Arm’s new chip could be a powerful but efficient conductor for real-world use of agentic AIs.



Humanity has long regarded intelligence as an ability unique to human beings. The capacities to think, remember, reason, and solve problems were considered central to the human mind itself. To understand language, anticipate the future, and engage in creative thought was believed to belong exclusively to humanity.
Yet today, humanity stands before an entirely new kind of presence.
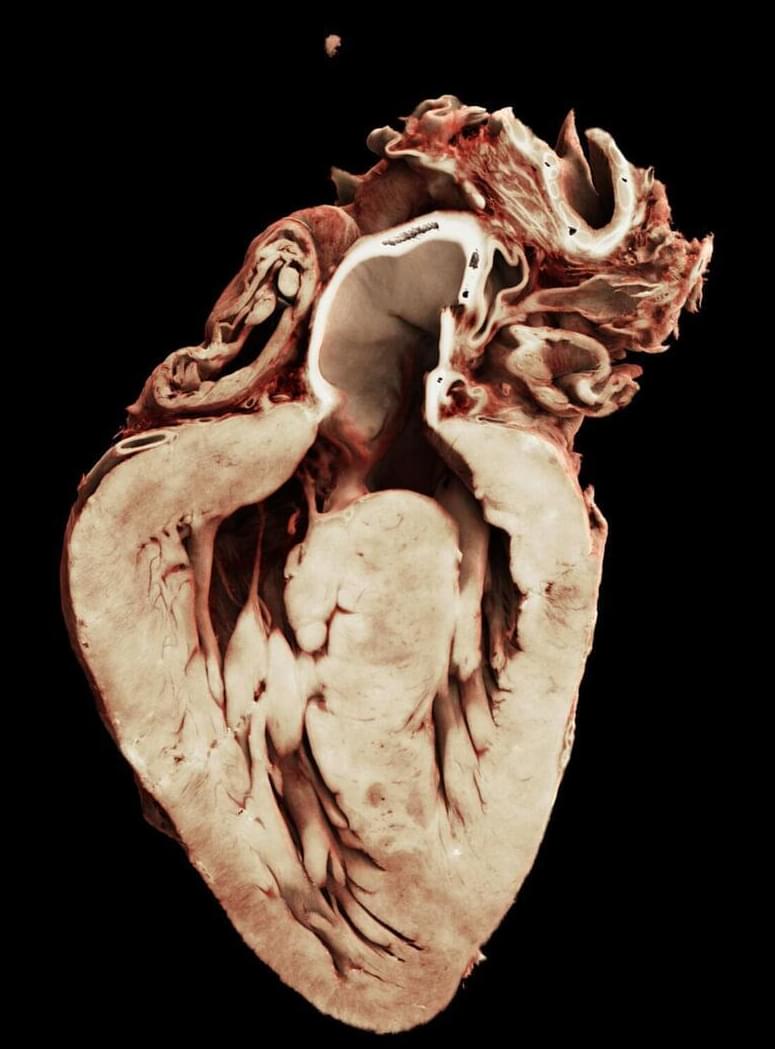
Researchers from UCL (University College London) and the ESRF (The European Synchrotron) have produced the first three-dimensional map of the heart’s electrical wiring in Tetralogy of Fallot, one of the most common congenital heart problems, revealing anatomical features that may explain why many patients develop heart conduction disorders in this condition.
The research, part of the Human Organ Atlas international collaboration, can be used for surgical training and lead to even better outcomes for patients. The research appears in JTCVS Structural and Endovascular.
Congenital heart disease affects around 1% of the population worldwide. In many cases, babies must undergo life-saving heart surgery shortly after birth. Although survival rates are now high, many patients develop complications later in life, particularly abnormal heart rhythms or contraction patterns. Surgeons have long known that these problems can arise when the heart’s delicate electrical conduction system, which is invisible during surgery, is disturbed.
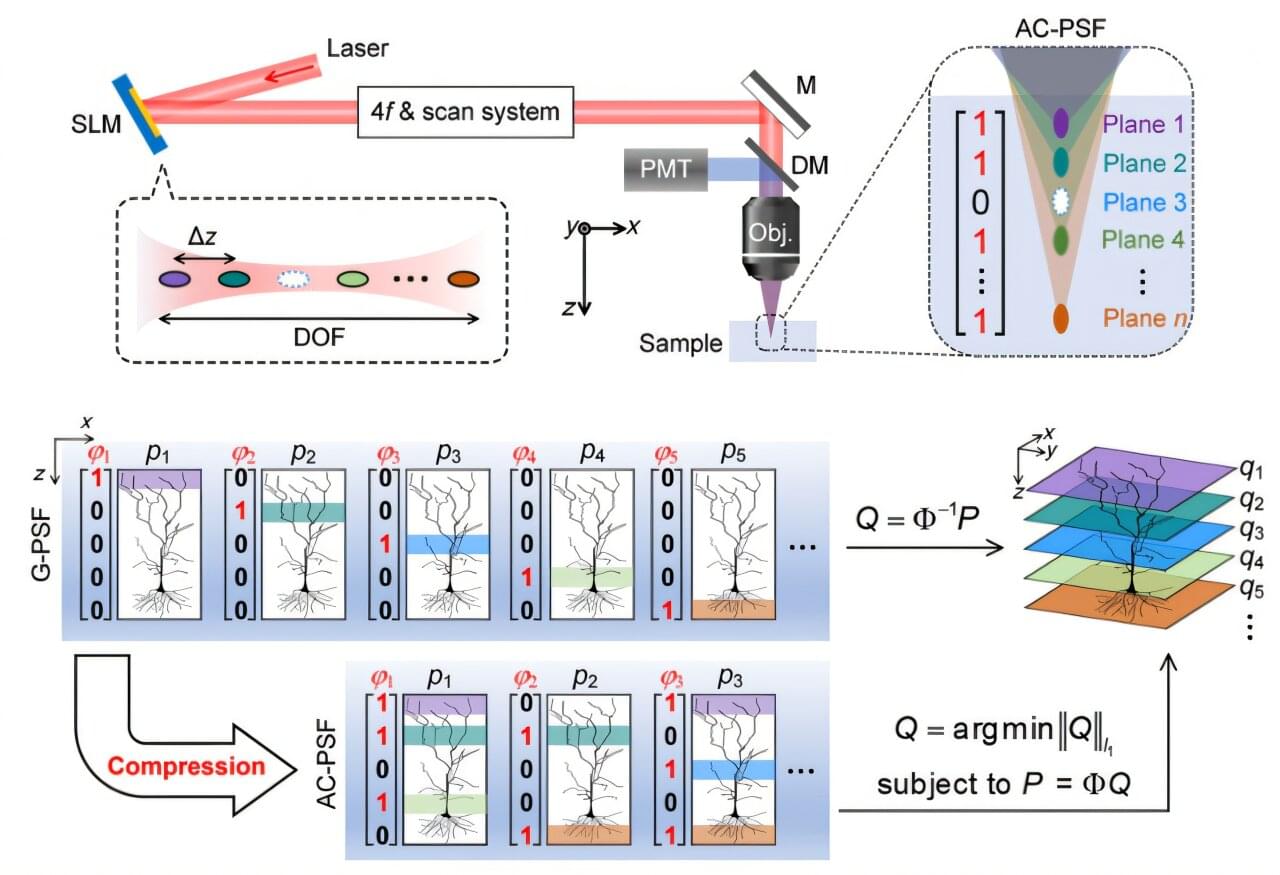
A research team from HKU Engineering has pioneered a fundamentally new imaging strategy known as AIMED (Arbitrary illumination microscopy with encoded depth), which utilizes a sub-sampling approach. By integrating innovations in axial optical encoding with advanced computational image reconstruction, the AIMED technology enables a substantial increase in 3D imaging speed while enhancing photon safety, all with minimal additional system complexity. This breakthrough demonstrates significant advantages across efficiency, image quality, and system compatibility.
This work was conducted by the OMEGA laboratory under the leadership of Professor Kenneth K. Y. Wong of the Department of Electrical and Computer Engineering at the University of Hong Kong (HKU). The study is published in the journal Advanced Photonics.
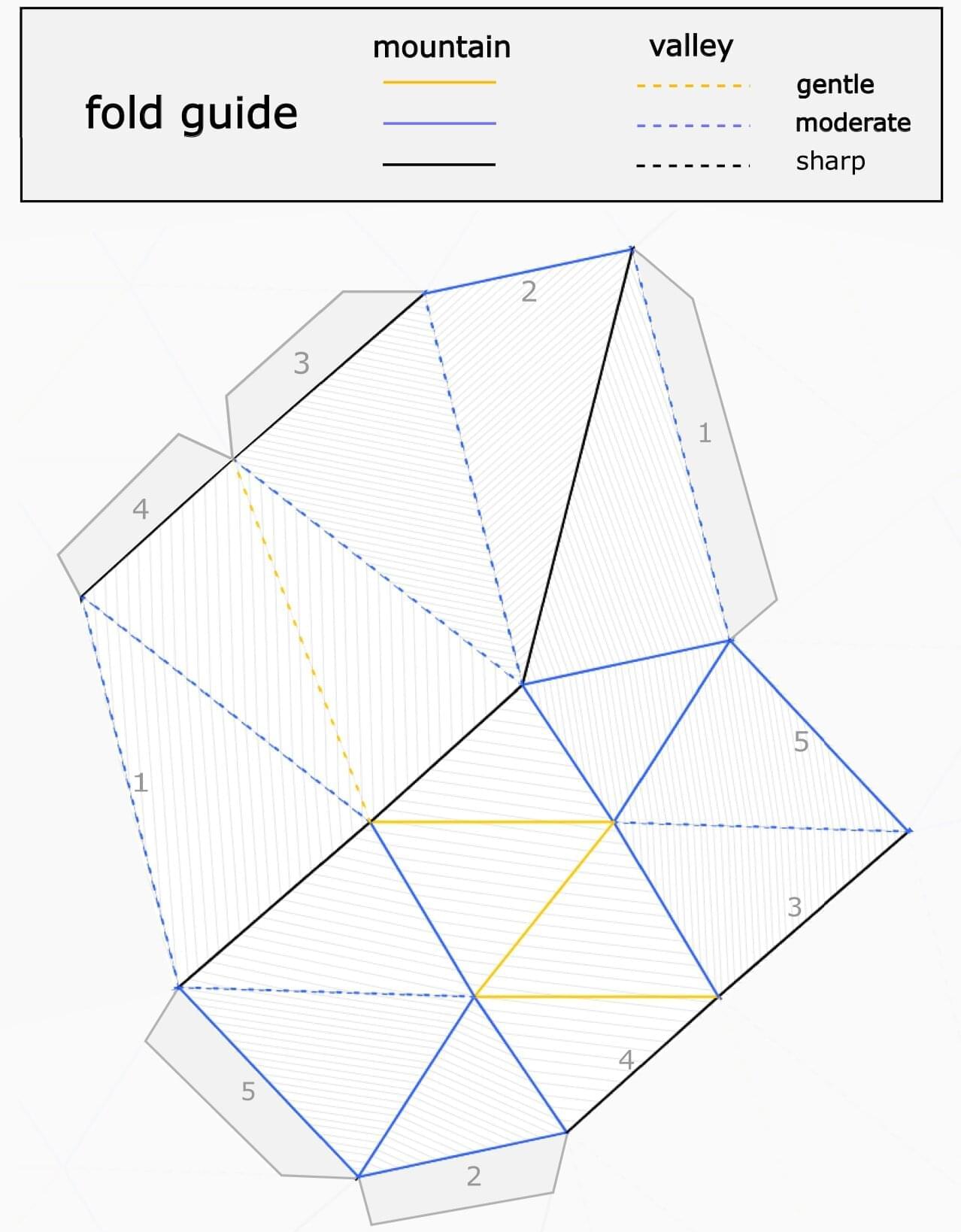
Most people wouldn’t think that it would take rigorous mathematical proof to show how many folds it takes to make a donut shape out of paper. Yet, no one could quite figure it out until recently.
In a new paper, published in Proceedings of the National Academy of Sciences, mathematician Richard Evan Schwartz provides detailed proof of where the line is drawn when it comes to the fewest folds required to construct a torus—the proper name for the shape of a donut—from a piece of paper.

Astronomers may have discovered one of the clearest examples yet of a rare “pair-instability” supernova. It is a catastrophic explosion thought to completely destroy some of the most massive stars in the universe, leaving behind no remnant. The paper outlining the properties of this rare explosion was posted to the arXiv preprint server on May 15.
The event, SN 2023vbw, was first detected by the Zwicky Transient Facility in October 2023 in the outskirts of a small, metal-poor dwarf galaxy about 1.3 billion light-years away. It was tentatively classified as a Type II supernova—the kind produced when a massive star exhausts its nuclear fuel, collapses under gravity, and explodes. But several of its properties refused to fit that picture.
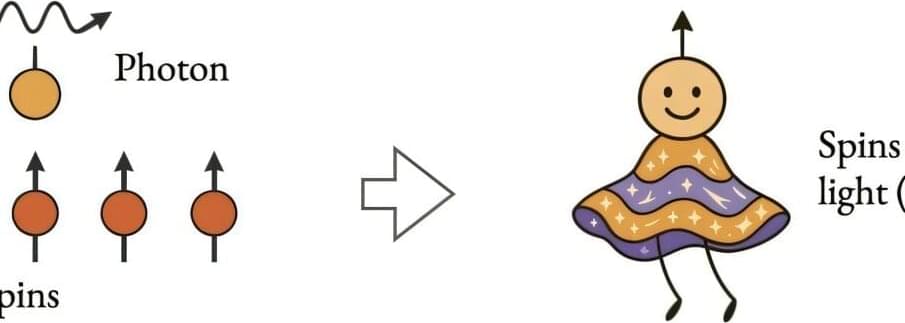
Quantum entanglement is a state in which particles are entwined with each other. In this entwined state, the properties of one particle influence the other, even when they aren’t physically close to each other. This phenomenon has often been observed in small quantum systems with only a few particles in them, where researchers can use it to store and process quantum information. Rice University professor Qimiao Si is interested in understanding and applying quantum entanglement to macroscopic systems with vast numbers of particles.
In a paper recently published in Nature Communications, Si described a method that could lead to not only better understanding of quantum entanglement in quantum materials but also more ready usage of quantum entanglement in macroscopic systems. His theory posits this can be done by coupling quantum materials to quantum light.
“In this theory, by placing matter in a small mirrored cavity and pushing it towards what is called the quantum critical point, we can then introduce photons and induce quantum entanglement in the photon-matter hybrid,” said Si, the Harry C. and Olga K. Wiess Professor of Physics and Astronomy and director of the Extreme Quantum Materials Alliance.

Hackers are ruthless. They can take control of your computer, delete files and disappear without a trace. However, FIU cybersecurity researcher Weidong Zhu has discovered a way to transform a computer’s storage chip into an additional tool for cyber defense. Working with collaborators at the University of Florida, Zhu created a system that makes data on these chips last longer—extending the lifespan of your files in the critical window after your computer is compromised. The work is published in the journal Proceedings of the 2025 ACM SIGSAC Conference on Computer and Communications Security.
“Our system extends recoverable data history up to 126 days,” said Zhu, an assistant professor at FIU’s Knight Foundation School of Computing & Information Sciences whose work is part of the Center for Integrated Security, Privacy, and Trustworthy AI (CIERTA). “Even if your computer is infected, your data can survive on your drive.”
Storage chips, known as solid-state drives (SSDs), have intrigued cybersecurity researchers for years. As hardware—not software—they offer unique safety benefits during an attack.
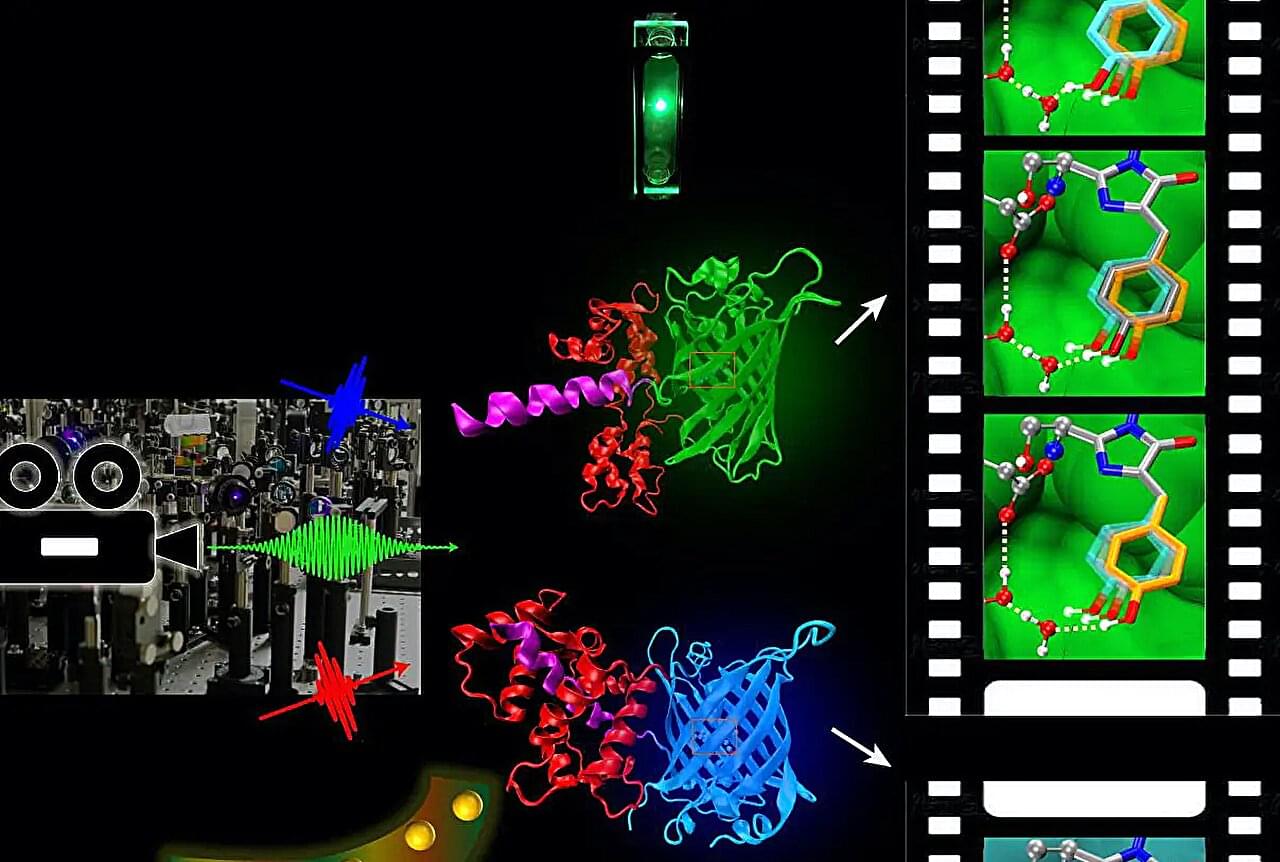
The latest production from the “molecular movie” imaging technology developed at Oregon State University is a new, inexpensive way of dealing with a common environmental pollutant. Based on short-pulse lasers, the imaging technology allows chemical and biological actions to be measured as they are occurring, one high-speed frame at a time.
The measurements occur on the level of the femtosecond—one-millionth of one-billionth of a second. A femtosecond is to a second roughly as a second is to 32 million years.
“We’re able to slow down the observation of chemical processes and understand the exact sequences of biochemical reactions,” said Chong Fang, professor of chemistry at OSU, who unveiled the technology in 2014. “It’s a really powerful tool to study, understand and tune biological processes. Now we have extended the tool set to delineate a wide array of chemical processes.”
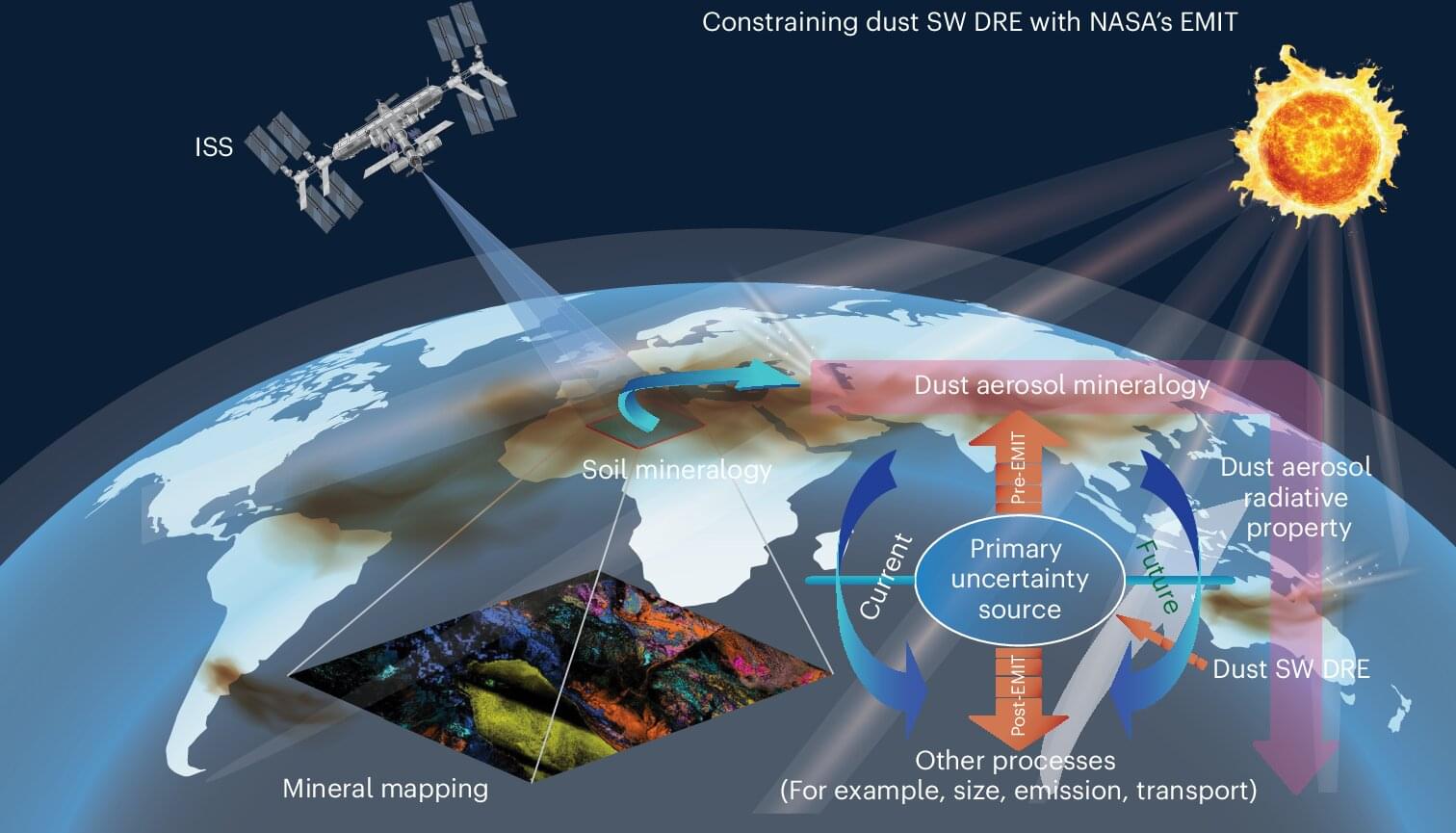
New research from a team of scientists led by Cornell is transforming how researchers understand one of the atmosphere’s most abundant and least understood constituents: mineral dust.
Mineral dust, composed of tiny particles lifted from arid regions including the Sahara, Middle East and East Asia, plays a complex role in Earth’s climate system. These particles both scatter and absorb radiation, influence cloud formation and even fertilize ecosystems. But until recently, scientists lacked reliable global data on the surface soils’ mineral composition, particularly on the prevalence of light-absorbing iron oxides.
Using high-resolution data from a NASA mission aboard the International Space Station, the team has reduced long-standing uncertainty about how airborne dust particles affect Earth’s energy balance through interactions with sunlight. The findings are published in the journal Nature Geoscience.