💬 Editorial: Current evidence supports iatrogenic transmission of cerebral amyloid angiopathy but not AlzheimerDisease; a definitive causal link between contaminated growth hormone exposure and AD remains speculative.
Neurodegenerative diseases caused by protein misfolding (eg, Alzheimer disease [AD], frontotemporal lobar degeneration, Parkinson disease) share many similarities with prion diseases. All demonstrate template-directed protein misfolding and propagation in vivo. However, with 1 exception, they have not exhibited interindividual or zoonotic transmission as observed in iatrogenic Creutzfeldt-Jakob disease and variant Creutzfeldt-Jakob disease, respectively. An important unresolved question is whether other proteinopathies are transmissible between individuals, and if so, their potential impact on public health. To address these concerns, several prion centers have re-assessed cases of iatrogenic Creutzfeldt-Jakob disease due to cadaver-derived human growth hormone (c-hGH) and dura mater grafts. Although amyloid β (Aβ) plaques and cerebral amyloid angiopathy were commonly seen, tau pathology necessary for a diagnosis of AD was not.1,2 Thus, while there is adequate evidence that cerebral amyloid angiopathy may be acquired through iatrogenic mechanisms, iatrogenic transmission of AD pathology remained speculative.3
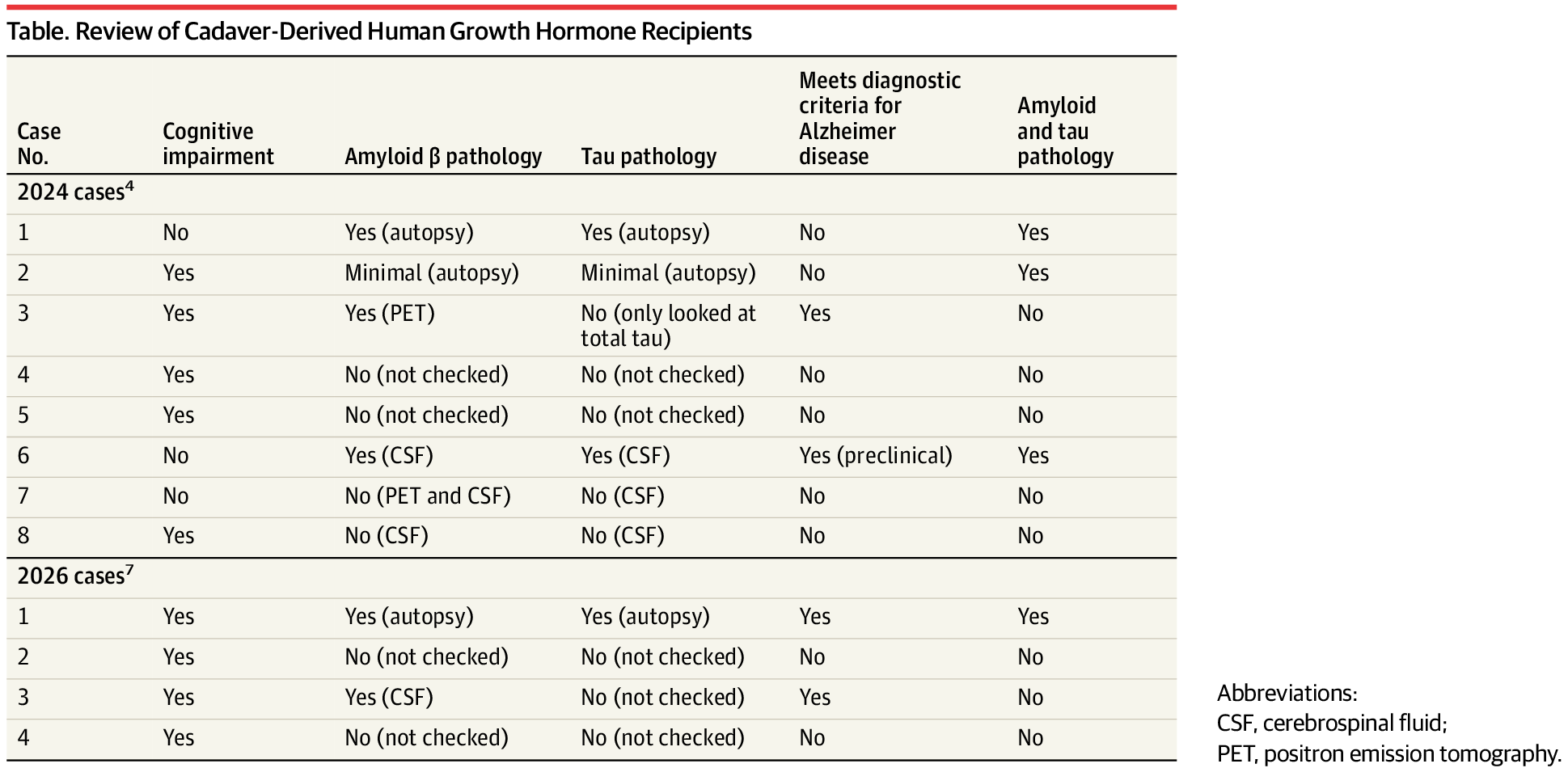
In 2024, Banerjee et al published an article entitled, “Iatrogenic Alzheimer’s Disease in Recipients of Cadaveric Pituitary-Derived Growth Hormone.”4 This assertion of iatrogenic AD (iAD) was largely predicated on the detection of Aβ seeds contaminating the c-hGH used in 8 recipients who later presented with concerns of cognitive impairment. The recipients had a variety of premorbid neurologic conditions that led to the need for hGH, many of which are themselves associated with later-life neuropathology, perhaps most notably radiotherapy and epilepsy. This report was met with some skepticism, given how the cases were diagnosed and the lack of biological evidence to confirm AD pathology in most participants.5,6
In this issue of JAMA Neurol ogy, the same group presents a report of an autopsy-confirmed case of AD in a c-hGH recipient and describes the clinical phenotype of 3 other c-hGH recipients.7 In their autopsy case, they describe cerebral amyloid angiopathy and high-level AD neuropathologic change (A3B3C3), providing the strongest confirmation of an AD diagnosis in their cohort. Additionally, this individual had limited premorbid medical conditions (complex partial seizures) and required hGH due to idiopathic growth hormone deficiency. They describe the clinical presentation as a mixed primary progressive aphasia phenotype and remark that 3 other c-hGH recipients presented similar primary progressive aphasia phenotypes. One of these was diagnosed with atypical AD due to unspecified single-photon emission computed tomography imaging findings and the other through a reduced Aβ42/40-cerebrospinal fluid ratio.