Six months of high-intensity aerobic exercise preserves the dopamine-producing neurons in the brain, a small pilot study finds.



Roger Penrose and Brian Cox discuss how Roger got interested in physics, the Big Bang, and the role of beauty in mathematics.
Do you agree with Roger’s thoughts on string theory?
With a free trial, you can watch the full conversation NOW at https://iai.tv/video/our-future-theor… the Big Bang to the fabric of spacetime and the nature of consciousness, our core scientific assumptions frame how we understand and perceive reality. But there are many challenges to our current understanding. What if the very foundations of our theories are flawed? Should we reconsider our understanding? And how radically might our view of the universe have to change? Join Roger Penrose, Nobel Prize Laureate and winner of the Wolf prize, in collaboration with Stephen Hawking, with legendary physicist and science communicator, Brian Cox, to explore whether the flaws in our current theories are at some fundamental level insurmountable, or whether they can be extended or changed to overcome these challenges. #physics #cosmology #bigbang Awarded the 2020 Nobel Prize in Physics for his work on black holes, Roger Penrose is a world-renowned mathematician and physicist. In recent years, he has investigated the relationship between physics and the mind, famously arguing that quantum mechanics plays an essential role in solving the mysteries of human consciousness. Penrose has made numerous appearances on media such as BBC, Closer to Truth, and The Joe Rogan Experience. In 1994, he was knighted for his services to science. Famed for his poetic take on the cosmos, physicist and broadcaster Brian Cox has become one of the world’s most recognizable voices in science communication. A former musician turned particle physicist, Cox has played a key role in major experiments at CERN and the Large Hadron Collider, while also captivating millions through BBC series such as Wonders of the Universe, The Planets, and Forces of Nature. Cox has been showered with praise for his contributions, appointed Commander of the Order of the British Empire (CBE), and is the recipient of the Institute of Physics Kelvin Medal and the Michael Faraday Prize. Beyond his work as a Royal Society professor of physics at the University of Manchester, Cox advocates for public scientific literacy and political responsibility in science funding. His style blends rigorous physics with a deep sense of awe — bringing relativity, entropy, and quantum theory into living rooms around the globe. His rare ability to fuse clarity with wonder has earned global acclaim. The Institute of Art and Ideas features videos and articles from cutting edge thinkers discussing the ideas that are shaping the world, from metaphysics to string theory, technology to democracy, aesthetics to genetics. Subscribe today! https://iai.tv/subscribe?utm_source=Y… 0:00 Intro 0:44 Brian Cox on how Roger Penrose inspired him 1:39 — Beauty in mathematics 3:00 — How Roger struggled with maths at school 6:51 — How Roger got interested in physics 9:27 — What theory is best for explaining the beginning of the universe? 12:12 — A key new discovery in cosmology 18:44 — The big bang is not quantum mechanical For debates and talks: https://iai.tv For articles: https://iai.tv/articles For courses: https://iai.tv/iai-academy/courses.
From the Big Bang to the fabric of spacetime and the nature of consciousness, our core scientific assumptions frame how we understand and perceive reality. But there are many challenges to our current understanding. What if the very foundations of our theories are flawed? Should we reconsider our understanding? And how radically might our view of the universe have to change? Join Roger Penrose, Nobel Prize Laureate and winner of the Wolf prize, in collaboration with Stephen Hawking, with legendary physicist and science communicator, Brian Cox, to explore whether the flaws in our current theories are at some fundamental level insurmountable, or whether they can be extended or changed to overcome these challenges.
#physics #cosmology #bigbang.
Awarded the 2020 Nobel Prize in Physics for his work on black holes, Roger Penrose is a world-renowned mathematician and physicist. In recent years, he has investigated the relationship between physics and the mind, famously arguing that quantum mechanics plays an essential role in solving the mysteries of human consciousness.
Join us for a deep-dive conversation with Dr Emil Kendziorra, CEO of Tomorrow.bio, as we explore the cutting edge of cryopreservation and medical biostasis.
Dr Kendziorra, who began his career in cancer research with a summa cum laude degree from the University of Göttingen, shares his remarkable journey from academic science to entrepreneurship, and ultimately to founding Tomorrow.bio — a company he considers his life’s work.
In this interview, he discusses the motivations behind his pivot from traditional longevity research to the frontier of cryomedicine, the operational and emotional lessons learned from cryopreserving over 20 human patients and 10 pets, and his vision for making this technology more accessible in the future.
Tomorrow.bio stands as Europe’s fastest-growing cryopreservation company, with over 800 members across 200+ cities and 45+ countries. The company offers both whole-body cryopreservation and brain-only preservation, using a transparent pricing model designed for long-term value.
With a recent €5 million Seed round and a mission to advance medical biostasis, Emil discusses the unique challenges and opportunities of operating in this space, the ethical considerations of a technology that may not achieve revival for decades, and his long-term vision for the field.
Key Points.
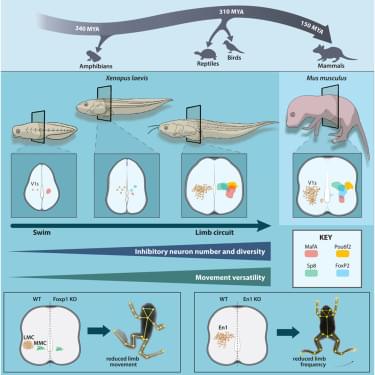
Frog metamorphosis reveals how spinal circuits adapt to new motor demands. Vijatovic et al. demonstrate that the shift from tail-to limb-based locomotion coincides with expansion and diversification of V1 inhibitory neurons. Cross-species comparisons identify a conserved blueprint of tail and limb locomotion, with Engrailed-1 a global regulator of movement frequency.


This is a ~1 hour talk and discussion, comprising part 1 of a conversation with a really interesting young neuroscientist, as well as friend, collaborator, and our Center member, Nicolas Rouleau (https://allencenter.tufts.edu/nicolas… goes over unconventional aspects of neuroscience touching on free will, cybernetics, consciousness, and a lot more. We start a discussion which is continued in part 2. For more information:
Nic’s website: www.rouleaulab.com.
X account: @DrNRouleau.
Recent papers to check out:
Sellar, E.P., Rouleau, N. (In Review). A cybernetic framework for synthetic biological intelligence in the era of neural tissue engineering. Preprint doi: 10.31234/osf.io/md2wf_v1.
Kansala, C., Cicek, E., Nkansah-Okoree, V., Golding, A., Murugan, N.J., Rouleau, N. (In Review). Superstitious conditioning forms the experience of free will under causal determinism. Preprint doi: 10.31234/osf.io/fk3yt_v2.
Roskies, A. \& Rouleau, N. (Forthcoming, In Press). Research on brain organoids should prioritize questions of agency, not consciousness. AJOB Neuroscience.