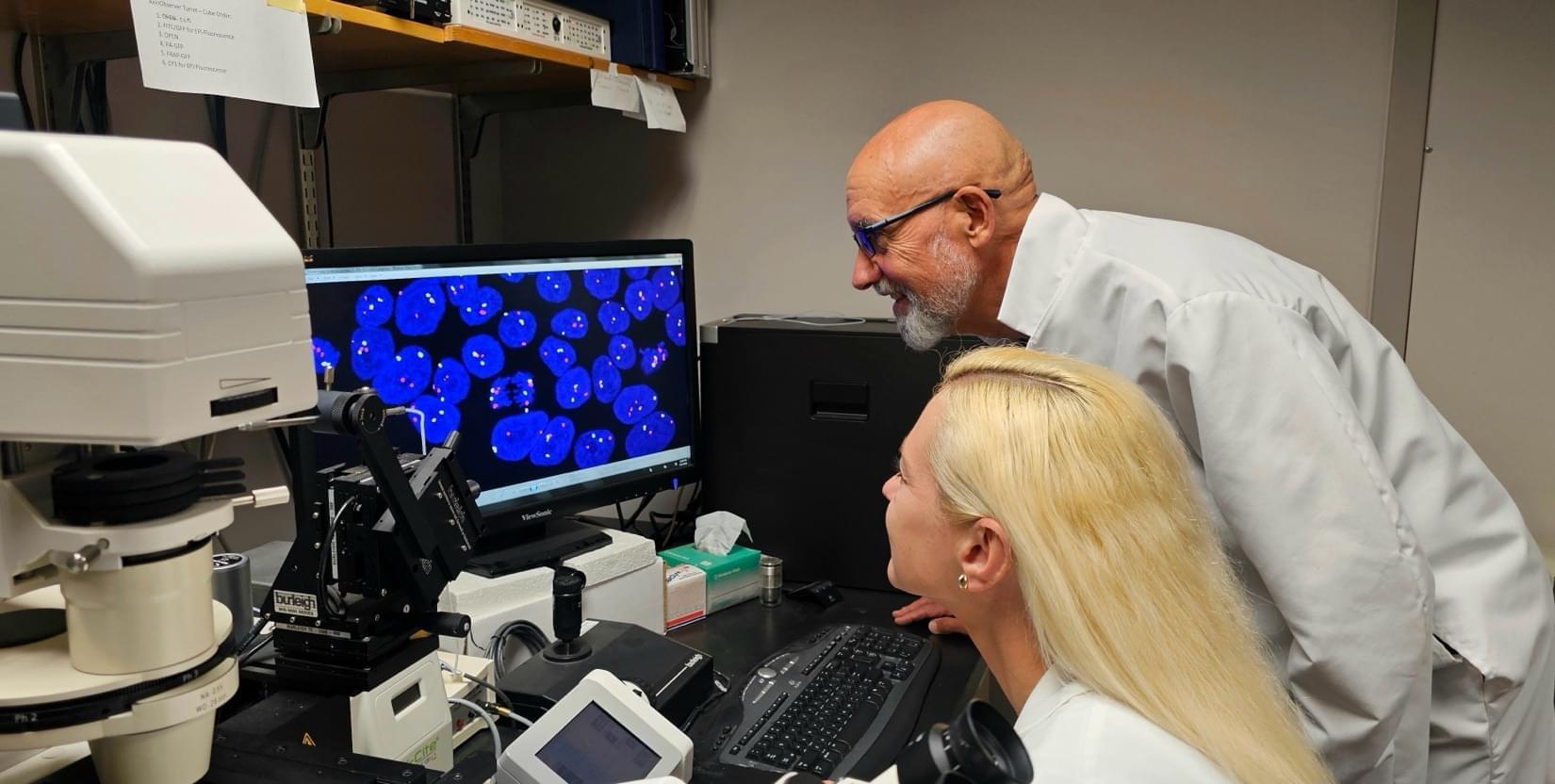
Scientists use Allen Institute cell lines to learn how the genetic defect spreads in stem cells

When disaster leaves only a handful, can a civilization restart? We explore genetic bottlenecks, colony failure, and the limits of survival.
Get Nebula using my link for 50% off an annual subscription: https://go.nebula.tv/isaacarthur.
Watch my exclusive video Surviving a New Ice Age: https://nebula.tv/videos/isaacarthur–… out Practical Engineering: https://nebula.tv/practicalconstructi… 🛒 SFIA Merchandise: https://isaac-arthur-shop.fourthwall… 🌐 Visit our Website: http://www.isaacarthur.net ❤️ Support us on Patreon: / isaacarthur ⭐ Support us on Subscribestar: https://www.subscribestar.com/isaac-a… 👥 Facebook Group:
/ 1,583,992,725,237,264 📣 Reddit Community:
/ isaacarthur 🐦 Follow on Twitter / X:
/ isaac_a_arthur 💬 SFIA Discord Server:
/ discord Credits: Genetic Bottlenecks – How Few People Can Start a World? Or Restart One? Written, Produced & Narrated by: Isaac Arthur Select imagery/video supplied by Getty Images Chapters 0:00 Intro 10:56 Restoration 19:26 Practical Engineering.
Check out Practical Engineering: https://nebula.tv/practicalconstructi…
🛒 SFIA Merchandise: https://isaac-arthur-shop.fourthwall…
🌐 Visit our Website: http://www.isaacarthur.net.
❤️ Support us on Patreon: / isaacarthur.
⭐ Support us on Subscribestar: https://www.subscribestar.com/isaac-a…
👥 Facebook Group: / 1583992725237264
📣 Reddit Community: / isaacarthur.
🐦 Follow on Twitter / X: / isaac_a_arthur.
💬 SFIA Discord Server: / discord.
Credits:
Genetic Bottlenecks – How Few People Can Start a World? Or Restart One?
Written, Produced & Narrated by: Isaac Arthur.
Select imagery/video supplied by Getty Images.
Chapters.
0:00 Intro.
10:56 Restoration.
19:26 Practical Engineering
JUST PUBLISHED:Click here to read the latest free, Open Access article from BMEF.
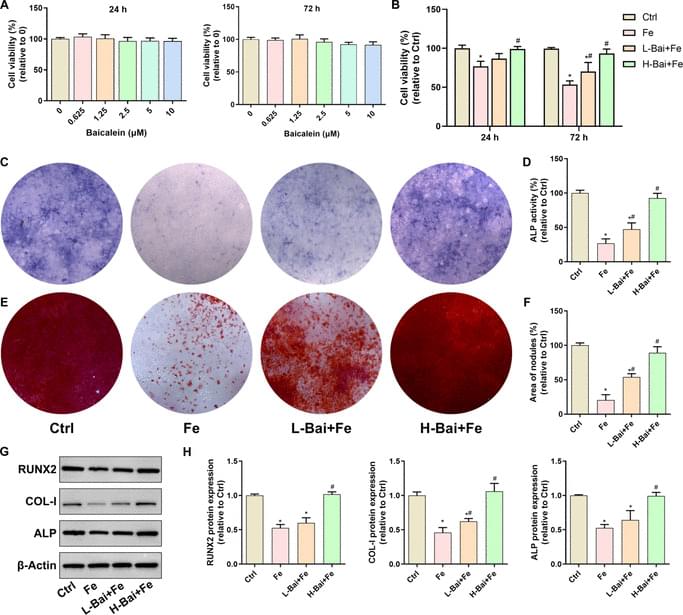
The transcription factor Nrf2 orchestrates cellular defenses against redox imbalance and lipid peroxidation, partly through regulating the expression of 2 key gatekeepers of ferroptosis: SLC7A11 and GPX4 [44]. As such, the Keap1/Nrf2 pathway is recognized as a master regulator of ferroptosis in osteoblasts [45]. Under stress conditions, Nrf2 dissociates from the Keap1–Nrf2 complex, translocates into the nucleus, and initiates the transcription of genes containing antioxidant response elements [46]. Previous studies have reported that Nrf2 activation protects osteoblasts from ferroptosis in bone tissue and alleviates osteoporosis [28,47]. Consistently, we observed that under iron overload conditions, baicalein restored nuclear Nrf2 levels and the expression of downstream targets GPX4 and SLC7A11. Both genetic and pharmacological inhibition of Nrf2 abolished the cytoprotective and pro-osteogenic effects of baicalein. These findings suggest that baicalein prevents ferroptosis in osteoblasts via activation of the Nrf2/GPX4 pathway.
Clinically, iron overload conditions, such as transfusion-induced iron overload in thalassemia and hereditary hemochromatosis, are strongly associated with low bone mass and increased fracture risk [48,49]. Current treatment options (e.g., iron chelators, phlebotomy, and anti-resorptive agents) fail to simultaneously address iron overload and bone damage. Baicalein has undergone human safety and pharmacokinetic studies, which indicate no significant side effects even at high doses [50,51]. Our study demonstrates that baicalein not only prevents bone loss by protecting osteoblasts from ferroptosis but also effectively reduces systemic iron storage. Although beyond the scope of this work, baicalein’s known anti-osteoclastogenic effects may synergistically contribute to its overall bone-protective actions in iron overload conditions. These findings suggest that baicalein is a promising therapeutic agent for iron overload-related bone disorders. Although clinical trials are warranted, the dose of baicalein used in our study was extrapolated from clinically tolerated doses in humans, thereby supporting the potential feasibility of its clinical application.
In summary, this study provides the first definitive evidence that baicalein effectively inhibits iron overload-induced ferroptosis in osteoblasts by activating the Nrf2/GPX4 signaling pathway, thereby promoting bone formation and preventing bone loss. Our findings not only elucidate the mechanism by which baicalein functions as a novel ferroptosis inhibitor in bone protection but also highlight its role as a “dual-function” therapeutic strategy—combining iron chelation and anti-bone-loss capacities. Given its favorable safety profile and existing human pharmacokinetic data, our results provide strong preclinical evidence supporting the clinical translation of baicalein for the treatment of iron overload-related bone diseases. Targeting the ferroptosis pathway, particularly via Nrf2/GPX4 activation by baicalein, represents a highly promising novel strategy for preventing and treating iron overload-induced bone loss.
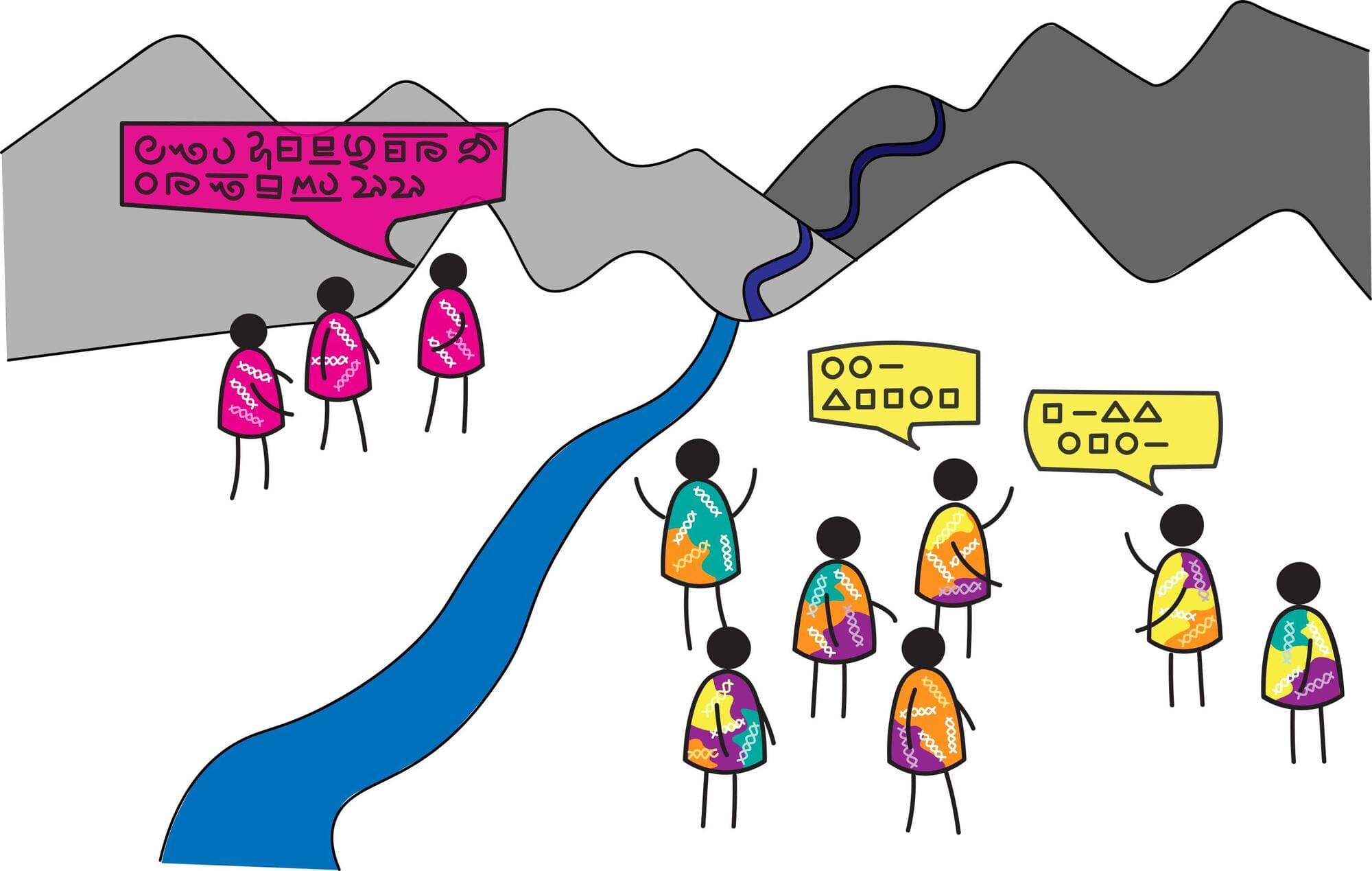
Languages and human DNA both capture aspects of human diversity. But how are they related? A new international study led by the University of Zurich finds a clear but counterintuitive pattern: regions with high genetic diversity tend to have more similar languages, while isolated populations with low genetic diversity show greater linguistic diversity. The research is published in the journal Proceedings of the National Academy of Sciences.
At first glance, the findings seem surprising. One might expect regions with greater genetic diversity, often shaped by migration and population mixing, to also show greater diversity in language. But the study reveals the opposite.
“We were struck by how robust this inverse relationship is across the globe,” says Anna Graff, lead author of the study and linguist at the University of Zurich. “Places where people have mixed more tend to be genetically diverse, but their languages are structurally more similar. In contrast, places with long-term isolation show less genetic diversity, yet much greater diversity in how languages are structured. Crucially, this relationship holds after adjusting for a wide range of confounding factors, including deep population history such as the timing of continental settlement.”
The performance garnered a huge media attendance, allowing the team to accomplish their goal of bringing attention to SCID. Frishkopf hopes to perform the piece in a concert in the future.
Genome Music Raises Rare Disease Awareness from Concerts to Contests
From a serendipitous idea to physical compositions, Kantipuly and her collaborators have demonstrated the power of music to bring people together and work for a good cause. Recently, the team connected with another composer, Casey McPherson, who also produces genetic music but in more modern styles and the founder of To Cure a Rose, a nonprofit organization focused on developing a cure for a rare genetic disease.
Skin aging is driven by intrinsic and extrinsic factors. Epigenetic alterations are one primary hallmark of aging and powerful biomarkers of biological skin age. To investigate epigenetic skin aging mechanisms and their regulation as a skin longevity approach across diverse ethnicities and phototypes, we assessed epidermal methylomes from white, African, and Asian donors.
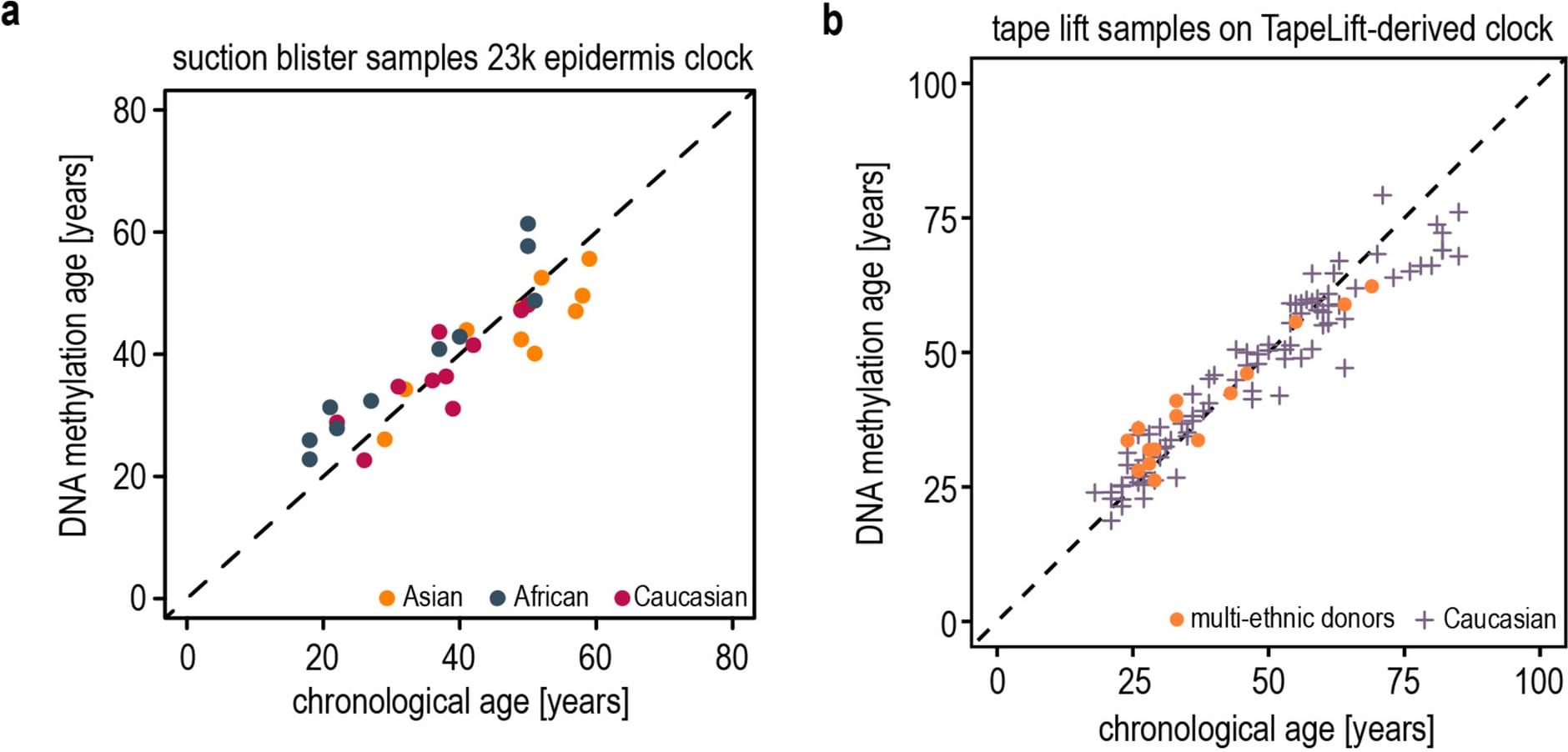
We collected epidermis samples from 17 multi-ethnic donors with diverse phototypes using a newly established tape-stripping method followed by array-based DNA methylation profiling to investigate the robustness of DNA methylation clocks across diverse ethnic backgrounds. Additionally, we conducted a clinical study with 60 participants representing Fitzpatrick phototypes I–VI. Diverse clinical parameters and biological skin age of the volunteers were determined at baseline and after applying a serum containing the natural epigenetic inhibitor dihydromyricetin (DHM) for 8 weeks to investigate skin longevity effects across phototypes.
Data analysis revealed that age-dependent DNA hypermethylation is conserved across populations and affects genes essential for keratinocyte vitality and longevity. A newly developed epidermal methylation clock accurately predicted biological age in multi-ethnic cohorts, confirming the robustness of epigenetic age estimation across phototypes. Topical application of a DHM-containing serum significantly reduced epidermal DNA methylation age. Epigenetic rejuvenation was associated with clinical improvements, including reduced skin roughness and wrinkle visibility and occupancy, and increased dermal echogenicity.
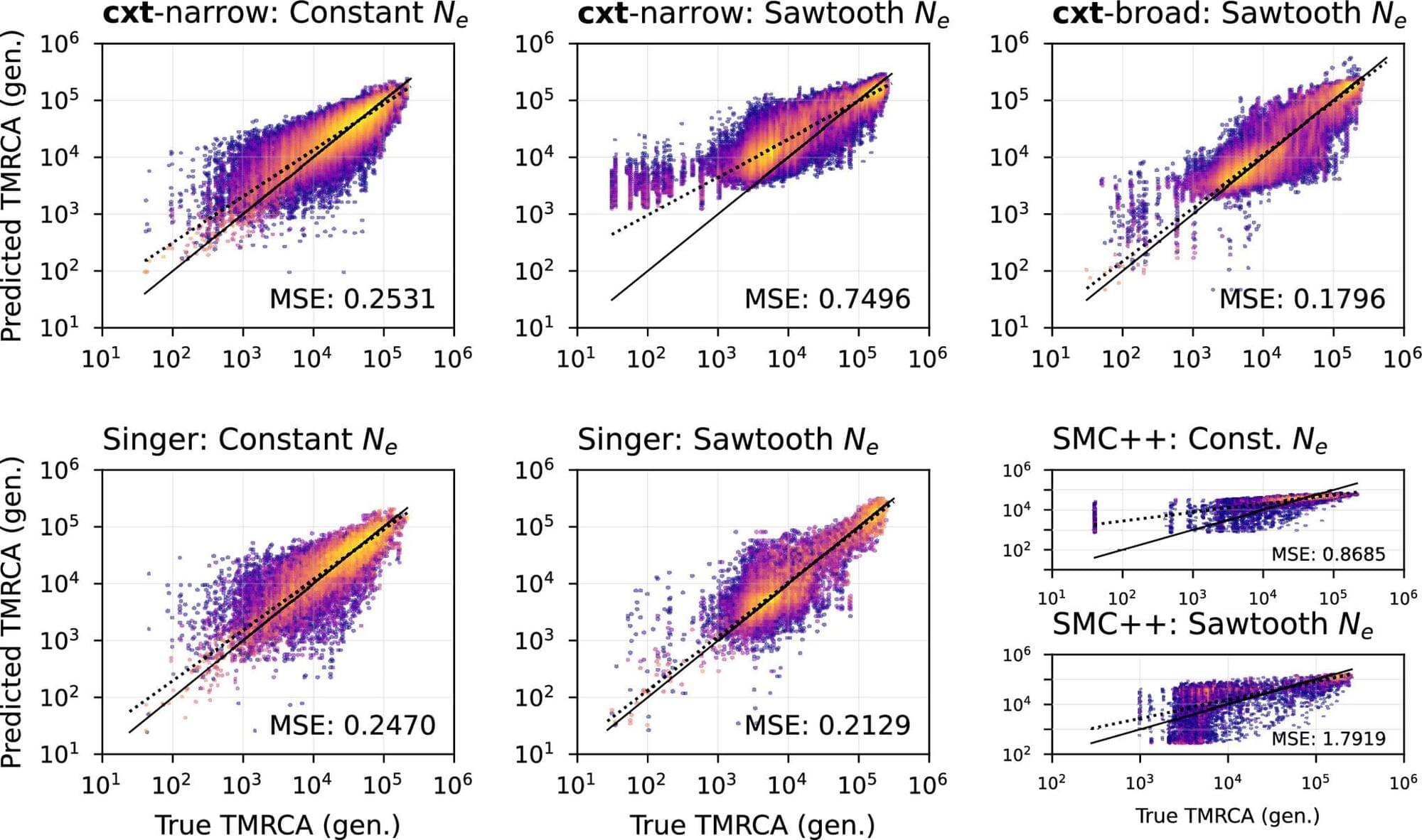
Researchers at the University of Oregon have developed an artificial intelligence tool that can read genetic code the way large language models like ChatGPT read text. Scanning the genome for biological mutation patterns, the computer model traces pairs of genes back in time to their last common ancestor.
It’s the first language model designed for population genetics, said Andrew Kern, a computational biologist in the UO College of Arts and Sciences. As described in a paper published April 10 in the Proceedings of the National Academy of Sciences, the AI tool offers scientists a fast and flexible alternative to classical methods for reconstructing evolutionary history.
In practice, it can help researchers like Kern understand when disease-resistance genes emerged in a population, for example, or when species evolved key traits.
