A new international study co-led by investigators from Mass General Brigham and the Eye & ENT Hospital of Fudan University shows that a gene therapy for a rare form of genetic deafness successfully restored hearing in most participants, with results lasting up to 2.5 years. The results, the largest clinical trial of gene therapy for inherited hearing loss to date and the longest follow-up reported so far, are published in Nature. According to the authors, these latest findings reinforce earlier trials that show gene therapy can be used to treat some forms of inherited deafness, helping guide future research and care.
“It’s remarkable to see patients go from complete deafness to being able to hear,” said the study’s corresponding author, Zheng-Yi Chen, DPhil, the Ines and Fredrick Yeatts Chair in Otolaryngology and an associate scientist at Mass Eye and Ear, a member of the Mass General Brigham healthcare system. “For many patients, that also means the ability to develop and use speech.”
Genetic mutations account for up to 60% of hearing loss present at birth. In this study, researchers used a gene therapy they developed to treat autosomal recessive deafness 9 (DFNB9), caused by mutations in the OTOF gene. The OTOF gene provides the body with instructions to make a protein called otoferlin, which is essential for hearing function. Without it, hair cells in the inner ear cannot pass sound signals to the brain, causing severe-to-complete deafness at birth. OTOF mutations account for about 2 to 8 in every 100 cases.
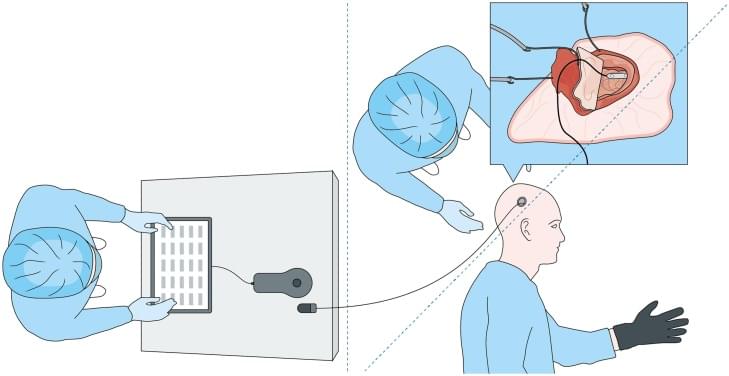
Gene therapies are designed to add a working version of mutated genes that lead to disease. Since a single faulty gene causes DFNB9, it is well-suited for gene therapy research. The treatment is a single injection into the inner ear that uses a harmless virus (AAV) to deliver a working copy of the OTOF gene to the cells needed for hearing.
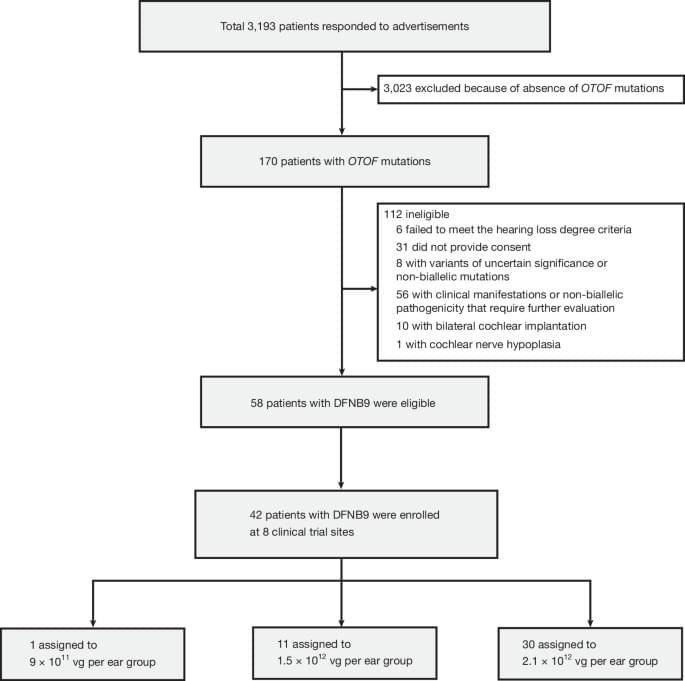
This latest trial enrolled 42 participants across eight sites in China, ranging in age from infants to adults (0.8 to 32.3 years). Each participant received one of three doses of a single gene therapy treatment: 36 in one ear and six in both ears. The research team then followed participants for up to 2.5 years, to see if treatment remained safe, affected their hearing and speech recognition. The researchers also sought to better understand why some participants may respond better than others.
“These multicenter trial results validate the effectiveness of our OTOF gene therapy,” said Yilai Shu, MD, PhD, a professor from Eye & ENT Hospital of Fudan University, who led the study. “The procedure can be broadly implemented in hospital settings, ensuring consistent delivery for a larger patient population.”
Abstract: Multicentre gene therapy for OTOF-related deafness followed up to 2.5 years https://www.nature.com/articles/s41586-026-10393-y.