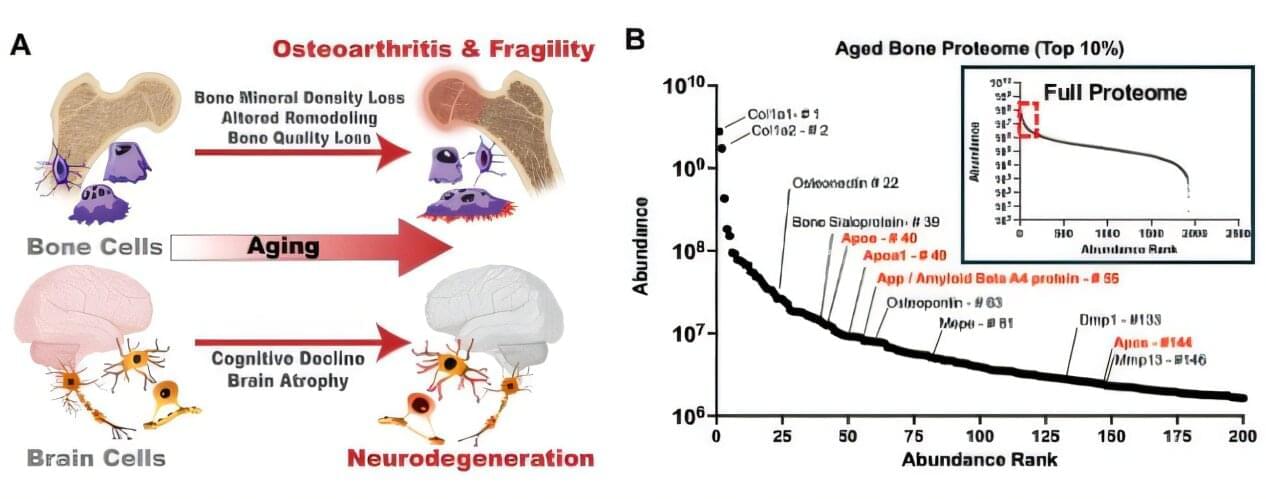
Scientists at the Buck Institute for Research on Aging, along with collaborators at UC San Francisco, have discovered that APOE4, the most common genetic risk factor for Alzheimer’s disease, causes bone quality deficits specifically in female mice, through a mechanism that is invisible to standard imaging and can emerge as early as midlife. The findings, published in Advanced Science, reveal an unexpected biological link between Alzheimer’s risk and skeletal health, and identify a new molecular pathway that could one day inform earlier diagnosis of cognitive decline or guide treatment for bone quality loss in women who carry the APOE4 gene.
“What makes this finding so striking is that bone quality is being compromised at a molecular level that a standard bone scan simply will not catch,” says Buck professor Birgit Schilling, Ph.D., a senior author of the study. “APOE4 is quietly disrupting the very cells responsible for keeping bone strong, and it is doing this specifically in females, which mirrors what we see with Alzheimer’s disease risk.”
Physicians have long observed that people with Alzheimer’s disease suffer bone fractures at higher rates, and that a diagnosis of osteoporosis in women is actually the earliest known predictor of Alzheimer’s. But the underlying mechanism connecting brain and bone health has remained elusive.