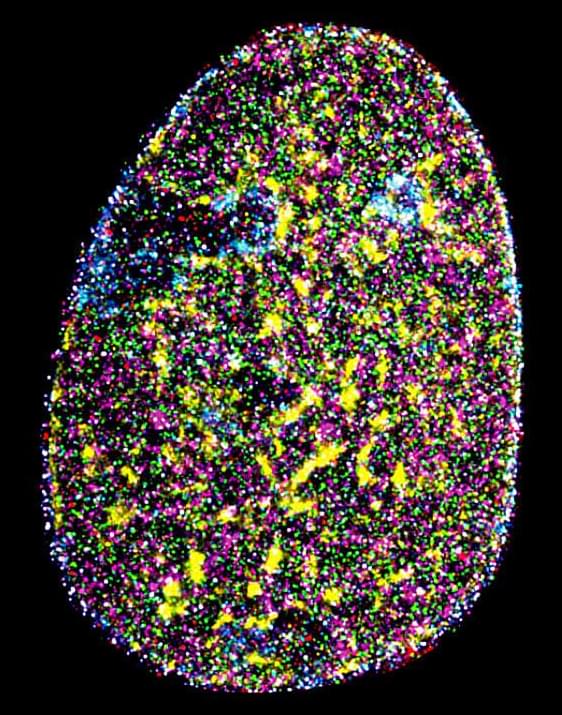
Researchers at the Indian Institute of Science (IISc) have implemented an advanced microscopy technique to visualize multiple biomolecules inside the nucleus of a cancer cell simultaneously at incredibly high resolution. The biomolecules they visualized include critical components of the cell’s transcription machinery and proteins that provide structural support to the nucleus—providing one of the first detailed maps of nuclear organization.
The human body is composed of trillions of cells. Each cell is an intricately organized meshwork of millions of proteins, nucleic acids, and many other molecules vital for the cell’s health. “Building novel technologies to visualize many biomolecules in individual cells is crucial to push the boundaries of biological research,” says Mahipal Ganji, Assistant Professor at the Department of Biochemistry (BC) and corresponding author of the study published in Nature Communications. Conventional imaging techniques, however, allow scientists to visualize only two or three biomolecules in each cell at a time.
In the study, the researchers turned to a microscopy technique called DNA-Points Accumulation for Imaging in Nanoscale Topography (DNA-PAINT), which allows for the visualization of biomolecules inside cells at incredible detail—far beyond the limits of conventional microscopes. It uses small fluorescent DNA fragments or tags that briefly attach to specific targets inside the cell and light up like tiny, blinking signals when a laser beam is shined on them.