Researchers have a better understanding of the nocebo effect and the neuroscience behind it all. Opposite of the better-known placebo effect, where positive expectations trigger genuine pain relief, the nocebo effect is the experience from negative expectations, created by prior experience, verbal suggestion, or social observation, which can drive anxiety and make pain worse.
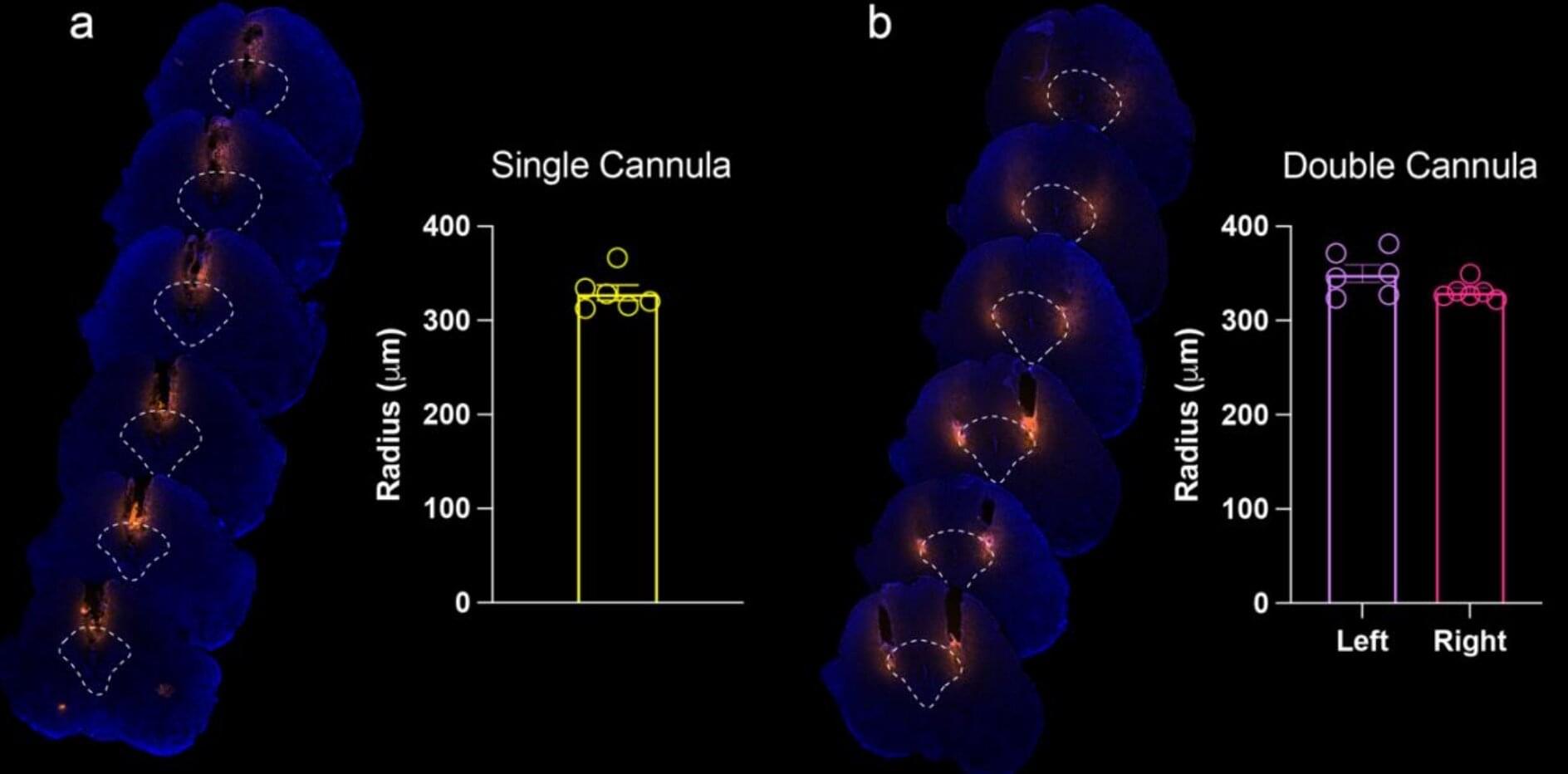
A new study published in Nature Communications, by researchers at the University of Toronto Mississauga and McGill University, identified a brain pathway through which negative expectations can amplify pain. The findings, generated independently by the two labs without prior coordination, converged on the neurochemical cholecystokinin (CCK), which has previously been linked to nocebo pain responses in humans.
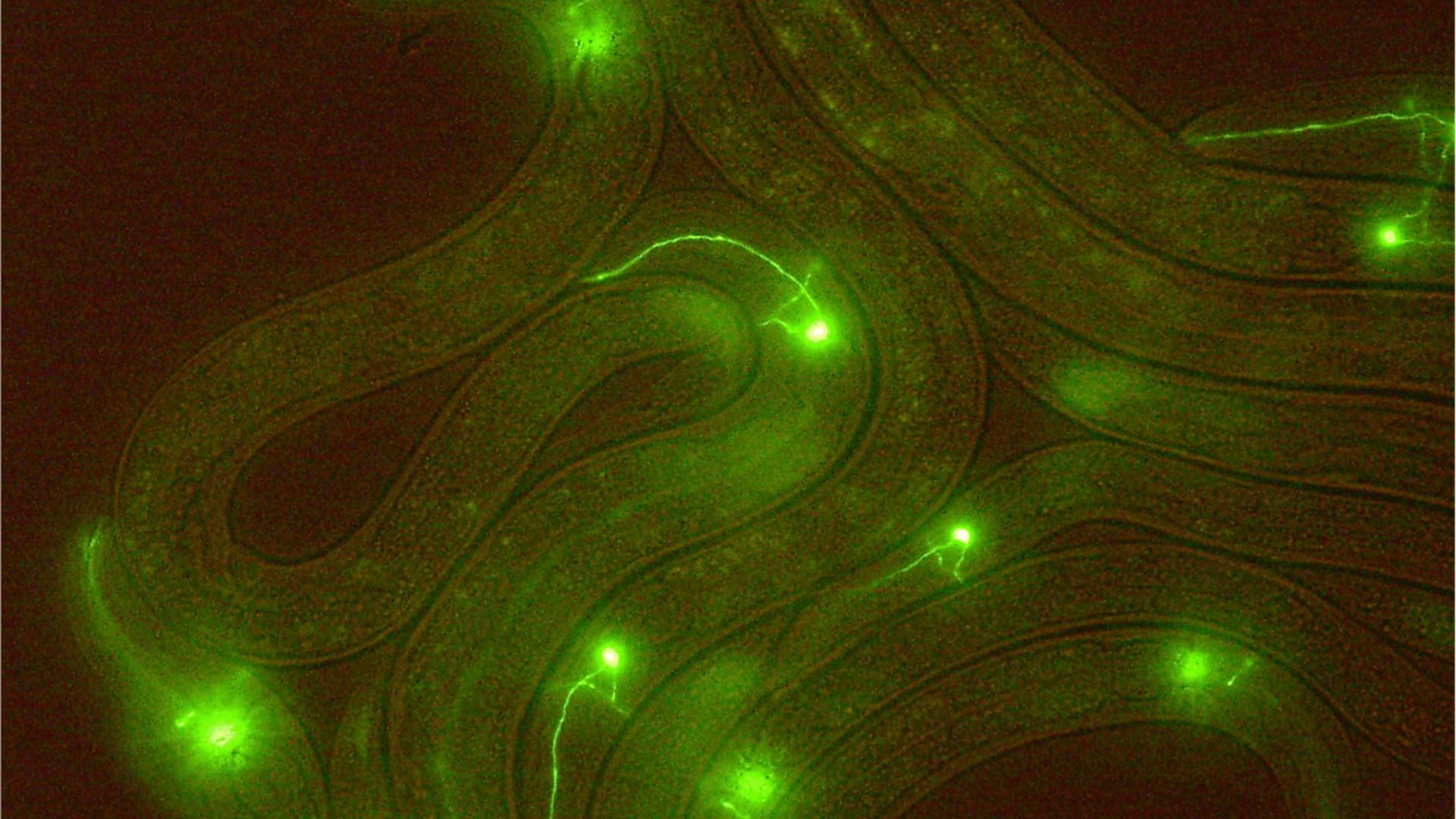
The researchers identified a specific brain pathway through which CCK acts, traveling from the brain’s anterior cingulate cortex (ACC), a region involved in the emotional dimensions of pain, to a midbrain structure called the lateral periaqueductal gray (lPAG), where it increases pain sensitivity.