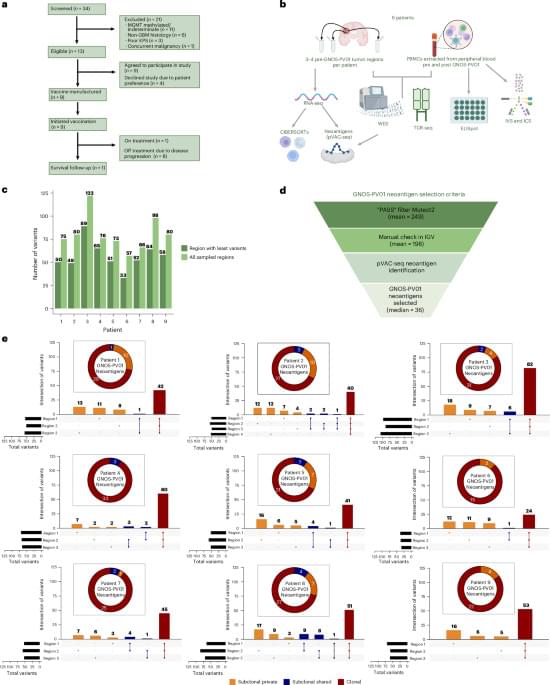
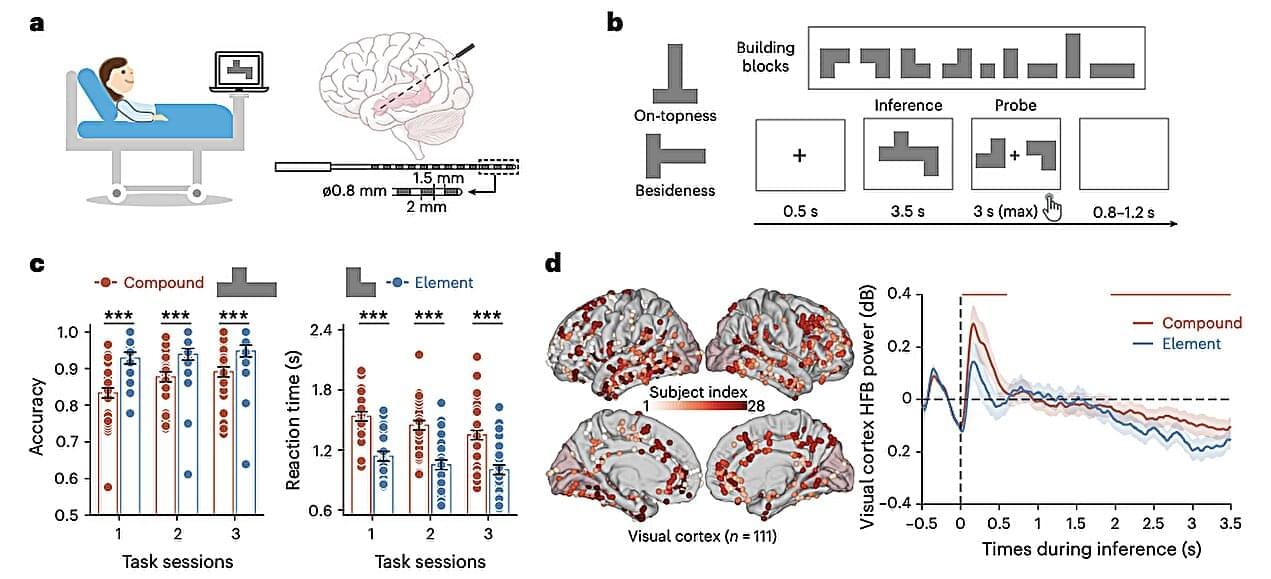
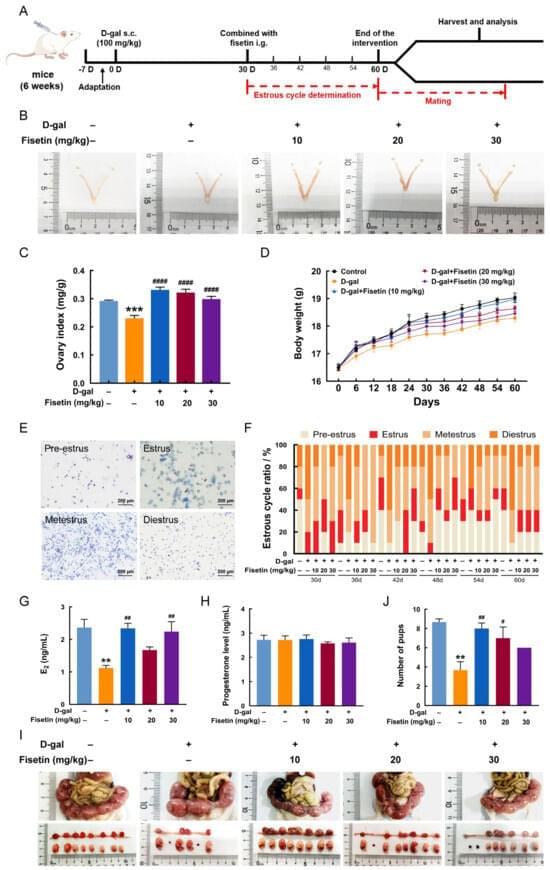
This talk is part of the “New Ideas in NCC Research” workshop of the Bamberg Mathematical Consciousness Science Initiative (BAMΞ). For more talks and details, see https://www.uni-bamberg.de/en/bamxi/r…
Abstract: In recent years, the search for the neural correlates of consciousness (NCC) has been complemented, and influenced, by the ongoing efforts to test neuroscientific theories of consciousness. A key insight from these efforts, though, is that many theories remain underdeveloped and not fully specified, making it harder to establish stringent tests for their predictions. In this talk, I will present a novel methodological approach that represents scientific theories as networks of beliefs structured in a core-periphery manner. These Prediction Maps visualize theoretical claims and empirical predictions, and illustrate their inferential relations. This framework further facilitates systematic theory testing by allowing researchers to evaluate the evidential weight of different components of a theory, and to identify which experimental results would constitute the most informative tests. To do so, we apply graph-theoretic and network analysis metrics, quantifying the centrality of specific predictions. I argue that this approach can advance efforts to arbitrate between theories of consciousness and to identify their most promising candidate mechanisms as NCCs.