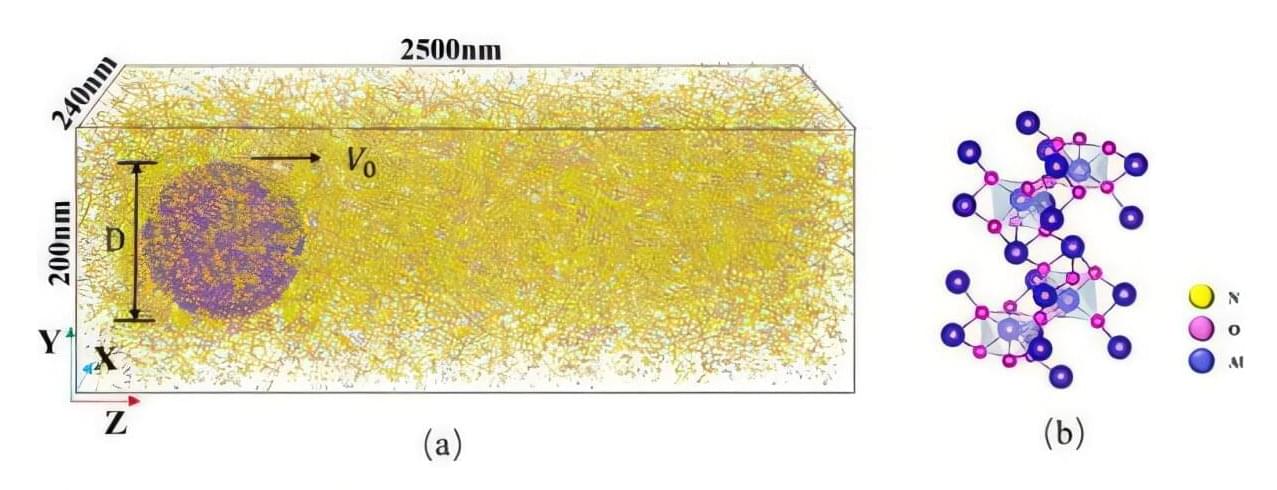
When rockets fire into space, the insides of their engines become an extreme environment where temperatures soar and tiny particles are thrown around at hypersonic speeds. These particles behave in ways that break long-held assumptions, according to new research that could help improve the durability, safety and performance of future space and defense technologies.
The study shows that particles traveling at hypersonic speeds do not remain spherical, instead melting and deforming mid-flight in ways that change how heat, drag and energy move through rocket systems. The findings, published in Physics of Fluids, have led researchers to develop a new drag model that more accurately predicts particle behavior under extreme conditions.
The work was led by researchers from the Southeast University–Monash University Joint Research Institute, Monash University and Shanghai University.