Scientists have uncovered a previously unknown brain feedback system that links deep sleep, growth hormone release, and wakefulness.



“We are in a time of circulating flesh.”
Stelarc said that to me 13 years ago. In 2026, it reads less like art criticism and more like a status report.
He had grown an ear on his arm. He had hung himself from hooks 25 times. He had let strangers on the internet choreograph his muscles through electrical stimulation, his body remote-controlled across continents.
Most people called it spectacle. I think it was inquiry.
Because long before deepfakes, before voice cloning, before AI agents wearing our faces, was already asking the question we now cannot avoid:
Where does the body end and the network begin?

Scientists may have found the brain’s “pain switch”—and how to turn it off. New research from the University of Colorado Boulder points to a little-known brain circuit that may determine whether short-term pain fades away or becomes a long-lasting problem. The findings suggest that this pathway plays a key role in turning temporary pain into chronic pain that can persist for months or even years.
The study, conducted in animals and published in the Journal of Neuroscience, focused on a region called the caudal granular insular cortex (CGIC). Researchers found that shutting down this circuit can both prevent chronic pain from developing and stop it after it has already begun.
“Our paper used a variety of state-of-the art methods to define the specific brain circuit crucial for deciding for pain to become chronic and telling the spinal cord to carry out this instruction,” said senior author Linda Watkins, distinguished professor of behavioral neuroscience in the College of Arts and Sciences. “If this crucial decision maker is silenced, chronic pain does not occur. If it is already ongoing, chronic pain melts away.”

Remember the early days of AI when a single monthly fee seemed like the ultimate golden ticket? It felt like having a limitless digital brain at our fingertips—until the dreaded usage limit pop-up appeared right in the middle of a critical project. Suddenly, that all-access pass felt more like a restrictive tether, leaving many of us frustrated by hidden caps and invisible throttles just when we needed peak performance the most.
It turns out, we were looking at AI pricing all wrong. Instead of a standard software subscription, artificial intelligence is much more like a utility—a highly measurable resource that actually makes more sense on a pay-as-you-go basis. Imagine a single, centralized workspace where you can seamlessly switch between the biggest powerhouse models on the market for your heavy-duty coding or reasoning, and then route simple summaries to lightning-fast, budget-friendly models.
No more juggling five different logins, and no more getting cut off; just total transparency and control over exactly what you spend.
We are finally entering an era where users hold the reins, and the chaotic days of unpredictable quotas are fading fast. I just published a new piece diving deep into how this shift toward unified, ledger-based AI platforms is completely changing the game for creators, developers, and everyday users alike.
Check out the full article at the link below to explore how this new approach works and why it is exactly the upgrade we have all been waiting for!
Remember late 2022 and early 2023? In tech years, it feels like a lifetime ago. That was when generative AI first exploded onto the scene, and the pricing was brilliantly, beautifully simple. You signed up for a basic flat subscription—usually around $20 a month—and you had the magic of the universe at your fingertips. If you were an enterprise team, maybe you stepped up to a specialized tier. But overall, the premise was the same.
An exploration of human AI versus alien AI and the idea of a galaxy wide data collection network operating at light speed to transfer information on the biology within it.
My Patreon Page:
/ johnmichaelgodier.
My Event Horizon Channel:
/ eventhorizonshow.
Music:

Many plastic products are designed to be used only once, yet the material itself lasts for years. But a new strategy is addressing this problem by creating products that self-destruct on command, known as living plastics. These materials incorporate activatable, plastic-degrading microbes alongside the polymers. One team reporting in ACS Applied Polymer Materials used two bacterial strains that worked together and completely broke down the material within just six days, without making microplastics.
Why scientists are rethinking plastics Zhuojun Dai, a corresponding author on the paper, explains that “the realization that traditional plastics persist for centuries, while many applications, like packaging, are short-lived, led us to ask: Could we build degradation directly into the material’s life cycle?”
Many microbes can break long polymeric chains into smaller pieces using enzymes. Because plastics are polymers, these enzymes or the microbes that make them could be incorporated into living plastics.

In October 2024, the US Department of Energy (DOE) — under the Joe Biden administration — opened applications for funding to support the initial domestic deployment of Generation III+ small modular reactor (SMR) technologies, with up to USD800 million to go to two “first-mover” teams, with an additional USD100 million to address so-called gaps that have hindered plant deployments. According to the solicitation documentation, a Gen III+ SMR is defined as a nuclear fission reactor that uses light water as a coolant and low-enriched uranium fuel, with a single-unit net electrical power output of 50–350 MWe, that maximises factory fabrication approaches, and the same or improved safety, security, and environmental benefits compared with current large nuclear power plant designs.
The solicitation was re-issued by the DOE in March 2025 to better align with President Donald Trump’s agenda on unleashing American energy and AI dominance.
In December last year, the DOE selected Tennessee Valley Authority (TVA) and Holtec Government Services to each receive USD400 million in federal cost-shared funding to support early deployments of advanced light-water small modular reactors in the USA. TVA’s application was selected for funding to accelerate the deployment of a GE Vernova Hitachi BWRX-300 at its Clinch River site in East Tennessee. Holtec plans to deploy two SMR-300 reactors — named Pioneer 1 and 2 — at the Palisades Nuclear Generating Station site in Michigan.
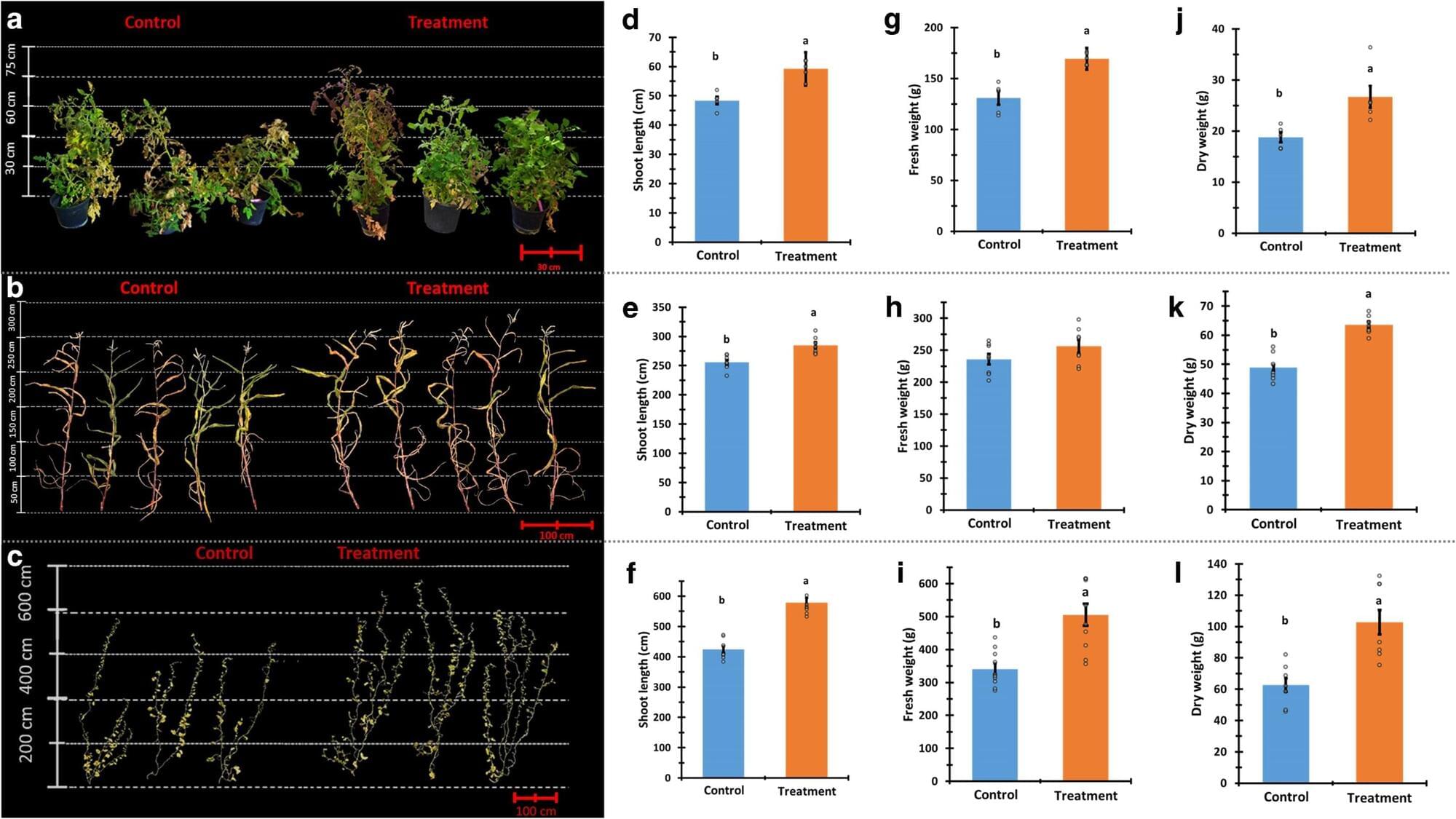
Can we have higher yields and better taste? Using a natural extract from the fungus Pseudozyma aphidis, this method improves the firmness and natural sugar content of crops like tomatoes and melons while significantly boosting production. This discovery offers a practical path to meeting global food demands without compromising the health of the planet or produce quality. Furthermore, because the approach uses stable microbial secretions instead of live cultures, it ensures consistent and reliable performance across various agricultural environments and climates.
Researchers at the Hebrew University of Jerusalem have identified a natural, eco-friendly way to significantly increase agricultural yields while also improving the quality and taste of produce. The study, led by Professor Maggie Levy alongside researchers Anton Fennec and Neta Rotem, focuses on an extract derived from the yeast-like fungus Pseudozyma aphidis.
As the global population continues to grow, the demand for higher agricultural output has historically led to the heavy use of synthetic fertilizers and pesticides. These chemicals often contribute to soil and water pollution and increase greenhouse gas emissions. The new research, published in the journal Plant Physiology, suggests that beneficial micro-organisms can offer a sustainable alternative to these traditional agricultural inputs.
It is hypothesized that environmental contamination by per-and polyfluoroalkyl substances (PFAS) defines a separate planetary boundary and that this boundary has been exceeded. This hypothesis is tested by comparing the levels of four selected perfluoroalkyl acids (PFAAs) (i.e., perfluorooctanesulfonic acid (PFOS), perfluorooctanoic acid (PFOA), perfluorohexanesulfonic acid (PFHxS), and perfluorononanoic acid (PFNA)) in various global environmental media (i.e., rainwater, soils, and surface waters) with recently proposed guideline levels. On the basis of the four PFAAs considered, it is concluded that levels of PFOA and PFOS in rainwater often greatly exceed US Environmental Protection Agency (EPA) Lifetime Drinking Water Health Advisory levels and the sum of the aforementioned four PFAAs (Σ4 PFAS) in rainwater is often above Danish drinking water limit values also based on Σ4 PFAS; levels of PFOS in rainwater are often above Environmental Quality Standard for Inland European Union Surface Water; and atmospheric deposition also leads to global soils being ubiquitously contaminated and to be often above proposed Dutch guideline values. It is, therefore, concluded that the global spread of these four PFAAs in the atmosphere has led to the planetary boundary for chemical pollution being exceeded. Levels of PFAAs in atmospheric deposition are especially poorly reversible because of the high persistence of PFAAs and their ability to continuously cycle in the hydrosphere, including on sea spray aerosols emitted from the oceans. Because of the poor reversibility of environmental exposure to PFAS and their associated effects, it is vitally important that PFAS uses and emissions are rapidly restricted.

Researchers at the São Carlos Institute of Physics at the University of São Paulo (IFSC-USP) in Brazil, led by Paulo Augusto Raymundo-Pereira, have created biodegradable, “wearable” sensors for plants to monitor their health, including the presence of pesticides. The sensors are made from carbon ink and are screen-printed onto transparent cellulose acetate bioplastics.
The study was published in Biosensors and Bioelectronics: X. The World Economic Forum selected wearable sensor engineering as one of the top ten emerging technologies of 2023 for its potential to improve plant health and increase agricultural productivity.
However, most wearable devices today are made from nonrenewable plastic polymers derived from petroleum and have poor adhesion to uneven, wavy, and curved surfaces.