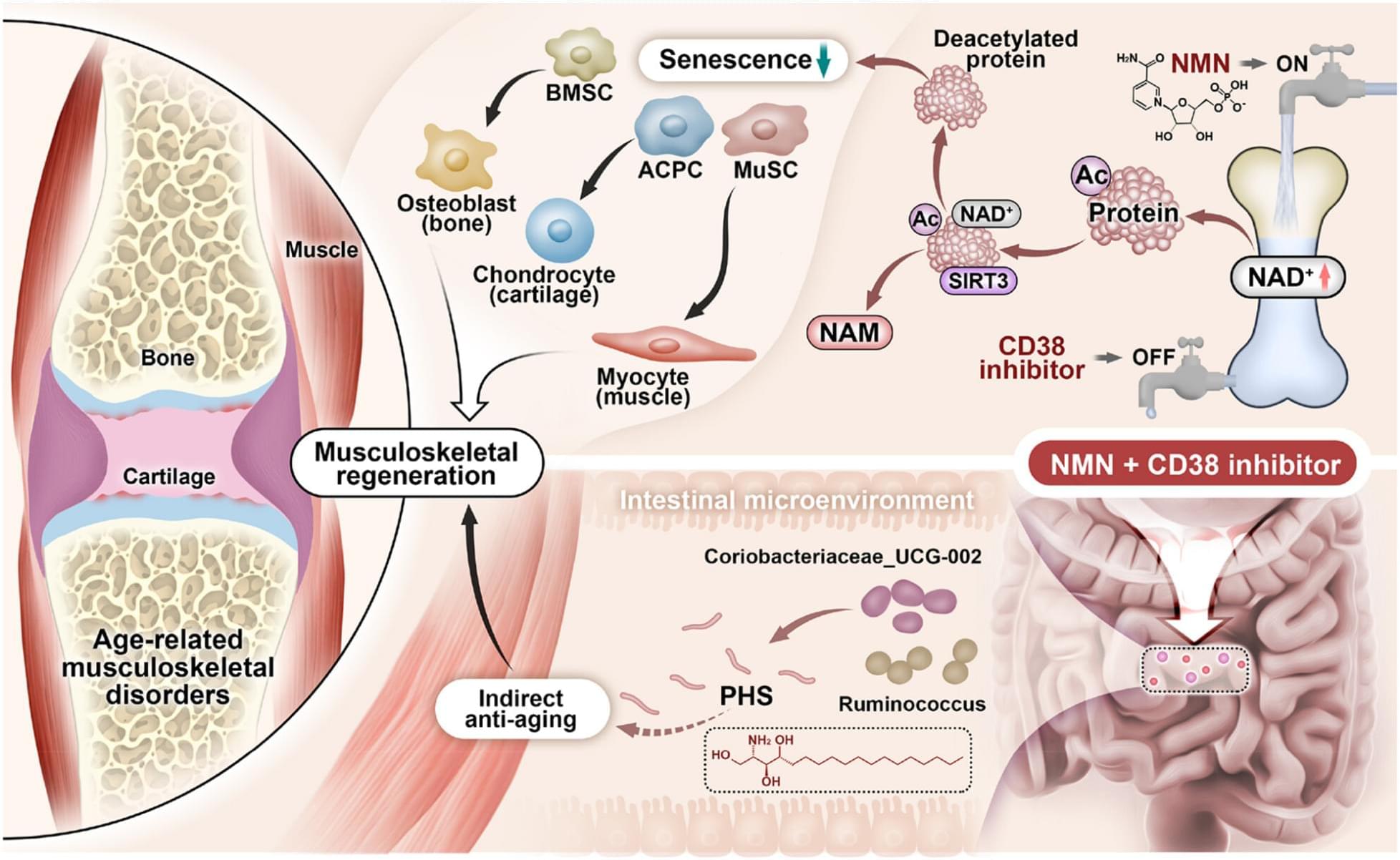
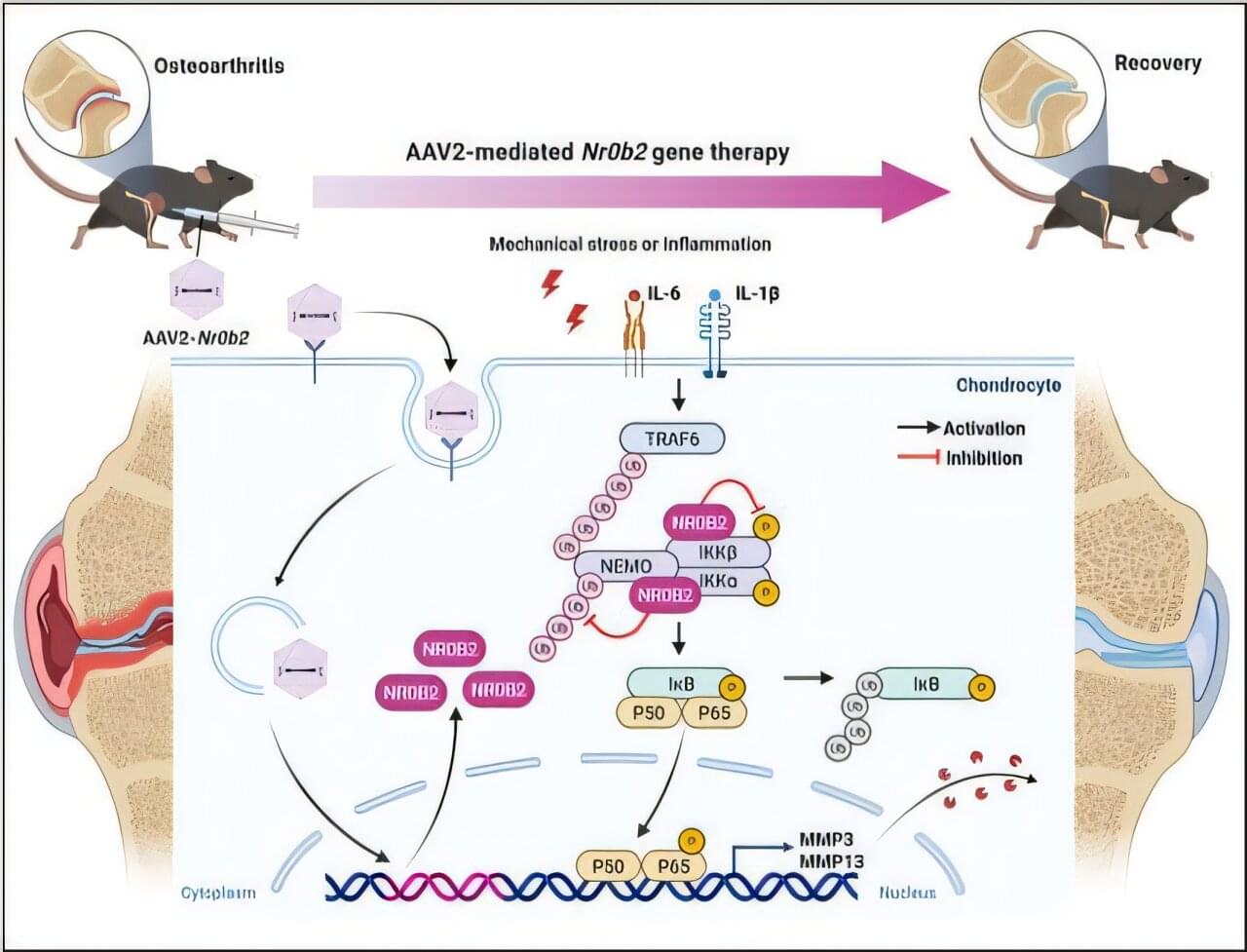
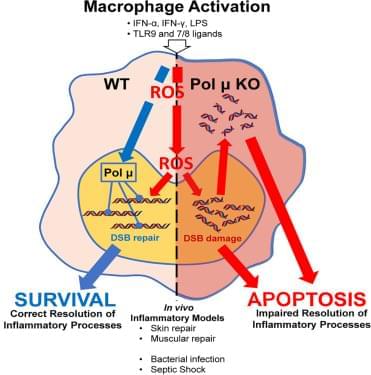
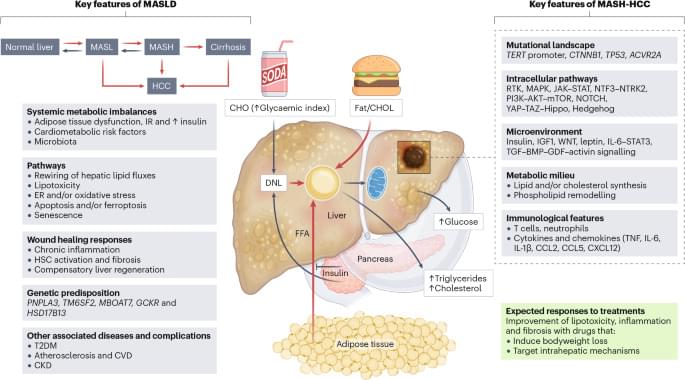
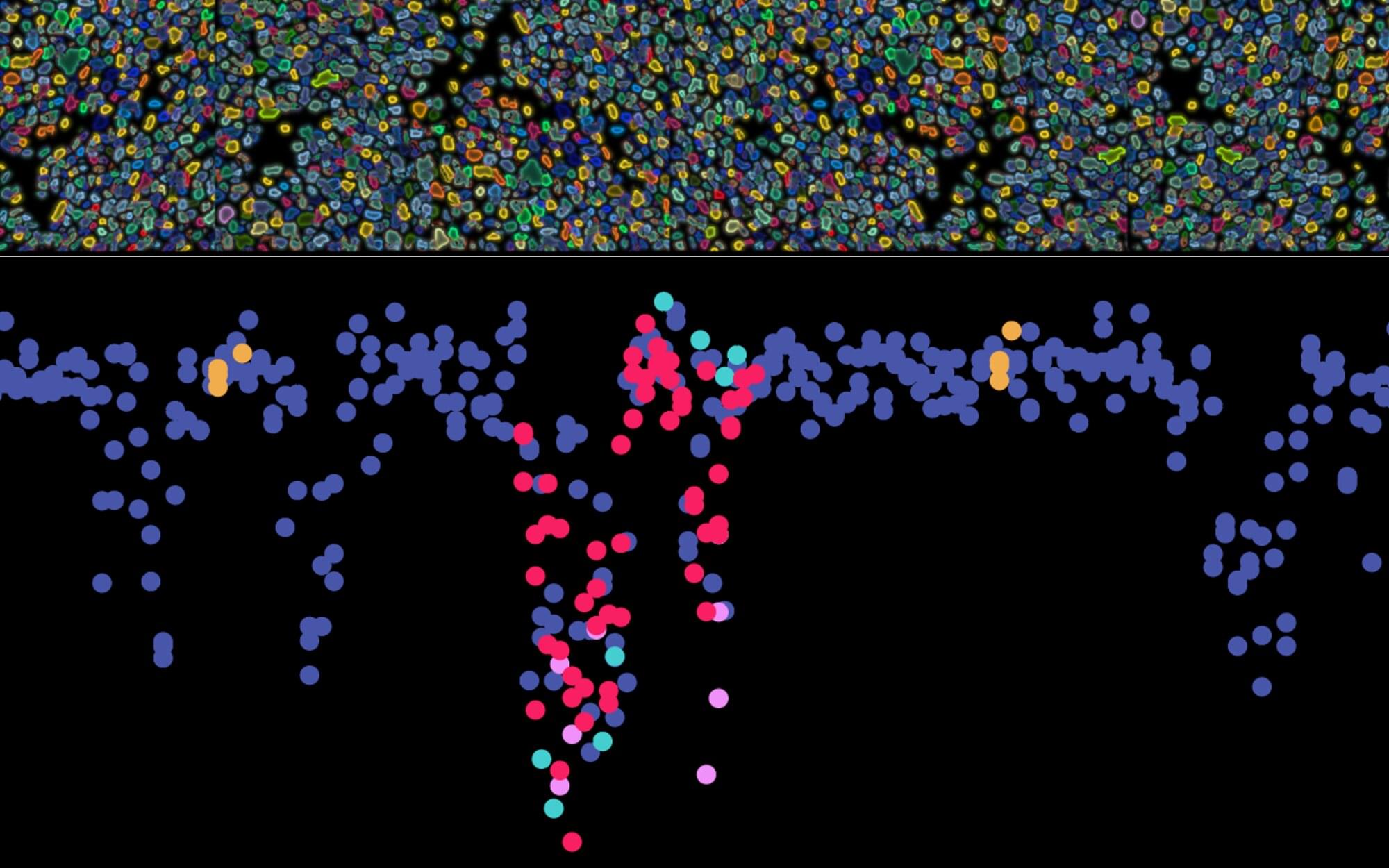
A novel synergistic drug combination (N + A) consisting of an NAD+ precursor (NMN) and an NAD+ consumption (CD38) inhibitor (API) promotes musculoskeletal regeneration in aging. Notably, increased NAD+ serves as a coenzyme for SIRT3, exerting a robust anti-senescence effect, thus promoting tri-lineage differentiation into chondrocytes, osteoblasts, and myocytes. Furthermore, oral administration of the N + A formulation modulated the intestinal microenvironment, promoting the gut microbiota-derived production of the metabolite PHS, thereby exerting indirect anti-aging effects in musculoskeletal disorders.