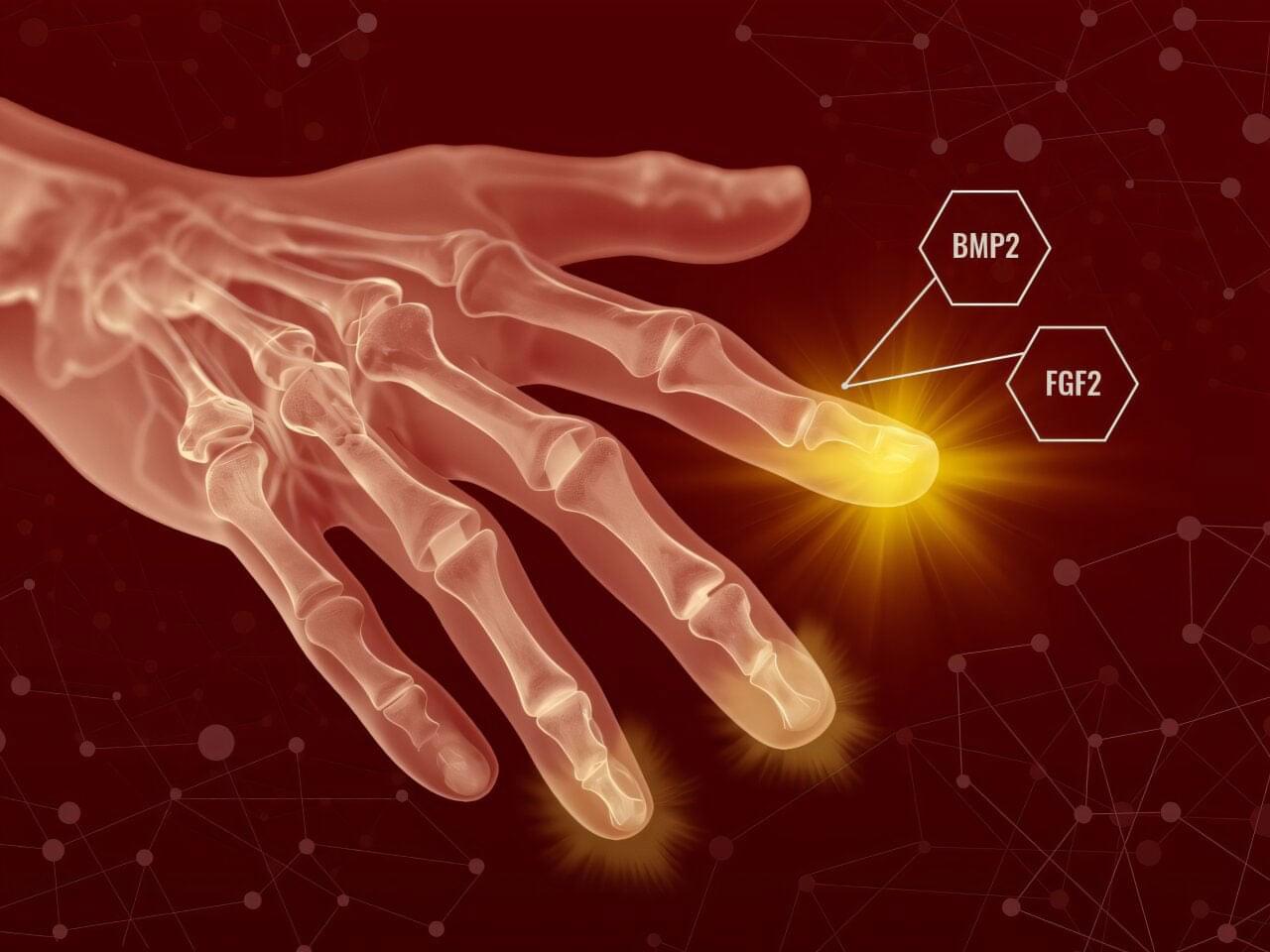
Researchers have successfully regenerated skeletal and connective tissue, although the new tissue was not perfectly formed. The result demonstrates a critical step forward in limb regeneration.

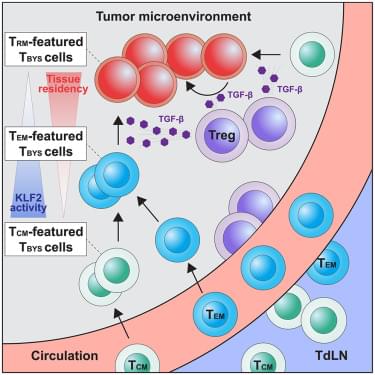
Lin et al. reveal that tumor-infiltrating bystander CD8+ T cells are heterogeneous and undergo a TCM-to-TEM-to-TRM differentiation trajectory driven by Treg-derived TGF-β signaling through suppression of KLF2. This mechanism sustains bystander CD8+ T cell heterogeneity and accumulation in tumors and provides a targetable mechanism for cancer immunotherapy.

New UCLA-led research finds that the use of telemedicine has not significantly increased visits and medical spending across all payer types. The findings, published in JAMA Network Open, could ease concerns among lawmakers that the telemedicine expansion that occurred during the COVID pandemic would result in large utilization and spending increases.
With the declaration of the COVID pandemic in 2020, the Centers for Medicare & Medicaid Services (CMS) changed key policies regarding telemedicine flexibility, such as introducing payment parity with in-person visits, waiving geographic restrictions, and eliminating out-of-pocket cost sharing.
Once the pandemic was declared over, however, lawmakers extended these changes to analyze how telemedicine impacted health care use and spending. Those CMS flexibilities are due to expire in 2027, and lawmakers continue to debate whether to permanently extend or modify them.
Researchers believe the findings could reshape our understanding of fundamental forces, space, and time.
#universe #cosmos #wion.
About Channel:
WION The World is One News examines global issues with in-depth analysis. We provide much more than the news of the day. Our aim is to empower people to explore their world. With our Global headquarters in New Delhi, we bring you news on the hour, by the hour. We deliver information that is not biased. We are journalists who are neutral to the core and non-partisan when it comes to world politics. People are tired of biased reportage and we stand for a globalized united world. So for us, the World is truly One.
Please keep discussions on this channel clean and respectful and refrain from using racist or sexist slurs and personal insults.
Check out our website: http://www.wionews.com

There is no dataset for grief.
No metric for justice.
No optimizer for legitimacy.
And yet we keep bringing the Hammer of AI to every problem we face. Climate change. Pandemics. Cancer. Energy. War. Political corruption. There is no problem that the omnipresent, all-knowing, all-mighty artificial superintelligence will not eventually crack.
This is a religion. Technology is its faith. Silicon Valley is its Promised Land. Entrepreneurs are its prophets. And we are all believers.
I should know. I used to be one.
In my latest piece on Singularity Weblog, I argue that some problems do bend to computation: fusion, protein folding, the genome. But others do not. They are not computable, only livable. And when we hammer them anyway, things break. Sometimes the thing that breaks is the problem. Sometimes it is us.

Researchers at the University of Rochester showed that one of those biological advantages can be moved into another mammal. By transferring a gene linked to the naked mole rat’s unusually high levels of high molecular weight hyaluronic acid (HMW-HA), the team improved health and modestly extended lifespan in mice.
The work, published in Nature in 2023, suggested that at least some longevity traits that evolved in long-lived animals may be adaptable beyond the species that developed them. The genetically modified mice lived healthier lives and had an approximate 4.4 percent increase in median lifespan compared with ordinary mice.
“Our study provides a proof of principle that unique longevity mechanisms that evolved in long-lived mammalian species can be exported to improve the lifespans of other mammals,” says Vera Gorbunova, the Doris Johns Cherry Professor of biology and medicine at Rochester.
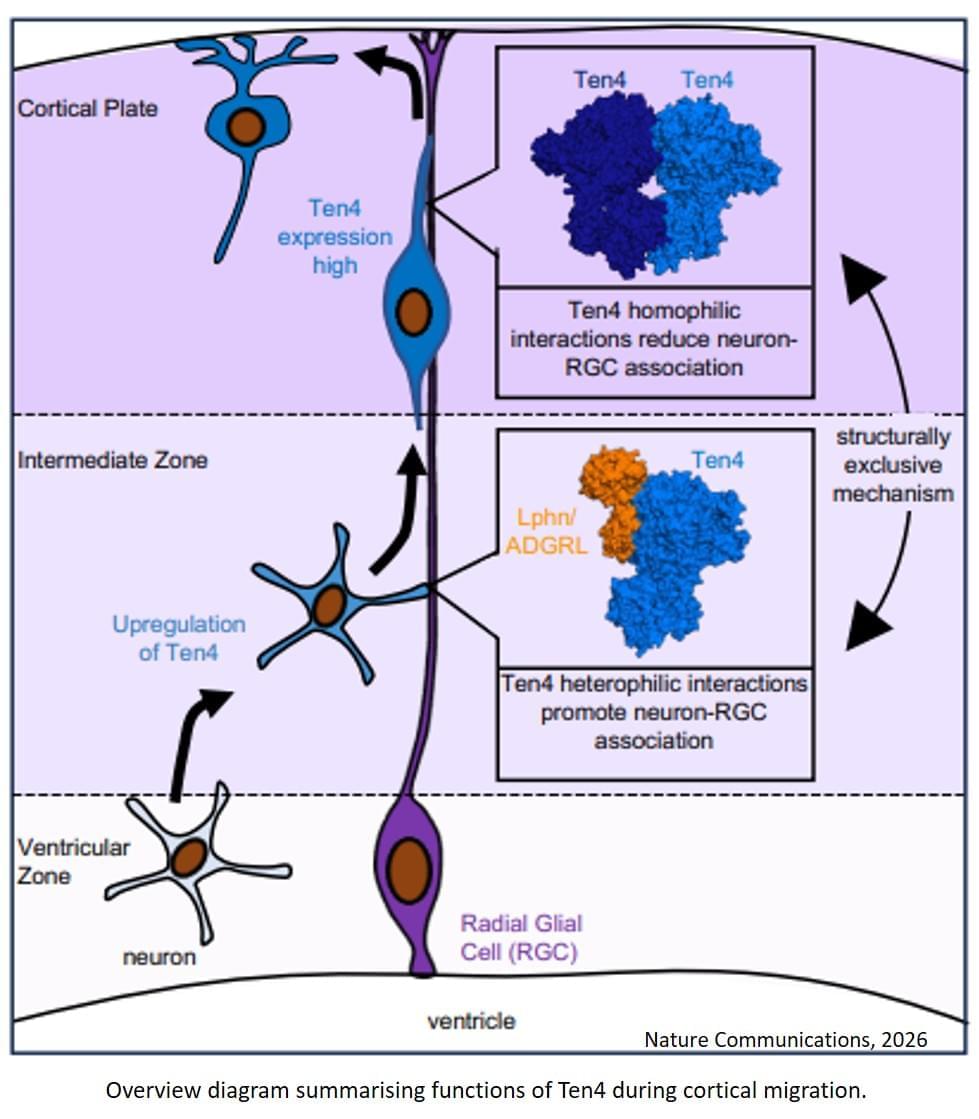
During brain development, neurons can regulate their movement until they reach their final destination thanks to a “molecular switch” involving the protein Teneurin 4 (Ten4). This protein can guide neuronal migration through mutually exclusive molecular pathways and determine the direction of nerve cells.
The discovery, published in the journal Nature Communications, improves our understanding of the molecular mechanisms that control neuronal migration and offers new insights into how the brain develops at the molecular level.
The study combines advanced techniques — structural protein studies, gene editing in animal models and super-resolution microscopy — to broaden our understanding of the origins of neurodevelopmental disorders and psychiatric or neurological conditions —schizophrenia, epilepsy, autism, bipolar disorder, etc. — which may be linked to errors in neuronal migration.

The threat is to the librarian. The threat is to the small, vanishing population of people who still go into the hexagons. Who still pull a book from the shelf. Who still spend three days reading it. Who still close it and feel changed. That practice is not a hobby. It is a technology. One older than print, older than the codex, possibly older than writing. It is a process of assembly inside one human skull. The kind of patient, sequential, focused and embodied attention that produces what we used to call understanding. AI does not produce that attention. AI produces a feeling that closely resembles attention while being something else, the way saccharin produces a feeling that closely resembles sweetness while being something else.
If this practice disappears, the Library will not notice. The books will not notice. The infinite hexagons will continue to extend in every direction. There will be no one in them. There will only be the queries, falling into the air, decaying into training data, generating fresh continuations for an audience that no longer reads them. Only, occasionally, glances at a summary.
This is the message Borges was telegraphing. This is what he saw, sitting in the National Library of Argentina, going slowly blind, surrounded by more books than any one man could read. He saw that the deepest threat to a literary culture was not the burning of books. It was the rendering of books unnecessary. He saw that a Library of Babel which contained every possible answer was, paradoxically, the most efficient instrument ever conceived for ending the practice of reading. And he saw, finally, that the only response available to a serious person was the response his narrator chose. To stop searching for the catalogue of catalogues. To return to one’s own hexagon. To pick up one particular book. To read it slowly. To die, eventually, a few leagues from where one was born, with one’s body falling through the fathomless air.
Join us on Patreon! / michaellustgartenphd.
Discount Links/Affiliates:
Blood testing (where I get the majority of my labs, for those who blood test with Quest): https://www.ultalabtests.com/partners… those who blood test with LabCorp: https://www.anrdoezrs.net/click-10161… At-Home Metabolomics: https://www.iollo.com?ref=michael-lus… Use Code: CONQUERAGING At Checkout Clearly Filtered Water Filter: https://get.aspr.app/SHoPY Epigenetic, Telomere Testing: https://trudiagnostic.com/?irclickid=… Use Code: CONQUERAGING NAD+ Quantification: https://www.jinfiniti.com/intracellul… Use Code: ConquerAging At Checkout Oral Microbiome: https://www.bristlehealth.com/?ref=mi… Enter Code: ConquerAging SiphoxHealth Blood Testing (ApoB, GrimAge): https://siphoxhealth.com/mlustgarten Green Tea: https://www.ochaandco.com/?ref=fqbtflod Use Code: ML10OFF Diet Tracking: https://shareasale.com/r.cfm?b=139013… If you’d like to support the channel, you can do that with the website, Buy Me A Coffee: https://www.buymeacoffee.com/mlhnrca Conquer Aging Or Die Trying Merch! https://my-store-d4e7df.creator-sprin… George’s YT channel: / @reprogrampodcast The Murphy Lab website: https://murphylaboratory.com/ X: @DrGJMurphy.
Blood Testing Essentials (Biological Age, CVD-Risk, Kidney Health and Function):
PhenoAge (Biological Age): https://www.ultalabtests.com/partners…
Risk-weighted ApoB (a better CVD predictor than LDL, non-HDL cholesterol, and ApoB): https://www.ultalabtests.com/partners…
Kidney health and function: https://www.ultalabtests.com/partners…