Targeting the neuro-immuno-tumor axis in GI cancer👇
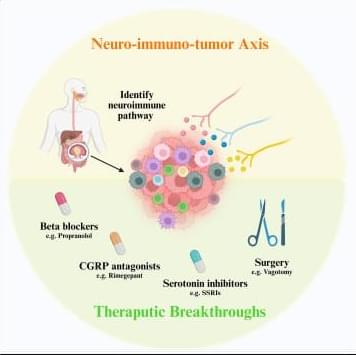
✅Recent advances in cancer biology highlight the neuro-immuno-tumor axis as a critical regulatory network within the gastrointestinal (GI) tumor microenvironment (TME). Tumors are closely innervated, and neural signals actively shape immune cell behavior, influencing disease progression and therapeutic response.
✅A key first step toward translational impact is the identification of dominant neuroimmune pathways operating in GI cancers. Understanding how neural inputs interact with tumor cells and infiltrating immune populations provides a mechanistic framework for disrupting pro-tumor signaling circuits.
✅Pharmacological strategies offer promising opportunities for therapeutic repurposing. β-blockers such as propranolol can attenuate stress-related adrenergic signaling, while CGRP antagonists like rimegepant target nociceptor-derived immunosuppressive cues. In parallel, serotonin inhibitors, including selective serotonin reuptake inhibitors (SSRIs), may modulate enteric and immune signaling to rebalance anti-tumor immunity.
✅Beyond drug-based approaches, surgical interventions such as vagotomy illustrate how physical disruption of neural inputs can reshape the tumor ecosystem. These strategies underscore the concept that nerves are not passive bystanders, but active drivers of tumor–immune interactions.
✅Together, these insights position the neuro-immuno-tumor axis as a powerful and underexplored therapeutic target. By integrating neurobiology with cancer immunology, future treatments may unlock more effective and durable anti-tumor responses in GI cancers.