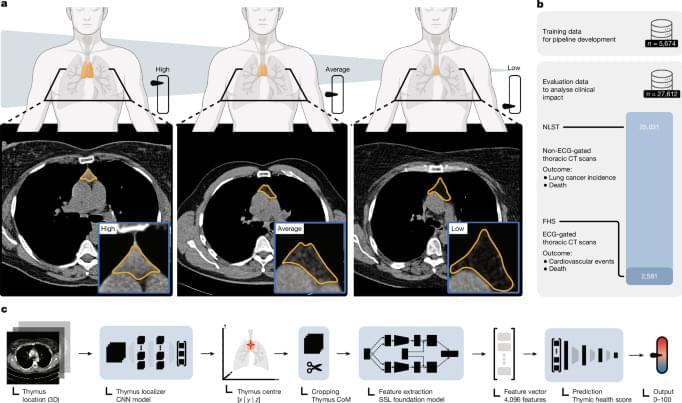
Assessing thymic function and health has highlighted the lifelong importance of the thymus as an organ that could be targeted to improve health outcomes, protect against disease and promote healthy ageing.

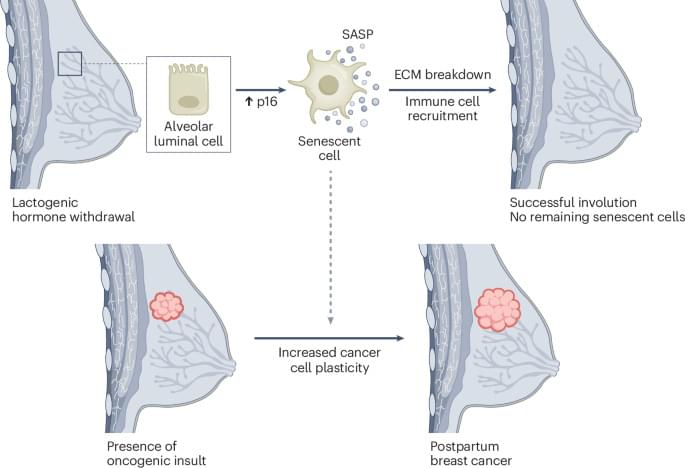
Cellular senescence has been linked to both beneficial and detrimental functions. Chiche, Djoual, Charifou and colleagues show that senescence supports normal postpartum mammary gland remodeling, but that when oncogenic events coincide with involution, senescent cells enhance tumorigenesis by regulating plasticity, invasion and metastasis.
LigandForge generates 150,000 peptide drug candidates in 3 minutes — a million times faster than existing methods, unlocking a tsunami of possible treatments.
A man with no medical background used ChatGPT, AlphaFold, and Grok to design a custom mRNA cancer vaccine for his dying dog — and her biggest tumor shrank 75%.
Meanwhile, scientists discovered a single protein that literally spreads aging through your bloodstream. These stories are each incredible on their own. But the big story is the implications for curing aging.
In this deep dive, I break down how these three breakthroughs fit together, what peptides and mRNA vaccines actually are (and how they’re different), and why this moment might be the most important inflection point in the history of drug design.
The age of custom AI cures isn’t coming. It’s here.
HUME BODY POD DISCOUNT UP TO 50% OFF:

Immortality is doubtful but what might be possible?
A central question in molecular biology is how cells protect their chromosomes from damage during repeated cell division. At the heart of this protective process is an enzyme called telomerase. Now an international research team has mapped the three-dimensional structure of telomerase in the yeast Saccharomyces cerevisiae, a widely used model organism in genetics.
Using cutting-edge technology, the scientists were able to visualize the architecture of this complex enzyme in unprecedented detail, uncovering unexpected features that may explain how it functions.
This major discovery was the result of an international collaboration between Pascal Chartrand, a professor in the Department of Biochemistry and Molecular Medicine at Université de Montréal, and researchers from Université de Sherbrooke and the MRC Laboratory of Molecular Biology in the U.K. Their findings were recently published in Science.
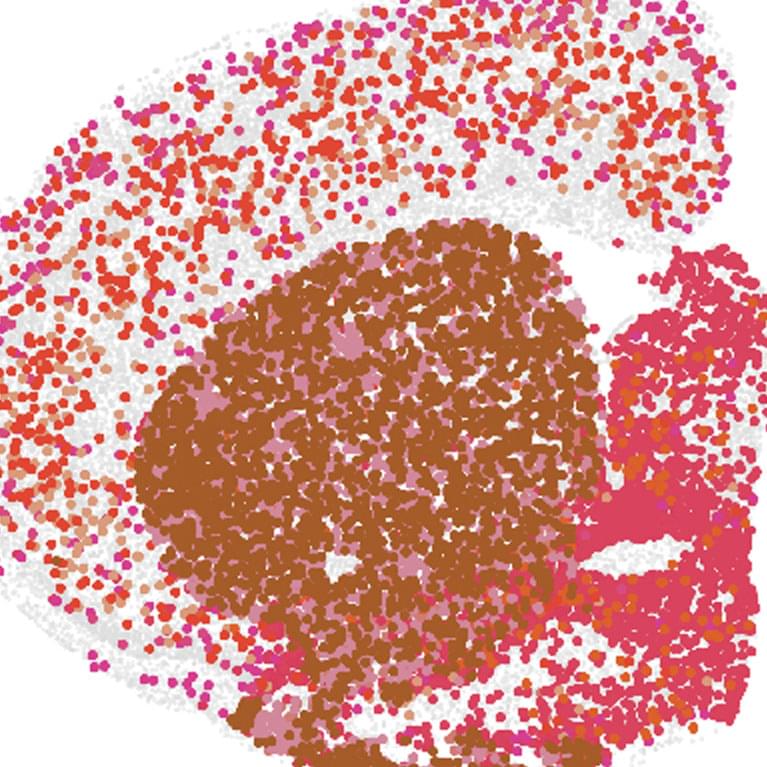
A new study from the Salk Institute maps how the aging brain changes at the epigenetic level — cell type by cell type.
The researchers created one of the most detailed single-cell atlases yet of the aging mouse brain, spanning 8 brain regions, 36 cell types, and hundreds of thousands of cells. They found major age-related changes in DNA methylation, chromatin structure, and gene activity, with some of the strongest changes appearing in non-neuronal cells.
This kind of work matters because it moves brain aging closer to mechanism — not just describing decline, but identifying the molecular regulatory shifts that may drive vulnerability to neurodegenerative disease.
Highlights Salk researchers create epigenetic atlas of cell type-specific changes in the aging mouse brain The atlas represents eight different brain regions and 36 different cell types, and shows clear epigenetic differences associated with different ages The new resource—available publicly on Amazon Web services—can be used to unravel age-related contributions to neurodegenerative diseases like Alzheimer’s, Parkinson’s, and ALS LA JOLLA—Neurodegenerative diseases affect more than 57 million people globally. The incidence of these diseases, from Alzheimer’s to Parkinson’s to ALS and beyond, is expected to double every 20 years. Though scientists know aging is a major risk factor for neurodegenerative diseases, the full mechanisms behind aging’s impact remain unclear.
You are invited to presentations by “The Immortality Thief: How Toxins Shorten Life — and How to Fight Back with Glutathione, TPE & Beyond.” Bill Faloon will also give an update on Age Reversal presentation. — March 26, 2026” Our doors open at 6:00 PM with the main event at 7:00 PM Live in Person AND in Zoom and YouTube Livestream Afterwards, enjoy a 5 star banquet and conversations with members on Health and Super Longevity.
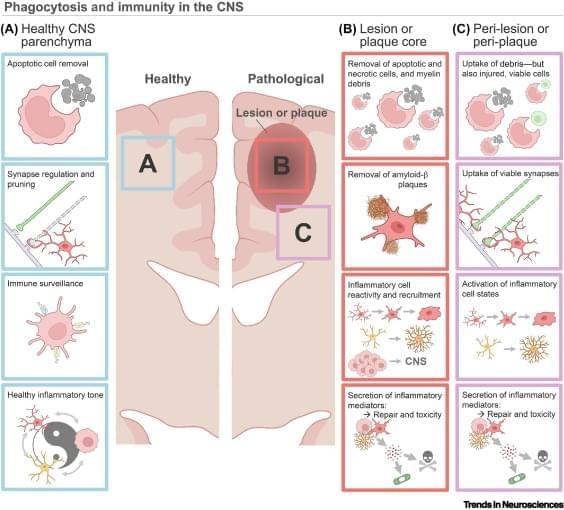
Brain phagocytosis and neuroinflammation.
Phagocytes in the central nervous system (CNS), including astrocytes, microglia, and macrophages, shape development and homeostasis by pruning synapses and removing apoptotic debris.
Phagocytosis is mediated by various ligand–receptor dyads and signaling pathways, enabling CNS phagocytes to respond to neuroimmune shifts across the lifespan and during pathology.
Phagocytosis pathways regulate recovery in various models of CNS pathology, including multiple sclerosis, CNS injury, ischemic stroke, and age-associated neurodegeneration.
Phagocytosis pathways are intimately integrated with the inflammatory cell state and remove viable cells in pathology-adjacent tissue, highlighting the complexity of targeting these systems.
To maximize benefit and minimize off target damage, new phagocytic-based approaches should optimize drug delivery timing and location, tailored to each CNS pathology. sciencenewshighlights ScienceMission https://sciencemission.com/resolution-of-inflammation