Treating fetuses for the enzyme deficiency might prevent early disease progression.

This is primarily about Rejuvant or AKG trials. When they first reported an 8 year age difference I did not truly believe it as that would mean I could take it, de-age, then age a bit, then just take it again. The tests however are ongoing and will feature people who are biologically older than their calendar age. There is also mention of 110 drugs/supplements that do “something” to mice. That would be quite a stack to take.
Dr Brian Kennedy presents potential interventions for extend our healthspan. Among them, Rejuvant – a CaAKG supplement which contain calcium + alpha-ketoglutarate shows better response for people who has biological age older than chronological age.
The first human trial of Rejuvant to reverse human biological age by 8 years in 7 month.
Rejuvant®, a potential life-extending compound formulation with alpha-ketoglutarate and vitamins, conferred an average 8 year reduction in biological aging, after an average of 7 months of use, in the TruAge DNA methylation test.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8660611/
Dr. Brian Kennedy is internationally recognized for his research in the basic biology of aging and as a visionary committed to translating research discoveries into new ways of delaying, detecting, and preventing human aging and associated diseases. He is a Professor in the Departments of Biochemistry and Physiology at National University Singapore and Director of the Centre for Healthy Ageing in the National University Health System. From 2010 to 2016 he was the President and CEO of the Buck Institute for Research on Aging. Currently, he remains as a Professor at the Institute. Dr. Kennedy also has an adjunct appointment at the USC Davis School of Gerontology. Dr. Kennedy is also actively involved Biotechnology companies, serving in consulting and Board capacities, as well as Scientific Director of Affirmativ Health. Dr. Kennedy also serves as a Co-Editor-In-Chief at Aging Cell.
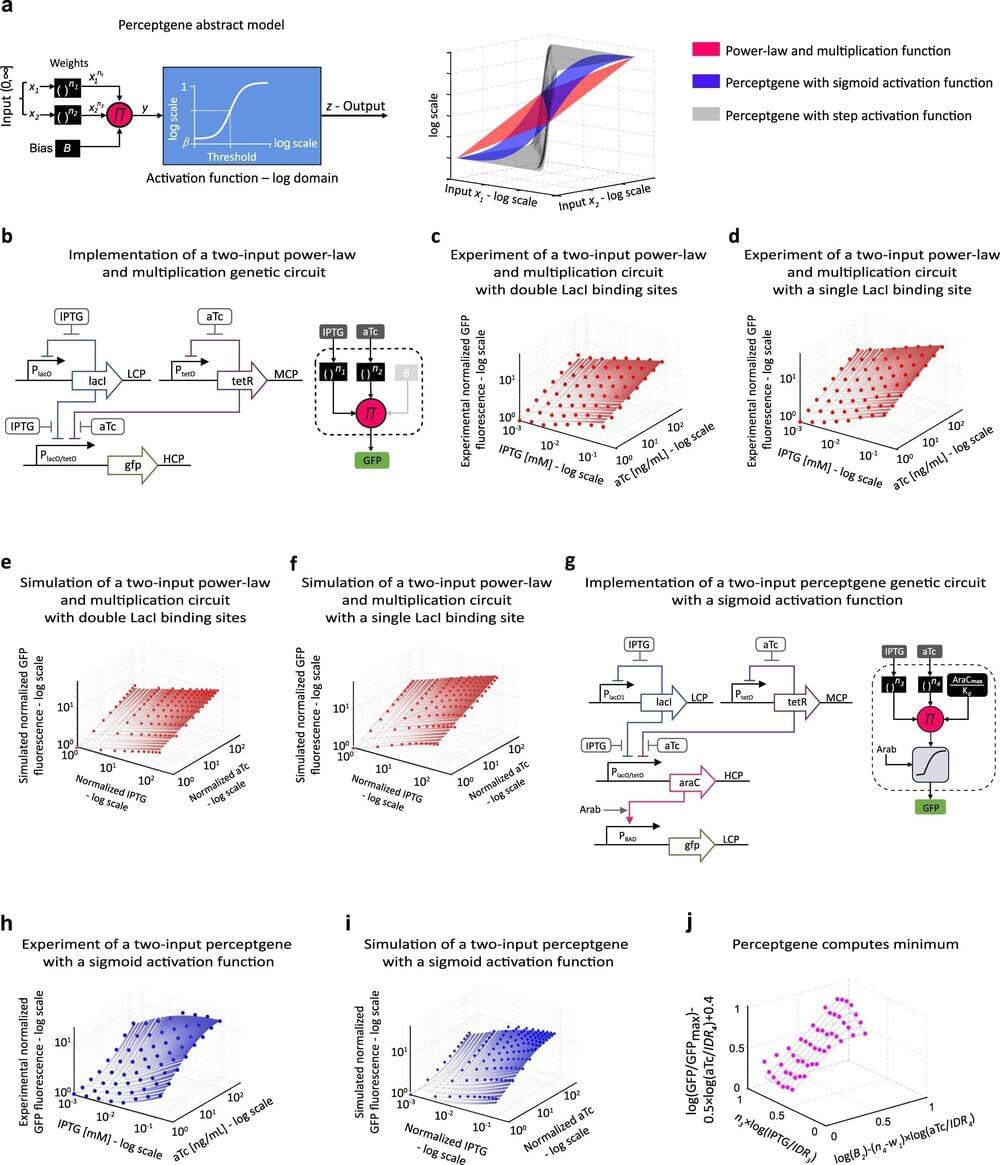
Bringing together concepts from electrical engineering and bioengineering tools, Technion and MIT scientists collaborated to produce cells engineered to compute sophisticated functions— biocomputers of sorts.
Graduate students and researchers from Technion—Israel Institute of Technology Professor Ramez Daniel’s Laboratory for Synthetic Biology & Bioelectronics worked together with Professor Ron Weiss from the Massachusetts Institute of Technology to create genetic “devices” designed to perform computations like artificial neural circuits. Their results were recently published in Nature Communications.
The genetic material was inserted into the bacterial cell in the form of a plasmid: a relatively short DNA molecule that remains separate from the bacteria’s “natural” genome. Plasmids also exist in nature, and serve various functions. The research group designed the plasmid’s genetic sequence to function as a simple computer, or more specifically, a simple artificial neural network. This was done by means of several genes on the plasmid regulating each other’s activation and deactivation according to outside stimuli.

The patients had some, although severely diminished, visual function during the day, however, at night they were essentially blind, with light sensitivity 10,000–100,000 times less than normal.
According to researchers at the Scheie Eye Institute at the Perelman School of Medicine at the University of Pennsylvania, adults with a genetic form of childhood-onset blindness experienced remarkable recoveries of night vision within days of receiving an experimental gene therapy.
The patients had Leber Congenital Amaurosis (LCA), a congenital blindness caused by GUCY2D gene mutations. The findings were published in the journal iScience. The researchers administered AAV gene therapy, which contains the DNA.

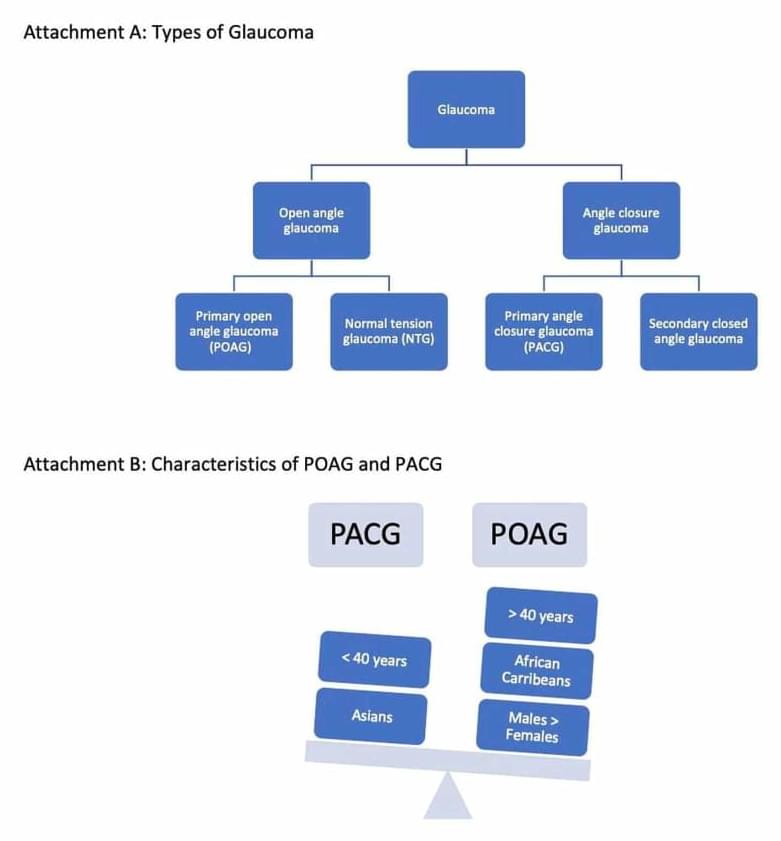
Glaucoma impacts tens of millions of people globally, and is the second leading cause of blindness, after cataracts. Now, Purdue University researchers have developed smart contact lenses that may help save people’s sight.
Glaucoma is a multifactorial optic degenerative neuropathy characterized by the loss of retinal ganglion cells. It is a combination of vascular, genetic, anatomical, and immune factors. Glaucoma poses a significant public health concern as it is the second leading cause of blindness after cataracts, and this blindness is usually irreversible. It is estimated that 57.5 million people worldwide are affected by primary open-angle glaucoma (POAG). People over 60 years of age, family members of those already diagnosed with glaucoma, steroid users, diabetics, as well as those with high myopia, hypertension, central cornea thickness of.
Watch any episode of “CSI,” and a character will use forensic DNA profiling to identify a criminal. A new study from San Francisco State University suggests that these forensic profiles may indirectly reveal medical information—perhaps even those of crime victims—contrary to what the legal field has believed for nearly 30 years. The findings, now published in the Proceedings of the National Academy of Sciences, could have ethical and legal implications.
“The central assumption when choosing those [forensic] markers was that there wouldn’t be any information about the individuals whatsoever aside from identification. Our paper challenges that assumption,” said first author Mayra Bañuelos, who started working on the project as a San Francisco State undergraduate and is now a Ph.D. student at Brown University.
Law enforcement uses the Combined DNA Index System (CODIS), a system organizing criminal justice DNA databases that uses specific genetic markers to identify individuals. Crime labs from national, state and local levels contribute to these databases and provide profiles from samples collected from crime scene evidence, convicted offenders, felony arrestees, missing persons and more. Law officials can use the database to try to match samples found in an investigation to profiles already stored in the database.
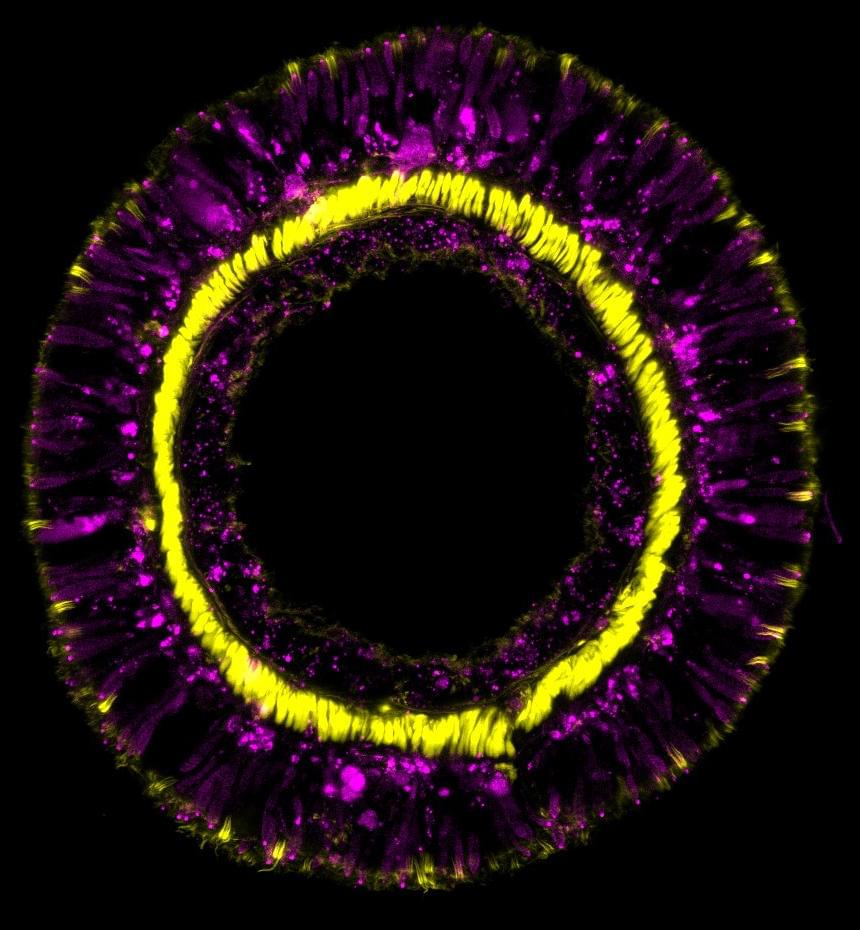
Sea anemones are seemingly immortal animals. They seem to be immune to aging and the negative impacts that humans experience over time. However, the exact reasons for their eternal youth are not completely understood.
The genetic fingerprint of the sea anemone Nematostella vectensis reveals that members of this incredibly ancient animal phylum employ the same gene cascades for neural cell differentiation as more complex organisms. These genes are also in charge of maintaining the balance of all cells in the organism during the anemone’s lifetime. These findings were recently published in the journal Cell Reports by a group of developmental biologists headed by Ulrich Technau of the University of Vienna.
Almost all animal organisms are made up of millions, if not billions, of cells that join together in intricate ways to create specific tissues and organs, which are made up of a range of cell types, such as a variety of neurons and gland cells. However, it is unclear how this critical balance of diverse cell types emerges, how it is regulated, and if the different cell types of different animal organisms have a common origin.
Join us on Patreon!
https://www.patreon.com/MichaelLustgartenPhD
TruDiagnostic Discount Link (Epigenetic Testing)
CONQUERAGING!
https://bit.ly/3Rken0n.
Bristle Discount Link (Oral Microbiome Quantification):
ConquerAging15
https://www.bmq30trk.com/4FL3LK/GTSC3/
Quantify Discount Link (At-Home Blood Testing)
https://getquantify.io/mlustgarten.
Cronometer Discount Link (Daily Diet Tracking):
https://shareasale.com/r.cfm?b=1390137&u=3266601&m=61121&urllink=&afftrack=
If you’d like to support the channel, you can do that with the website.