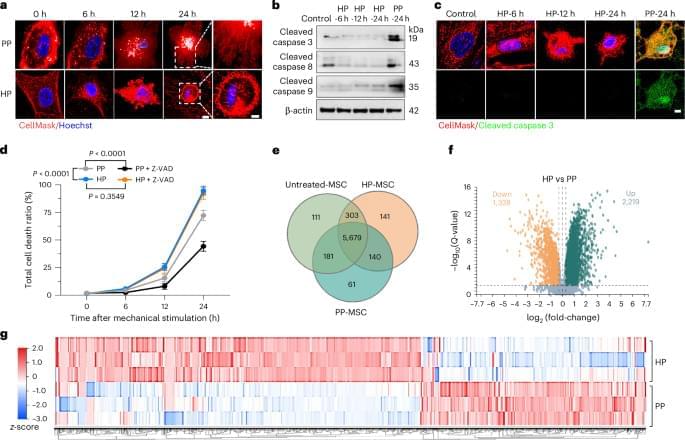
Intermittent hypobaric pressure extends the lifespan and rescues the osteoporosis phenotype in aged mice by activating the ion channel transmembrane protein 59 (TMEM59) and eliminating senescent cells via lysosome-dependent cell death.


In a surprising discovery, University of New Mexico researchers have found that OTULIN – an enzyme that helps regulate the immune system – also drives the formation of tau, a protein implicated in many neurodegenerative diseases, as well as brain inflammation and aging.
In a study published in the journal Genomic Psychiatry, the researchers reported that when they deactivated OTULIN, either by administering a custom-designed small molecule or knocking out the gene that codes for it, it halted the production of tau and removed it from neurons. The study was conducted on two different types of cells, some derived from a patient who had died from late-onset sporadic Alzheimer’s disease, and the rest from a line of human neuroblastoma cells that are frequently used in neuroscience research.
The discovery opens the door to potential treatments for Alzheimer’s and other neurodegenerative diseases, said Karthikeyan Tangavelou, PhD, a senior scientist in the lab of Kiran Bhaskar, PhD, professor in the Department of Molecular Genetics & Microbiology in the UNM School of Medicine.
Significance.
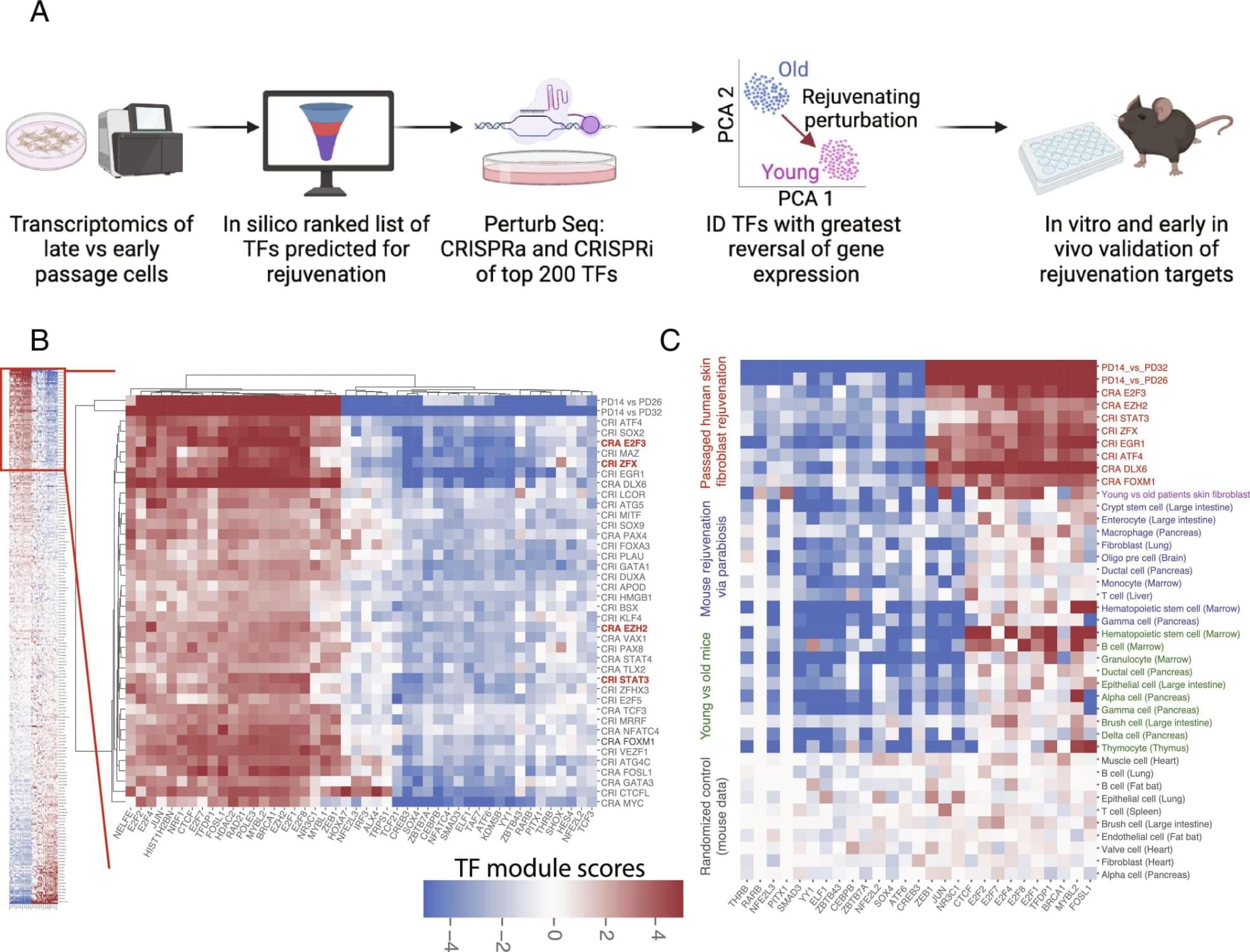
Cellular rejuvenation through transcriptional reprogramming has emerged as exciting approach to counter aging. However, to date, only a few of rejuvenating transcription factor (TF) perturbations have been identified. In this work, we developed a discovery platform to systematically identify single TF perturbations that drive cellular and tissue rejuvenation. Using a classical model of human fibroblast aging, we identified more than a dozen candidate TF perturbations and validated four of them (E2F3, EZH2, STAT3, ZFX) through cellular/molecular phenotyping. At the tissue level, we demonstrate that overexpression of EZH2 alone is sufficient to rejuvenate the liver in aged mice, significantly reducing fibrosis and steatosis, and improving glucose tolerance. Our work expanded the list of candidate rejuvenating TFs for future translation. Abstract.
Cellular rejuvenation through transcriptional reprogramming is an exciting approach to counter aging. Using a fibroblast-based model of human cell aging and Perturb-seq screening, we developed a systematic approach to identify single transcription factor (TF) perturbations that promote rejuvenation without dedifferentiation. Overexpressing E2F3 or EZH2, and repressing STAT3 or ZFX, reversed cellular hallmarks of aging—increasing proliferation, proteostasis, and mitochondrial activity, while decreasing senescence. EZH2 overexpression in vivo rejuvenated livers in aged mice, reversing aging-associated gene expression profiles, decreasing steatosis and fibrosis, and improving glucose tolerance. Mechanistically, single TF perturbations led to convergent downstream transcriptional programs conserved in different aging and rejuvenation models. These results suggest a shared set of molecular requirements for cellular and tissue rejuvenation across species. Sign up for PNAS alerts.
Get alerts for new articles, or get an alert when an article is cited. Cellular rejuvenation through transcriptional reprogramming is an exciting approach to counter aging and bring cells back to a healthy state. In both cell and animal aging models, there has been significant recent progress in rejuvenation research. Systemic factors identified in young blood through models such as heterochronic parabiosis (in which the circulatory systems of a young and aged animal are joined) rejuvenate various peripheral tissues and cognitive function in the brain (1–4). Partial reprogramming at the cellular level with the Yamanaka factors (four stem cell transcription factors) reverses cellular and tissue-level aging markers and can extend lifespan in old mice (5–8). These discoveries support the notion that transcriptional reprogramming is a powerful approach to improving the health of cells and tissues, and one day could be used as an approach for human therapeutics. However, to date, only a couple of rejuvenating transcription factor (TF) perturbations have been identified (9, 10) and most of them require the overexpression of TFs. We hypothesized that there are multiple other TF perturbations which could reset cells and tissues back to a healthier or younger state—rejuvenating them. Identifying complementary rejuvenating strategies is important as it will increase the chance of successful future translation. We developed a high-throughput platform, the Transcriptional Rejuvenation Discovery Platform (TRDP), which combines computational analysis of TF binding motifs and target predictions (Materials and Methods), global gene expression data of old and young cell states, and experimental genetic perturbations to identify which TF can restore overall gene expression and cell phenotypes to a younger, healthier state. We developed TRDP to be applicable to any cell type, and in both aging and disease settings, with the only requirements being baseline comparison of gene expression data comparing the older/diseased state to the younger/healthier state and the ability to perform genetic perturbations. To model aging in vitro as a validation of our approach, we used the canonical aging model of passaged fibroblasts (11, 12). We tested 400 TF perturbations via our screen and validated reversal of key cellular aging hallmarks in late passage human fibroblasts for four top TFs: E2F3, EZH2, STAT3, and ZFX. Moreover, EZH2 overexpression in vivo rejuvenated livers in aged mice—reversing aging-associated global gene expression profiles, significantly reducing steatosis and fibrosis, and improving glucose tolerance. These findings point to a conserved set of molecular requirements for cellular and tissue rejuvenation.
We sit down with José Cordeiro, author of The Death of Death, to uncover the timeline scientists believe could end aging — and even achieve immortality. From the promise of longevity escape velocity by 2030 to Ray Kurzweil’s bold prediction of immortality by 2045, José reveals the breakthroughs, roadblocks, and revolutionary ideas shaping the future of human life.
Join the community behind the first ever Biotech Freedom City:
https://joinimmortalis.com
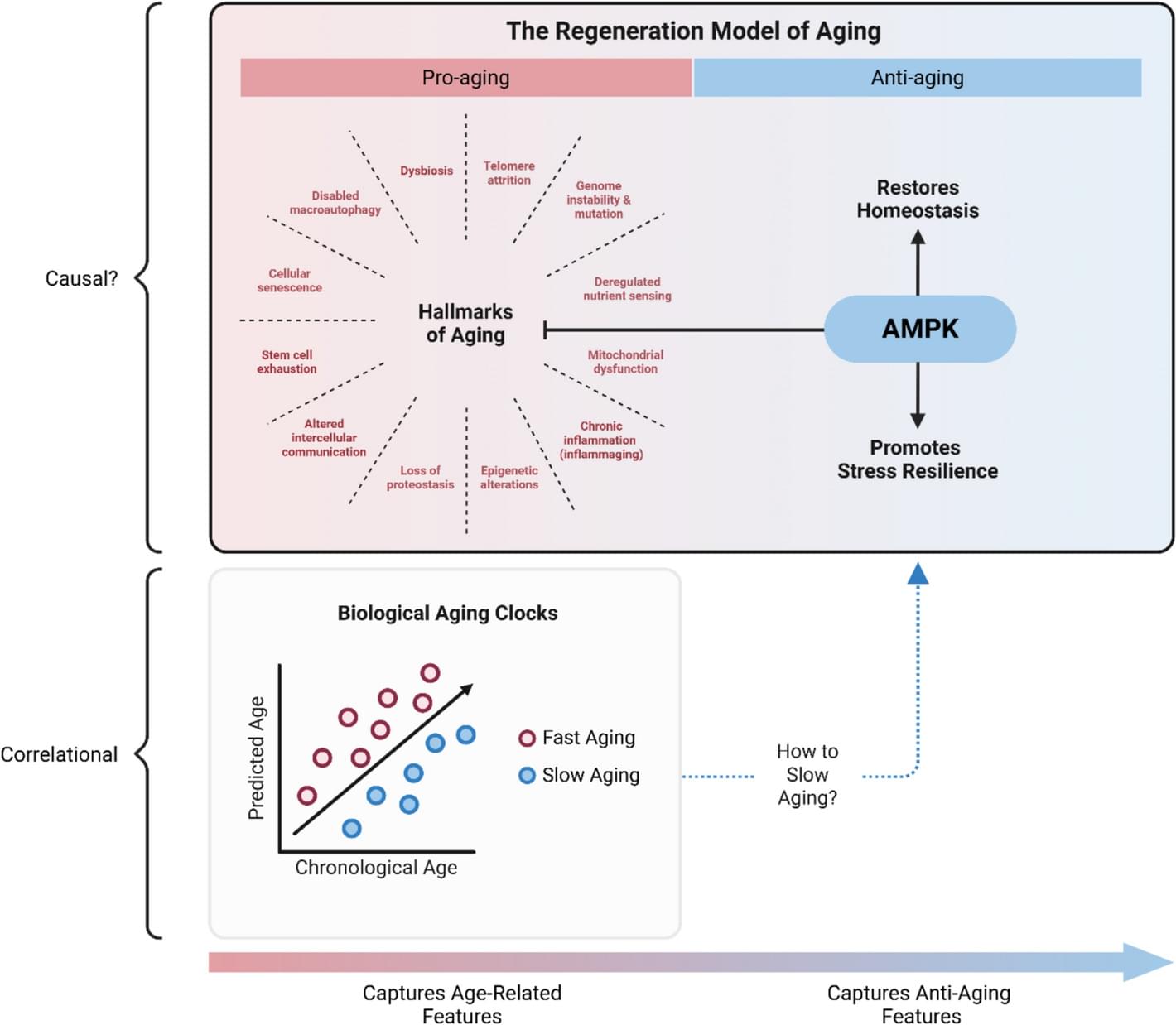
Aging is a primary risk factor for multi-morbidity and declining quality of life. The geroscience hypothesis states that targeting biological aging mechanisms may prevent or delay morbidity; however, translating theory into practice remains challenging. Unknown long-term risks and a lack of well-validated, responsive, and practical surrogate endpoints especially hinder the field’s preventive aspirations. This review addresses these obstacles by introducing the regeneration model of aging—a novel framework that integrates biological aging processes and distills the complexity of aging into a series of fundamental steps. The model provides insights into potential trade-offs of anti-aging interventions and can guide strategies to slow aging across diverse populations.

Hematopoietic aging extends far beyond the confines of the bone marrow, functioning as a central regulator of systemic decline through its influence on inflammation, immune dysregulation, and inter-organ communication. Moreover, reciprocal signaling from peripheral organs, such as the brain and gut, further shapes hematopoietic aging, highlighting the bidirectional nature of these interactions (Figure 3).
Join us on Patreon! https://www.patreon.com/MichaelLustgartenPhD
Discount Links/Affiliates:
Blood testing (where I get the majority of my labs): https://www.ultalabtests.com/partners/michaellustgarten.
At-Home Metabolomics: https://www.iollo.com?ref=michael-lustgarten.
Use Code: CONQUERAGING At Checkout.
Clearly Filtered Water Filter: https://get.aspr.app/SHoPY
Epigenetic, Telomere Testing: https://trudiagnostic.com/?irclickid=U-s3Ii2r7xyIU-LSYLyQdQ6…M0&irgwc=1
Use Code: CONQUERAGING
NAD+ Quantification: https://www.jinfiniti.com/intracellular-nad-test/
At SciCon 2024, John Cumbers, founder and CEO of SynBioBeta, explores the groundbreaking and controversial potential of synthetic biology and AI in brain and body replacement. He delves into stem cell research and AI’s role in regenerating brain function, while also addressing the provocative idea of gradually replacing parts of the brain and body. Cumbers discusses how these advancements could one day lead to life extension, challenging traditional views on aging, and raising ethical questions about the future of human biology.
SciCon (2024) is ResearchHub’s annual conference, which unites truth-seekers and innovators to push the boundaries of open science.
– ResearchHub’s mission is to accelerate the pace of scientific research. We are building a modern platform where people can collaborate on scientific research more efficiently, much like GitHub has done for software engineering. We believe scientific research should be accessible to everyone, collaborative, and prioritized.
Product: https://www.researchhub.com/
Website: https://researchhub.foundation/
GitHub: https://github.com/ResearchHub
In a nonrandomized phase 2 trial of adults with advanced dMMR/MSI-H noncolorectal cancers, combined nivolumab/ipilimumab showed an objective response rate of 63% and 6-month progression-free survival rate of 71%.
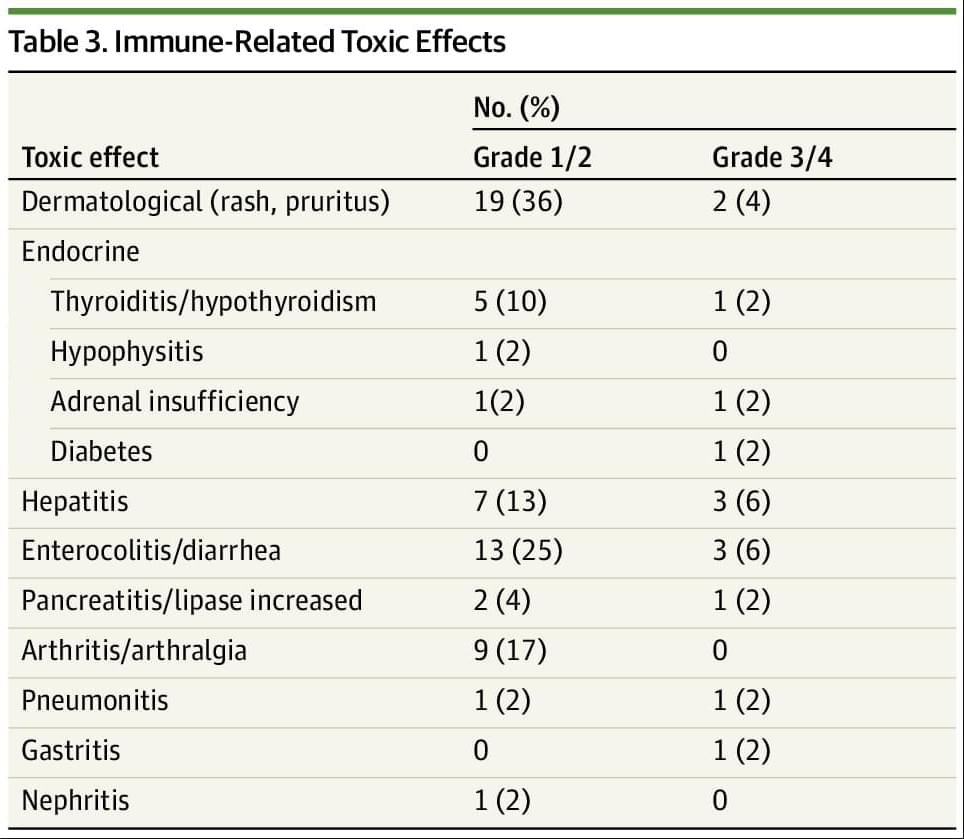
Main Outcomes and Measures The co-primary end points were objective response rate (ORR) and 6-month progression-free survival (6-PFS) as assessed by Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1, with the secondary end points being median overall survival (mOS), progression-free survival (PFS), and treatment-related toxic effects.
Results Overall, 52 participants were included. The median (range) age of participants was 62 (29−84) years; 41 (79%) were female individuals and 11 (21%) were male individuals. Overall, 52 patients representing 17 tumor types were enrolled, with the most common tumor type being endometrial cancer (26 [50%]). Twenty-seven patients (52%) were pretreated for metastatic disease. ORR was 63% (95% CI, 50% to 75%) with the median duration of response not being reached and 79% of responses being ongoing. The median PFS and OS have not been reached and the 6-month PFS was 71% (95% CI, 57%-81%). Overall, 12 patients (23%) experienced a grade 3/4 immune-related adverse event.
Conclusions and Relevance This nonrandomized clinical trial found that combined anti–PD-1/CTLA-4 blockade was associated with a high rate of durable responses in dMMR/MSI-H noncolorectal cancers, comparing favorably to published trials using anti–PD-1/programmed cell death ligand 1 monotherapy. Anti–PD-1/CTLA-4 blockade using nivolumab and ipilimumab may represent an alternative treatment option to monotherapy in this patient population.
A landmark international study that pooled brain scans and memory tests from thousands of adults has shed new light on how structural brain changes are tied to memory decline as people age.
The findings — based on more than 10,000 MRI scans and over 13,000 memory assessments from 3,700 cognitively healthy adults across 13 studies — show that the connection between shrinking brain tissue and declining memory is nonlinear, stronger in older adults, and not solely driven by known Alzheimer’s-associated genes like APOE ε4. This suggests that brain aging is more complex than previously thought, and that memory vulnerability reflects broad structural changes across multiple regions, not just isolated pathology.
Published in Nature Communications, the study, “Vulnerability to memory decline in aging revealed by a mega-analysis of structural brain change,” found that structural brain change associated with memory decline is widespread, rather than confined to a single region. While the hippocampus showed the strongest association between volume loss and declining memory performance, many other cortical and subcortical regions also demonstrated significant relationships. This suggests that cognitive decline in aging reflects a distributed macrostructural brain vulnerability, rather than deterioration in a few specific brain regions. The pattern across regions formed a gradient, with the hippocampus at the high end and progressively smaller but still meaningful effects across large portions of the brain.
https://www.nature.com/articles/s41467-025-66354-y
Genetic risk for Alzheimer’s and widespread brain shrinkage linked to greater memory loss — even in otherwise healthy adults.