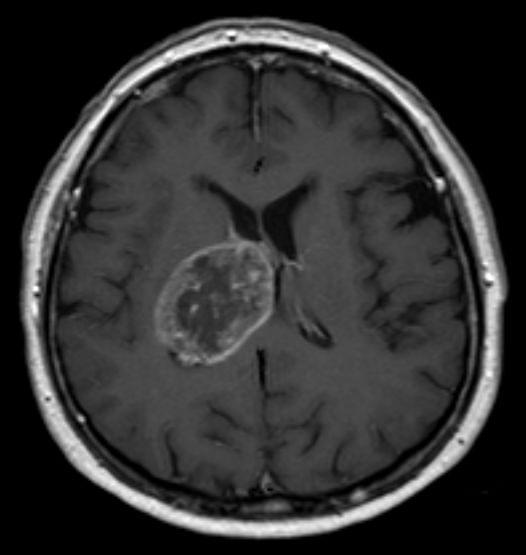
Ruhi Polara, PhD, who led the research alongside Robinson, further commented, “Essentially, CD47 is shielding ROBO2, allowing it to accumulate and drive tumor progression. When we remove CD47, ROBO2 is degraded, and the cancer cells lose their ability to grow and invade effectively.”
The findings reveal a previously unknown molecular pathway—CD47–ITCH–ROBO2—that controls how glioblastoma cells behave. This opens up new possibilities for treatment strategies that go beyond current approaches. While therapies targeting CD47 are already being tested in clinical trials for other cancers, they have shown limited success in glioblastoma so far. The new research suggests that directly targeting the CD47–ROBO2 pathway, or disrupting the stabilisation of ROBO2, could be a more effective strategy. “In summary, our study reveals a role of CD47 in regulating cellular plasticity suggesting that targeting ROBO2 could offer a promising alternative therapeutic strategy for GBM,” they stated.
“By understanding this mechanism, we now have new targets to explore,” Polara said. “This could lead to the development of therapies that specifically block the tumor’s ability to spread, which is one of the biggest challenges in treating glioblastoma.”