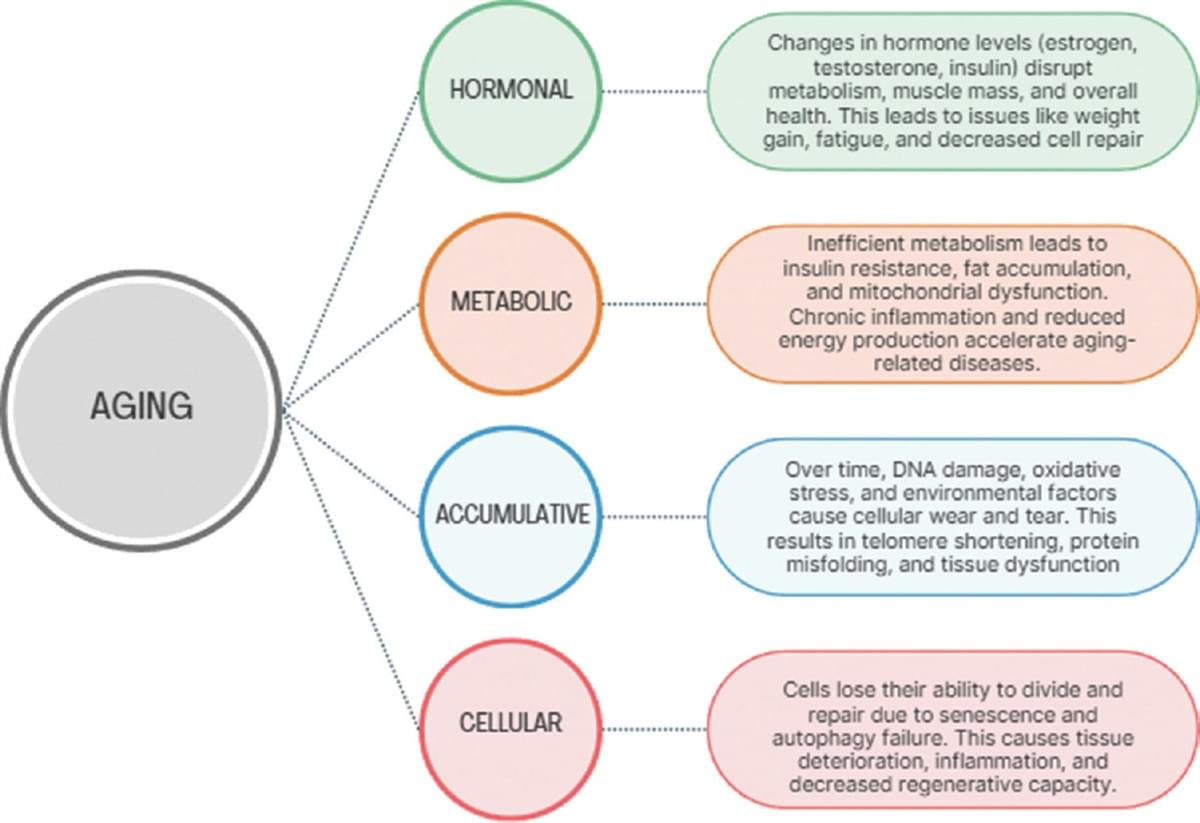
After receiving a stem cell transplant from his brother, a 63-year-old Norwegian man known as the “Oslo patient” has become one of only a handful of people to see their HIV (human immunodeficiency virus) go into long-term remission.
While HIV can now be controlled with medication that stops the virus from replicating, the virus remains in the body, rebounding when the drugs are stopped. So case studies like this one are invaluable for researchers working towards a full cure.
The Oslo man was given a bone marrow stem cell transplant to treat a rare type of blood cancer. Discovering at the last minute that his brother carried a rare genetic mutation previously shown to resist HIV, researchers led by a team from Oslo University Hospital closely tracked the operation’s impact on the virus.