and into brain regions linked to mood. In mice, the technology appeared to improve depression-like behaviour.
The idea sounds futuristic: a contact lens that could one day help treat depression by stimulating the brain through the eye. The work is still at a very early stage, with findings so far limited to a single mouse study.
The eye is already one of the body’s most useful access points for medical technology.
Isomorphic Labs announces it has raised $2.1 Billion in Series B funding. The financing round is led by Thrive Capital, and includes participation from existing backers Alphabet and GV alongside new investors MGX, Temasek, CapitalG, and the UK Sovereign AI Fund, significantly expanding Isomorphic Labs’ global capital base.
Isomorphic Labs was founded with the ambition to leverage the power of AI to reimagine and accelerate drug discovery to bring much-needed treatments to millions of patients globally. The company aims to apply its pioneering AI drug design engine (IsoDDE) to deliver biomedical breakthroughs and is advancing drug design programs across multiple therapeutic areas and drug modalities.
Read more in the news release below.
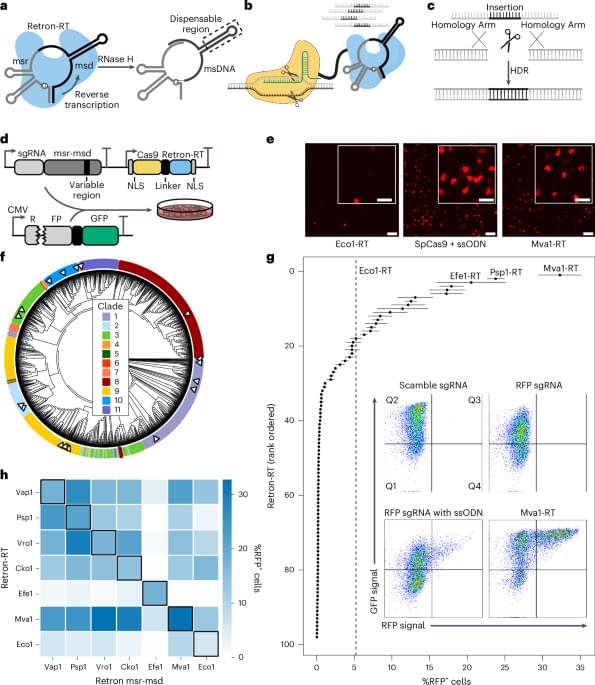
An extremely interesting new technology which combines bacterial retrons with CRISPR-Cas for localized generation of single-stranded DNA inserts and subsequent targeted genome editing. I remember reading about retrons as an obscure biological phenomenon years ago in a monograph called Mobile DNA III, so it’s awesome to see them leveraged in this way!
A metagenomic screen identifies retron reverse transcriptases for precise genome-editing applications.
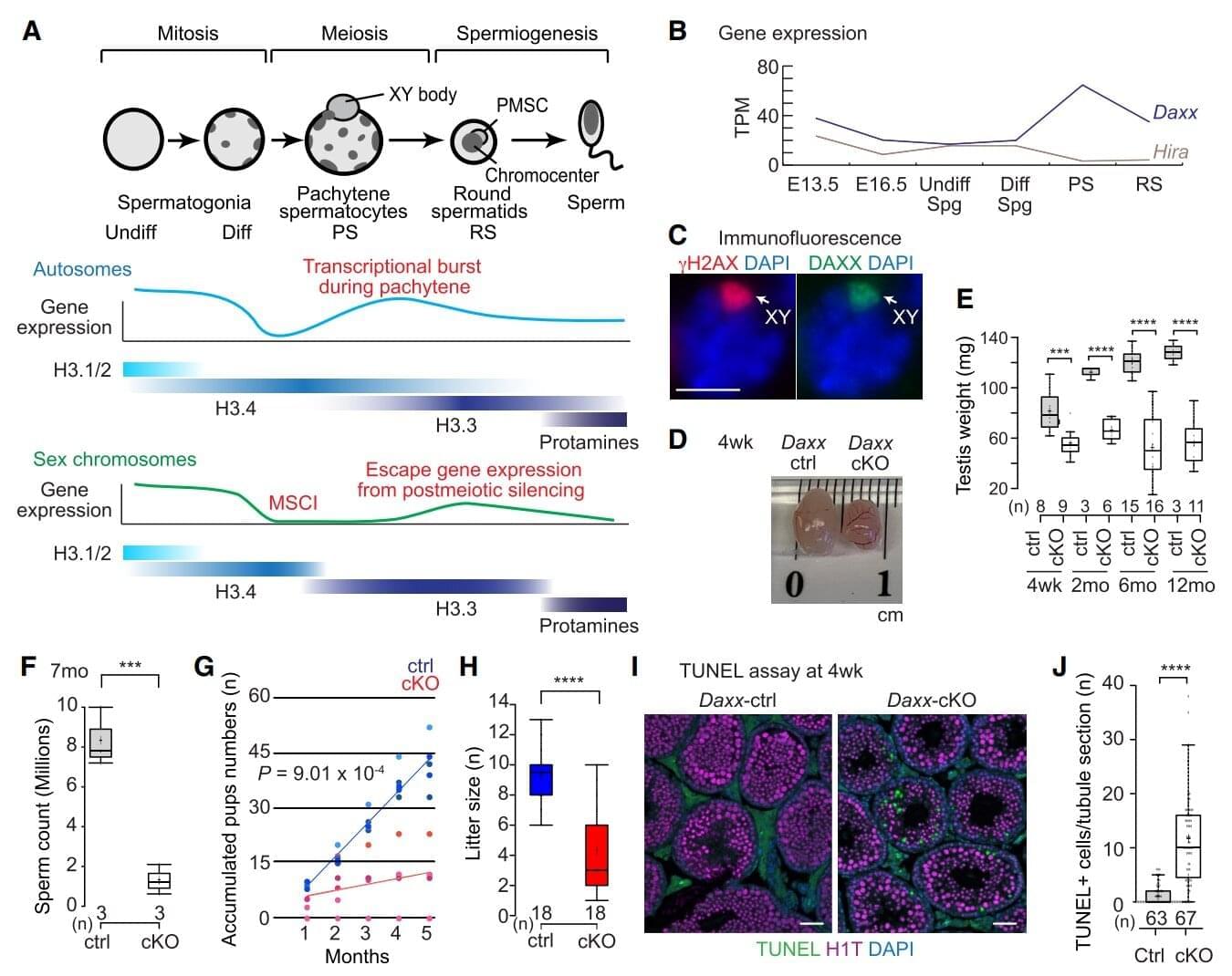
The causes of male infertility can be hard to diagnose, with many tests failing to detect genetic defects. Sometimes, infertility doesn’t even involve the genes themselves. It can arise from improper folding of the father’s DNA in the sperm. If a couple conceives, this mispackaged DNA can damage the lifelong health of the child.
“Paternal health is critical to sperm quality and the health of the offspring,” said Satoshi Namekawa, a professor of microbiology and molecular genetics. “Understanding the packing and folding of DNA in sperm cells is a fundamental question in modern biology.”
Namekawa and Ph.D. student Yu-Han Yeh have now unveiled an important new piece of this puzzle. They have identified a protein, called DAXX, that guides how sperm DNA is organized. DAXX silences thousands of genes so they don’t interfere with reproduction. It also keeps a handful of crucial genes turned on—shaping the delicate, early stages of embryonic development. The work was published recently in Genes & Development.