A new approach manufactures many high-quality diamond-based quantum memory chips simultaneously on a single wafer.


Researchers at the University of Bath have discovered a renewable, bio-based polymer membrane capable of efficiently capturing toxic “forever chemicals” from water, offering a potential new route to more sustainable water treatment. The paper is published in the journal ACS Applied Materials & Interfaces.
Perfluorooctanoic acid (PFOA), a member of the per-and polyfluoroalkyl substances (PFAS) family and once commonly used in non-stick coatings, has now been widely detected in water sources worldwide. High levels of exposure have been linked to cancers, hormone disruption, and immune system suppression, with governments around the world taking action to protect people and the environment.
Unlike many conventional water treatment materials that require frequent replacement or generate secondary waste, the new bio-based membrane can trap and hold over 94% of PFOA from water. It can later be treated with heat to remove the trapped pollutants, allowing the polymer to be reused and reprocessed into a new membrane.
A new paper in Genome Biology and Evolution, indicates that while the COVID-19 virus has developed rapidly since 2019, it has done so within limited genetic channels. These genetic limits have remained unchanged. Despite scientists’ earlier fears about dramatic, rapid evolution of the COVID-19 virus, it appears recent changes in the virus were relatively constrained; the virus altered by combining pre-existing mutations. The virus has not expanded the number of genetic routes it can take to evolve.
The paper is titled “Structural constraints acting on the SARS-CoV-2 spike protein reveal limited space for viral adaptation.”
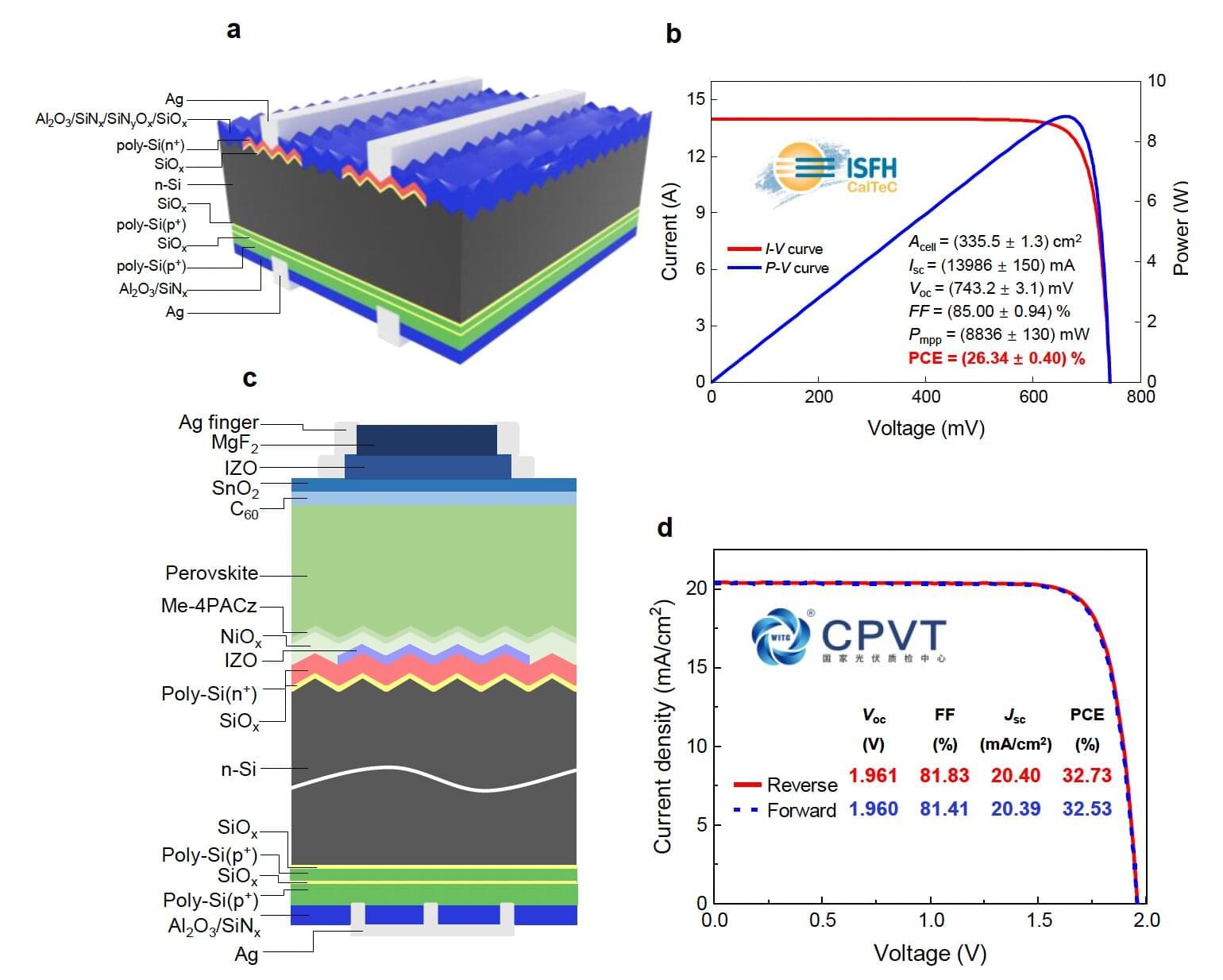
Solar cells, devices that can convert sunlight into electricity, are now widely used in many countries worldwide. Over the past few years, energy engineers have been exploring alternative designs that could further boost these devices’ power conversion efficiencies (PCEs) and ensure that they continue operating reliably over time.
Researchers at Soochow University, Zhejiang Jinko Solar Co. Ltd. and other institutes introduced a new bifacial solar cell design that could overcome some of the limitations of a recently introduced type of solar cell that leverage components known as tunnel oxide passivating contacts (TOPCon). Their design, outlined in a paper published in Nature Energy, combines TOPCon structures with perovskites, a class of materials with a unique crystal structure that efficiently absorbs light.
“Our work is rooted in a fundamental limitation of current TOPCon solar cells,” Kun Gao and Prof. Xinbo Yang, first author and co-senior author of the paper, respectively, told Tech Xplore. “In industrial TOPCon devices, a boron-diffused p+ emitter is still used on the front side, which introduces significant recombination losses and limits further efficiency improvements. A natural strategy is to replace this emitter with localized TOPCon contact.”
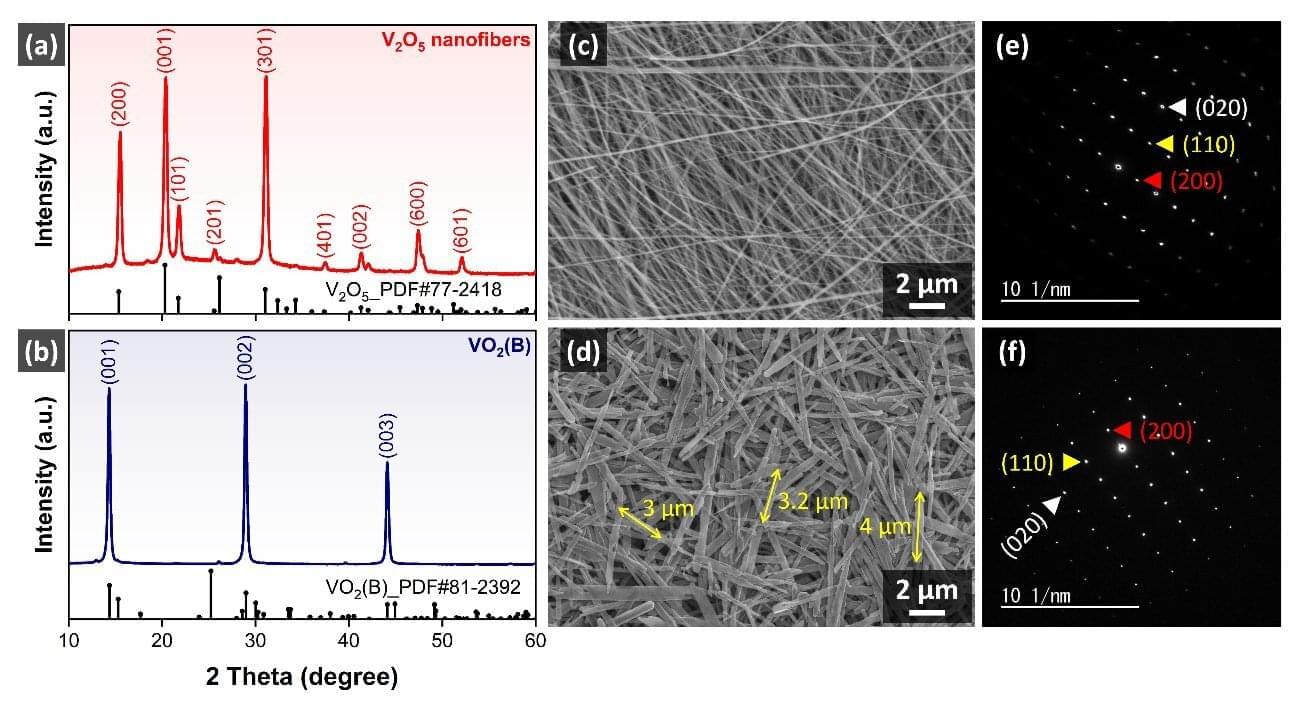
An international research team has successfully synthesized oriented belt-shaped vanadium dioxide (VO2(B)) single crystals via a hydrothermal reduction method, using one-dimensional vanadium pentoxide (V2O5) nanofibers as the starting material. This work, published in the journal ACS Sensors, provides a new material platform and design guidelines for the development of next-generation low-power gas sensors capable of operating at room temperature.
Volatile organic compounds (VOCs) emitted from industrial activities and vehicle exhaust are major urban air pollutants. Because VOCs pose serious environmental and health risks, developing effective monitoring for them is a global concern. Gas sensors can monitor for VOCs, but it has been a major challenge for scientists to develop sensors that work reliably at room temperature. Currently, metal oxide semiconductor gas sensors operate at 200°C–400°C.
“This heating requirement greatly increases power consumption and limits their use in portable devices, battery-powered systems, and large-scale Internet of Things sensor networks,” said Professor Shu Yin from the Institute of Multidisciplinary Research for Advanced Materials (IMRAM), Tohoku University (also affiliated with the Advanced Institute for Materials Research, WPI-AIMR).
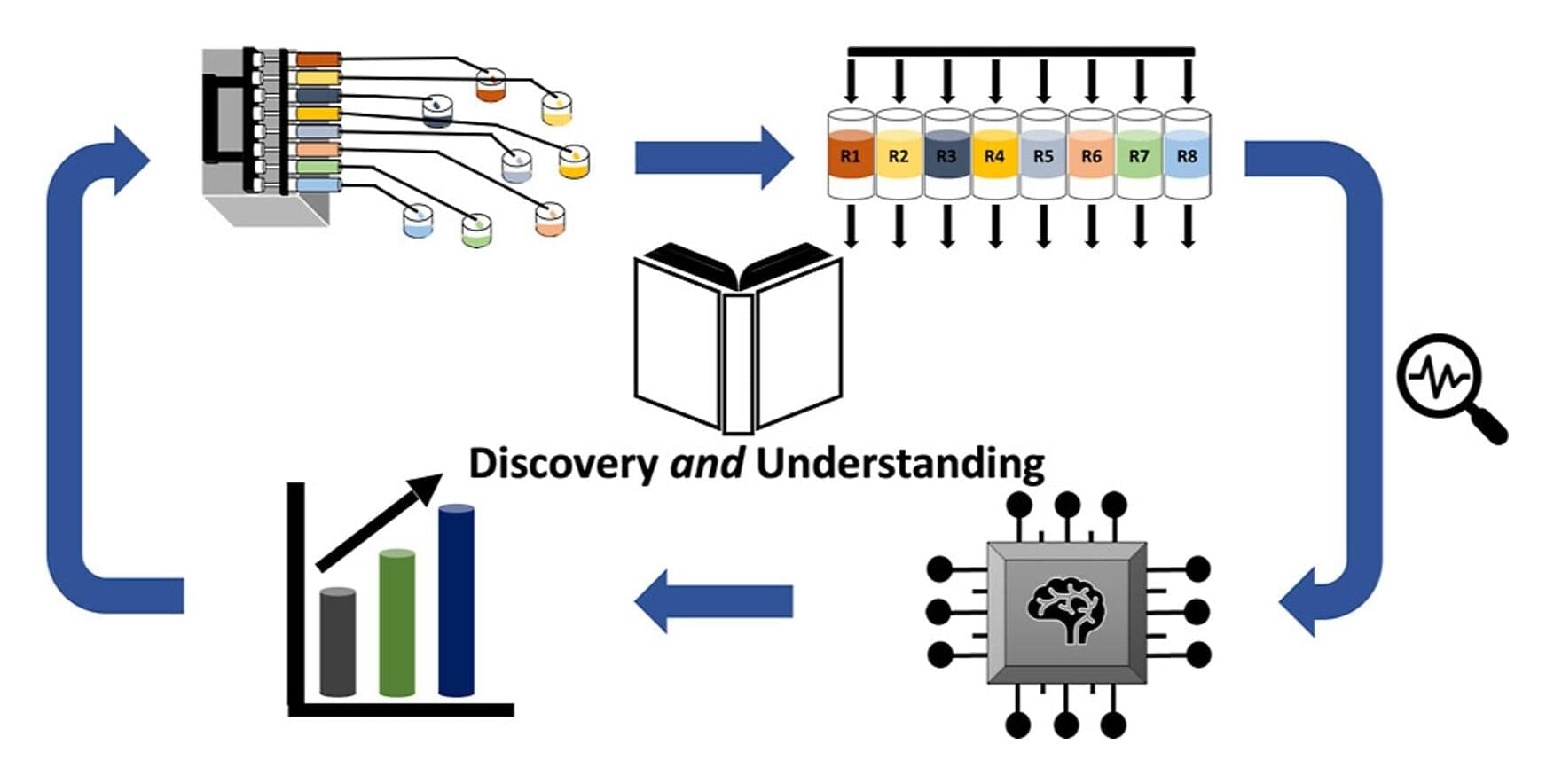
Self-driving laboratories (SDLs) powered by artificial intelligence (AI) are rapidly accelerating materials discovery, but can they also explain their results? Researchers from the Theory Department of the Fritz Haber Institute, in collaboration with BASF, and BasCat—UniCat BASF JointLab, show that they can.
Their new AI-driven strategy works hand-in-hand with SDLs to identify better catalysts while revealing the chemistry behind their performance. The approach was validated on the industrially crucial conversion of propane into propylene.
An SDL integrates an AI doing the experiment planning with lab automation and robotics. In the race to develop better materials, AI and SDLs are often celebrated for one main reason: speed.

You may not be able to hear it, but all solid materials make a sound. In fact, atoms—bound in lattices of chemical bonds—are never silent nor still: Under the placid surface of each and every object in our surroundings, a low hum hovers or a high-energy squeak titters.
As atoms vibrate in their lattices, they do so by either all moving in the same direction, in which case their collective vibration shows up as a low humming sound, or by moving in opposite directions from one another, giving rise to an energetic vibration that registers as a bright squeak or titter.
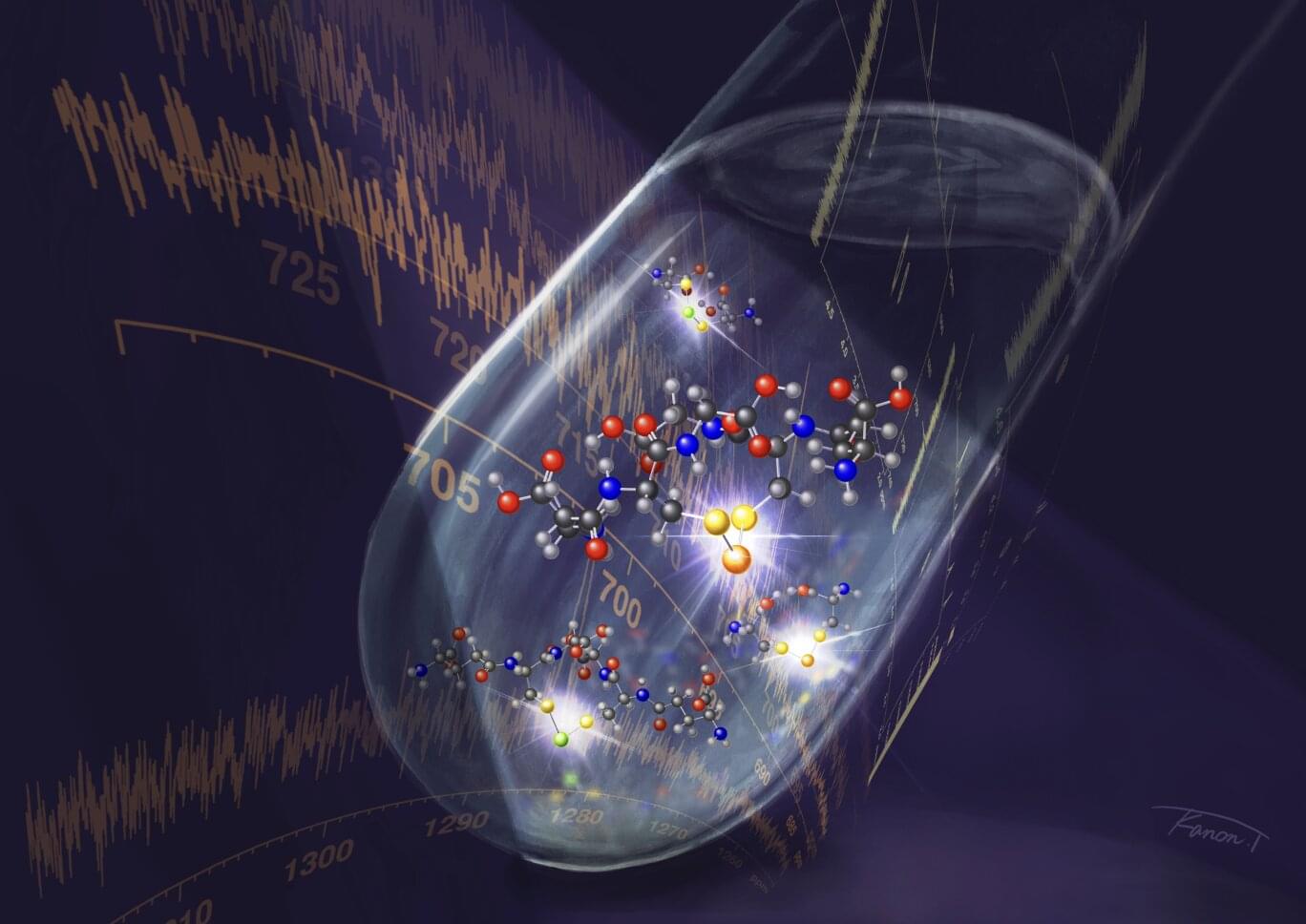
Toward the right side of the periodic table below oxygen, are the chalcogens, or “ore-forming” elements. The chalcogens that occur naturally, including sulfur, selenium and tellurium, are all somehow involved in biological processes. Molecules containing sulfur, like the antioxidant glutathione, play a central role in redox regulation, the balance between oxidation and reduction that is essential for maintaining cellular health.
Recent studies have suggested that the heavier selenium and tellurium are active in biological redox systems as well, but the instability of molecules containing chains of different chalcogen atoms has made structural analysis difficult.
Traditional methods have largely relied on mass spectrometry, which cannot be used to directly observe molecular bonds. This limitation motivated a team of researchers at Kyoto University to develop a method that would allow them to more clearly observe chains of chalcogens. The paper is published in the journal ACS Measurement Science Au.
A team of researchers from Tel Aviv University, in collaboration with colleagues from Japan, has taken an important step toward the next generation of electronics. The scientists achieved highly precise control of the internal structure of graphene—an exceptionally thin and strong material—using a minute, nearly negligible amount of energy.
The study was conducted under the supervision of Prof. Moshe Ben-Shalom of the School of Physics and Astronomy, together with Prof. Michael Urbakh and Prof. Oded Hod of the School of Chemistry. The experiments and calculations were led by Dr. Nirmal Roy and Dr. Pengua Ying, supported by Simon Salleh Atri, Yoav Sharaby, Noam Raab, and Dr. Youngki Yao. The findings were published in the journal Nature Nanotechnology.

Scientists in Geneva took some antiprotons out for a spin—a very delicate one—in a truck, in a never-tried-before test drive that has been deemed a success.
If this so-called antimatter had come into contact with actual matter, even for a fraction of an instant, it would have been annihilated in a quick flash of energy. So experts at the European Organization for Nuclear Research, known as CERN, had to be extra careful when they took 92 antiprotons on the road for a short ride on Tuesday.
The antiprotons were suspended in a vacuum inside a specially designed box and held in place by supercooled magnets.