The emergence of Organoid Intelligence as computing’s answer to the energy wall and the plasticity problem

Neurons, the uber-connected nerve cells that act as a main switchboard for the brain, are central to some incredibly complicated processes. They make it possible to think, walk, speak, and breathe. They even have built-in backup batteries to use in emergencies.
Yet the way individual neurons go about their business is surprisingly simple, according to a new Yale study.
How simple? Most of them operate entirely like tiny on-off switches.

For centuries, we’ve assumed that science has banished the transcendent and established that reality is entirely physical. But critics argue there are signs that a rigorous materialism might be holding science back. Increasingly, “emergence” is used to account for everything from consciousness to spacetime – a convenient placeholder for what materialist science may be unable to explain. Physicists like Heisenberg and Hawking concluded that science gives us models of reality, rather than final descriptions of its true nature, while there are scientists working in everything from biology to computer science who suggest that dualism is a productive metaphysical framework for their research. Materialism may have enabled science to reach beyond the dogmas of religion, but there are now those who are restlessly probing the limits of materialism itself.

The other 99% is used on the activity the brain generates on its own: neurons (nerve cells) firing and signalling to each other regardless of whether you’re thinking hard, watching television, dreaming, or simply closing your eyes.
Even in the brain areas dedicated to vision, the visuals coming in through your eyes shape the activity of your neurons less than this internal ongoing action.
In a paper just published in Psychological Review, we argue that our imagination sculpts the images we see in our mind’s eye by carving into this background brain activity. In fact, imagination may have more to do with the brain activity it silences than with the activity it creates.
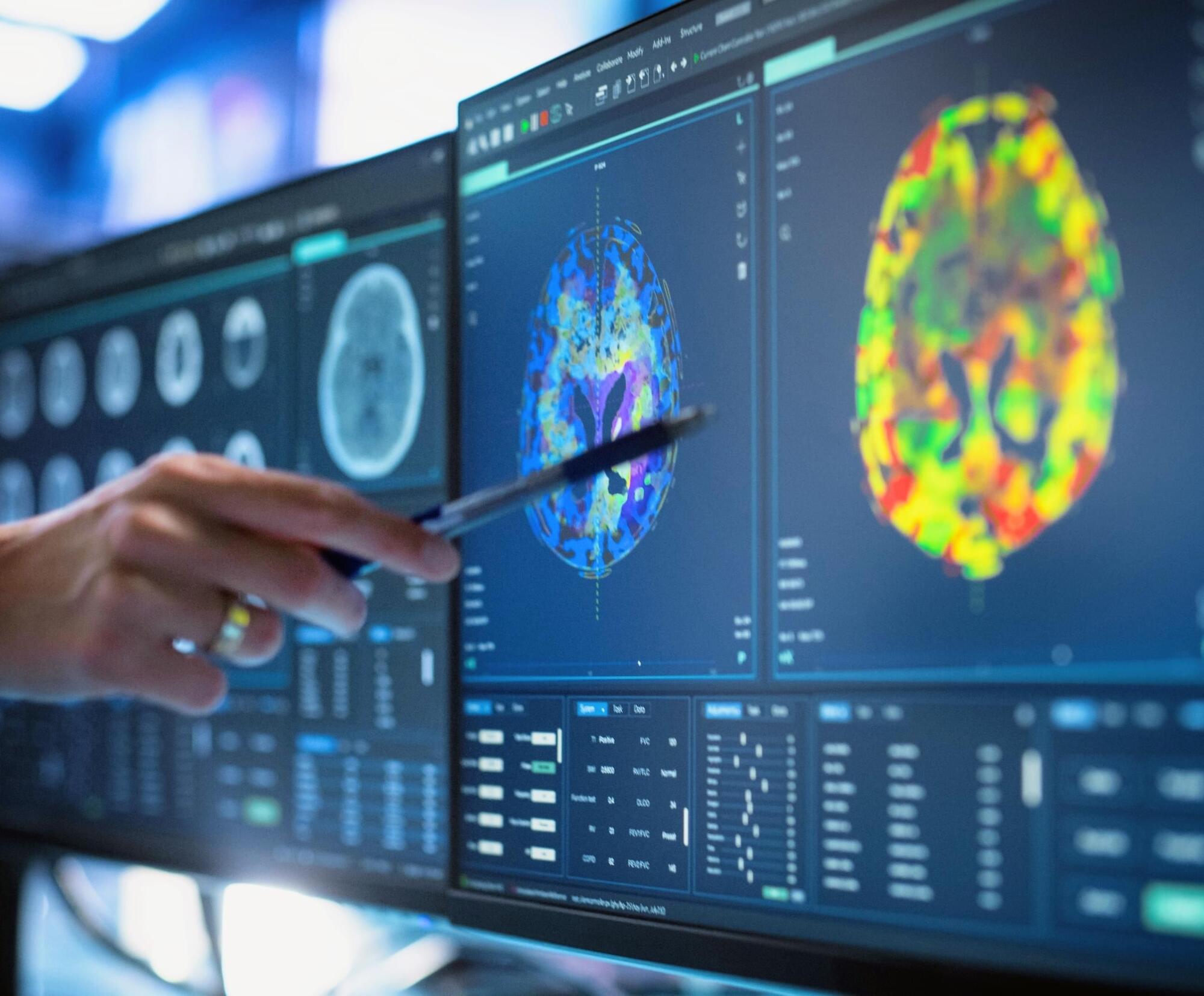
The expected explanation was straightforward: resilient people would respond more strongly to rewards, allowing positive outcomes to outweigh the negatives. But the brain scans told a different story.
In ten prefrontal and parietal regions tied to cognitive control, the resilient group showed stronger increases in activity when negative information appeared.
Their brains were not muting losses. They were engaging more circuitry to handle them.

13 years ago, I walked into Dr. Stuart Hameroff’s operating room with a camera, a microphone, and a single stubborn question:
Is consciousness computation?
Hameroff, an anesthesiologist and professor at the University of Arizona, and co-author with Sir Roger Penrose of the Orch OR theory, said no.
Emphatically. Unfashionably. Against the entire weight of mainstream neuroscience and Silicon Valley orthodoxy.
At the GF2045 conference, where I first met him, Ray Kurzweil went out of his way to declare Orch OR “totally wrong.” Others called it speculative. Untestable. Unscientific.
Today, in the age of large language models, that argument is no longer a niche dispute among philosophers and physicists. It is the decisive question of our century.