Heterogeneous catalysis—in which catalysts and reactants are of different phases, e.g., solid and gas—is important to many industrial processes and often involves solid metal as the catalyst. Ammonia synthesis, catalytic converters for automobile exhaust, methanol synthesis, carbon dioxide reduction, and hydrogen production are examples of such metal-catalyzed heterogeneous catalysis.
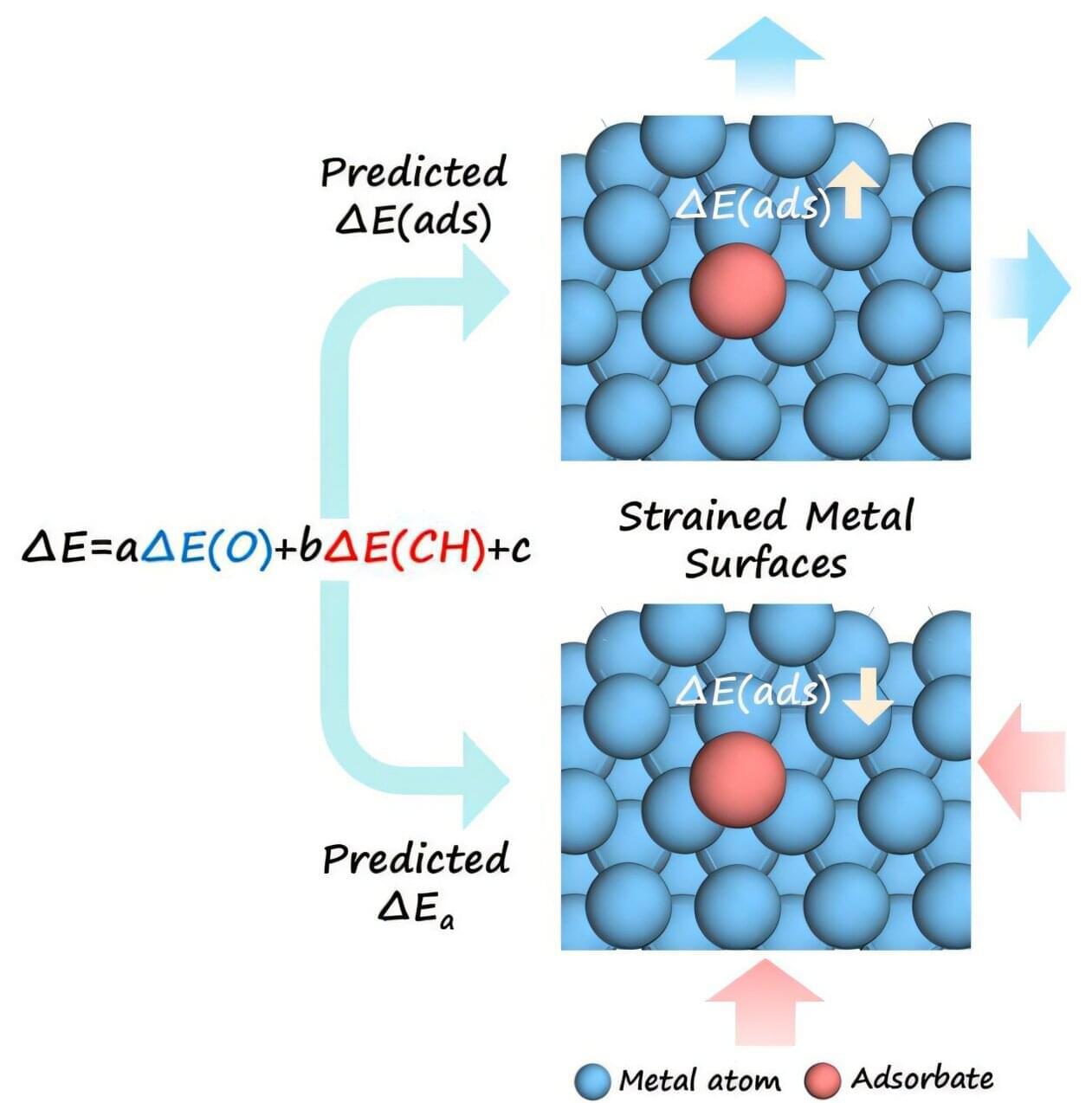
The electronic structure of metal surfaces governs the adsorption of reactants and intermediates, and thus the catalytic activity. For this reason, strain engineering —which tunes the electronic structure of a metal catalyst by stretching or compressing its crystal lattice—has emerged as an important strategy for enhancing catalytic performance. Unfortunately, scientists have not been able to quantify how metal strain influences adsorption energies and reaction barriers across different metal catalysts, thereby limiting the rational design of catalysts with desired properties.
To address this challenge, a research team from the Lanzhou Institute of Chemical Physics (LICP) of the Chinese Academy of Sciences has developed a method to predict how strain modifies adsorption energies and reaction barriers across diverse metal systems. The study is published in the journal Cell Reports Physical Science.