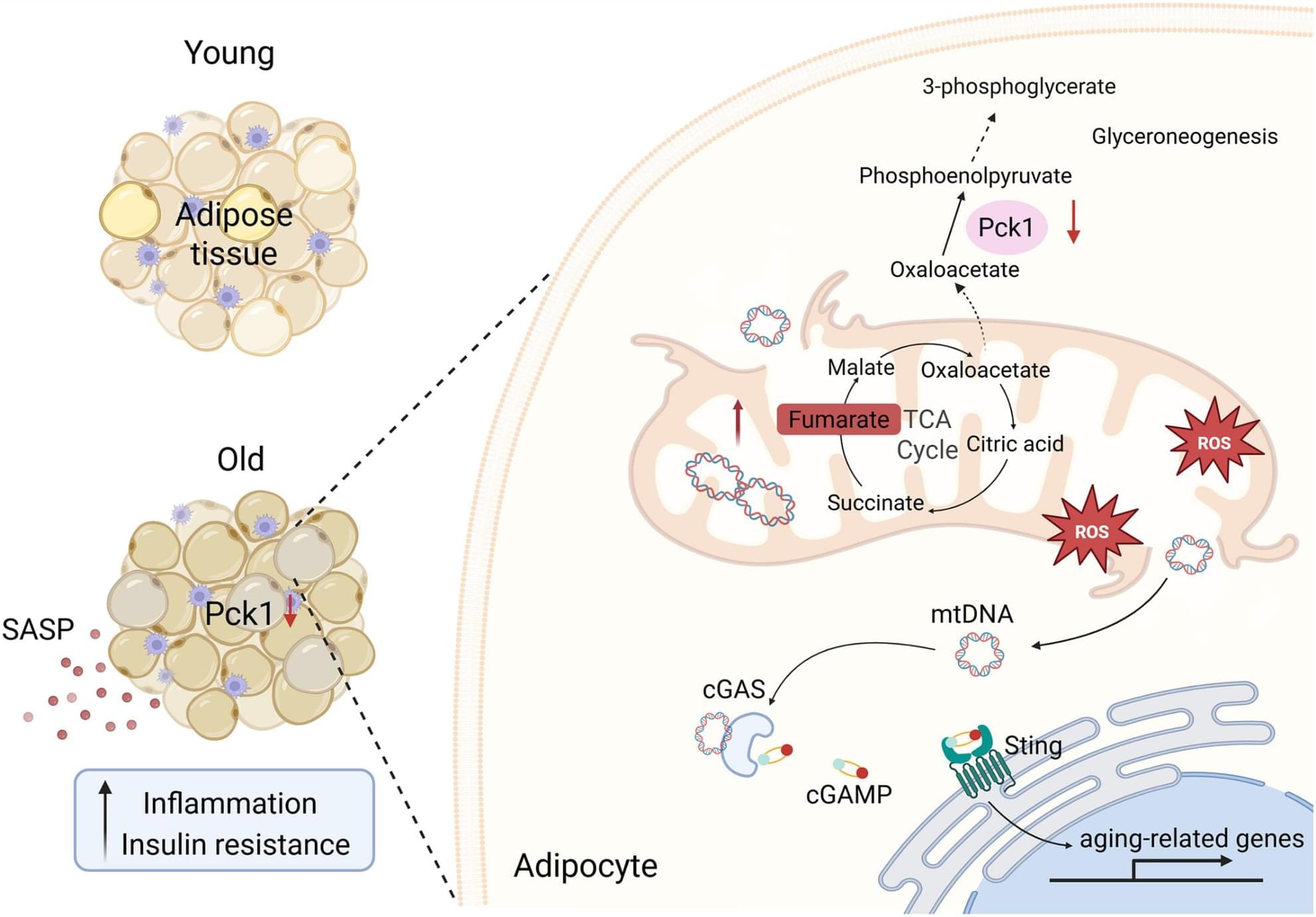
Adipose tissue is one of the most vulnerable tissues during aging, and its dysfunction plays a pivotal role in age-associated physiological impairments (Ou et al. 2022). As the primary energy storage and endocrine organ, adipose tissue serves as a central hub for maintaining energy and metabolic homeostasis (Zwick et al. 2018). Age-related accumulation of the dysfunctional adipose tissue, particularly visceral fat, induces chronic inflammation and insulin resistance, leading to metabolic disorders (Reyes-Farias et al. 2021 ; Zhang, Jiang, et al. 2024). Clearance of senescent cells in white adipose tissue (WAT) has been shown to significantly mitigate aging-related metabolic dysfunction in murine models, highlighting a pivotal role of WAT in organismal aging (Palmer et al. 2019 ; de Oliveira et al. 2025).
Senescent cells undergo metabolic reprogramming, resulting in abnormal accumulation of detrimental metabolites that further reinforce inflammatory and senescence pathways (Maqdasy et al. 2022 ; Dou et al. 2023 ; Zhang, Higgins, et al. 2024). These cells are identified by a combination of molecular features, including elevated senescence-associated β-galactosidase (SA-β-gal) activity, increased expression of cyclin-dependent kinase inhibitors p21 and p16, persistent DNA damage response, and secretion of the senescence-associated secretory phenotype (SASP) (Di Micco et al. 2021). Mitochondrial dysfunction is a critical driver of cellular senescence (Gonzalez-Freire et al. 2015), marked by impaired mitochondrial quality control, heightened oxidative stress, accumulation of mitochondrial DNA damage, and disruption of cytosolic nicotinamide adenine dinucleotide (NAD+ and NADH) balance (de Mello et al. 2018 ; Das et al. 2021). Emerging studies indicate that impaired TCA cycle metabolism reduces mitochondrial membrane potential, exacerbates mitochondrial damage, and contributes to cellular senescence (Kurhaluk 2024 ; Cappel et al. 2019). However, the precise mechanisms linking mitochondrial dysfunction to the onset and progression of cellular senescence and age-related inflammation remain incompletely understood.
Phosphoenolpyruvate carboxykinase 1 (Pck1) is well recognized as a rate-limiting enzyme in hepatic and renal gluconeogenesis; however, in adipose tissue, it predominantly facilitates glyceroneogenesis, playing a pivotal role in modulating mitochondrial-cytosolic carbon flux via TCA cycle cataplerosis. Our prior research demonstrates that Pck1 modulates replicative lifespan in yeast (Yuan et al. 2020), suggesting its potential role in aging processes. In this study, we showed that Pck1 was essential for mitochondrial function and adipose tissue aging. Pck1 deficiency induced premature adipocyte senescence, exacerbated age-related metabolic abnormalities and inflammation in WAT. Mechanistically, Pck1 deficiency impaired TCA cycle catabolism, leading to fumarate accumulation, which further promoted cellular senescence. These findings identify Pck1 as a key metabolic effector that links mitochondrial metabolism in adipocytes to adipocyte aging and inflammation, with potential implications for targeting age-related diseases.