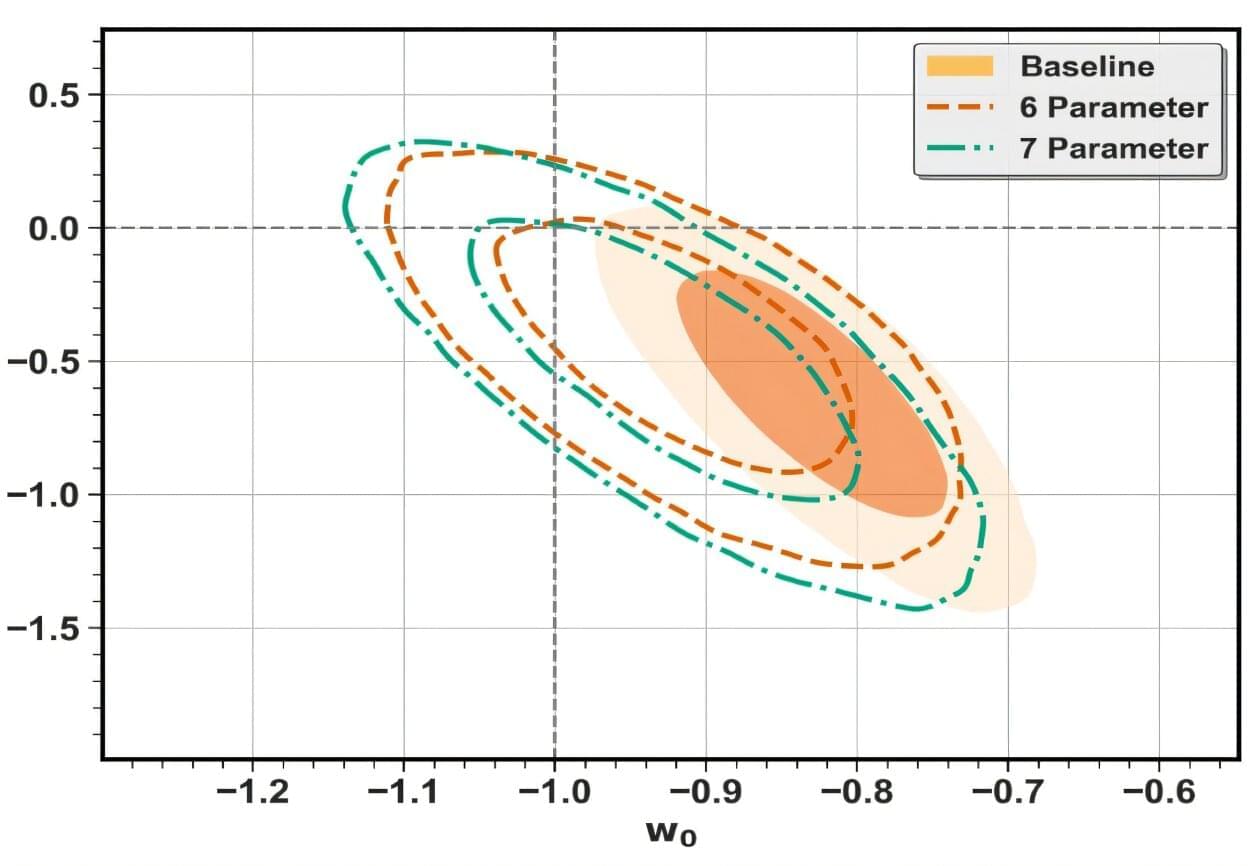
Cosmologists have long struggled to determine whether the universe’s accelerating expansion is being driven by a simple cosmological constant, or whether dark energy’s influence is evolving over time. In a new analysis published in Physical Review D, Samsuzzaman Afroz and Suvodip Mukherjee at the Tata Institute of Fundamental Research, Mumbai, have identified a subtle impact on the inference of the nature of dark energy, due to a tiny mismatch between a fundamental cosmological distance relation and two key datasets used to measure the properties of dark energy.
The result casts fresh doubt on the robustness of the recent claims that dark energy could be evolving over time—perhaps bringing us a step closer to solving one of cosmology’s most enduring challenges.