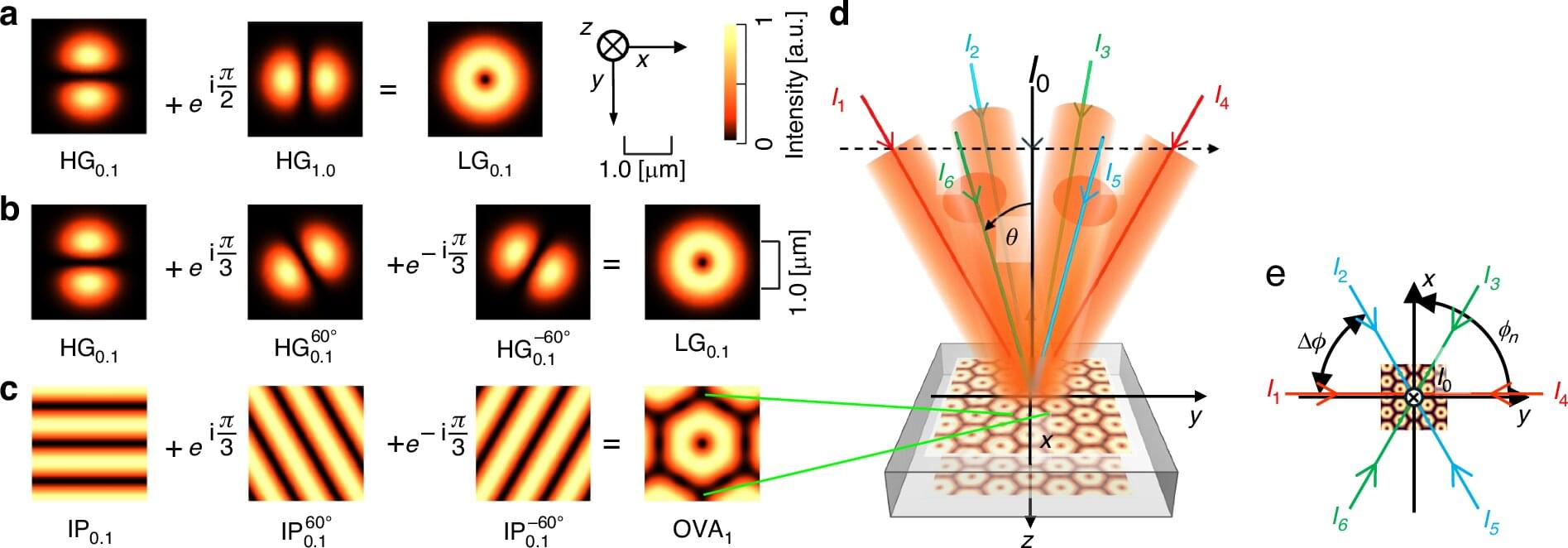
Optical vortices—light beams carrying orbital angular momentum (OAM)—are characterized by helical wavefronts and phase singularities. While they have been widely studied in recent decades, two fundamental limitations have restricted their broader impact: generating large numbers of vortices simultaneously and achieving high peak power in such configurations. Until now, large vortex arrays have been limited to low-power systems, whereas high-power demonstrations have typically involved only single vortices.
In a new paper published in Light: Science & Applications, a research team led by Professor Yoshiki Nakata at The University of Osaka reports the world’s first experimental realization of a megawatt-class large-scale optical vortex array comprising 3,070 phase-coherent vortices at a peak power of 58 megawatts. The result represents more than three orders of magnitude improvement in both vortex number and peak power compared with previous approaches.
Conventionally, Laguerre–Gaussian (LG) modes are expressed as the superposition of two Hermite–Gaussian (HG) modes with a π/2 phase shift. This constitutes the first revision of the HG–LG mode-conversion framework in three decades. The team reformulated this description into a three-mode representation that naturally integrates with multibeam interference geometry.