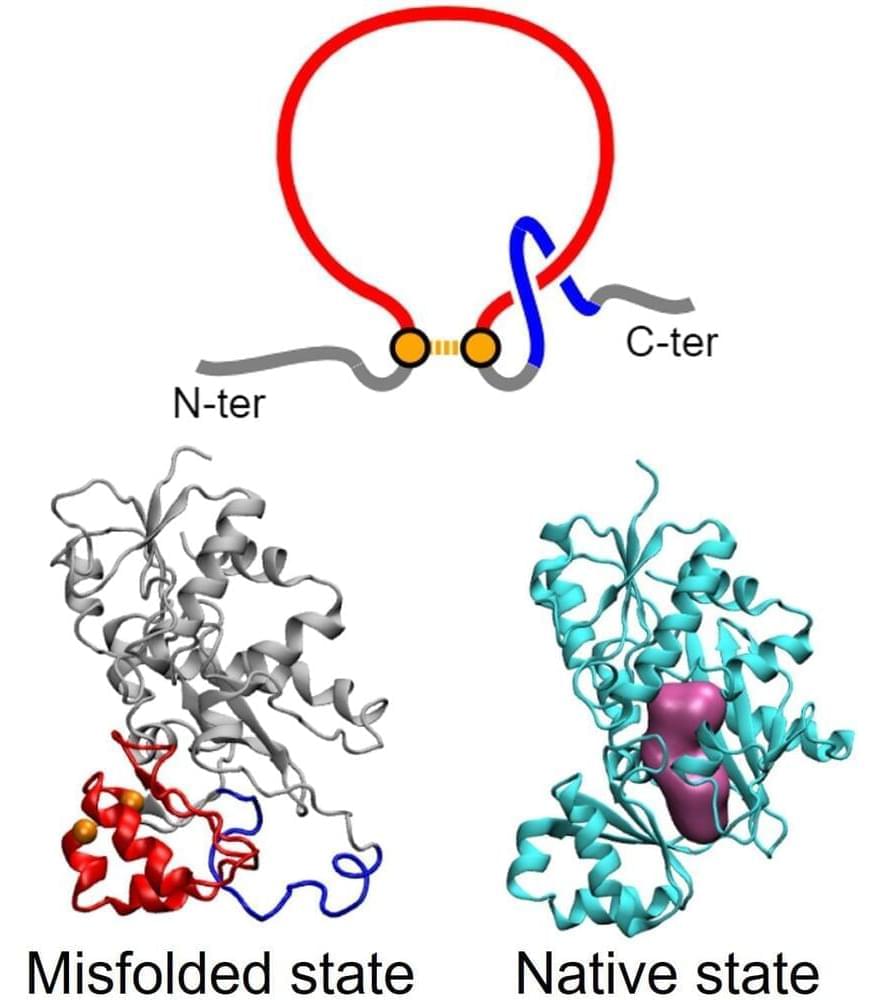
Imaging deep tissues with light is challenging. Visible light is often quickly absorbed and scattered by structures and molecules in the body, preventing researchers from seeing deeper than a millimeter within a tissue. If they do manage to probe further, substances like collagen or melanin often muddy the image, creating the equivalent of background noise through their natural fluorescence. As the authors explained, “Biological tissues have strong optical attenuation in the visible wavelength range (350–700 nm), due to the absorption of hemoglobin and melanin, as well as the tissue scattering, which fundamentally limits the imaging depth of high-resolution optical technologies.”
To wade out from these muddied waters, Yao and collaborator Vladislav Verkhusha, PhD, professor of genetics at Albert Einstein College of Medicine, developed a protein that absorbs and emits longer wavelengths of light in the near-infrared (NIR) spectrum. “Tissue is the most transparent in the 700‑1300 nm window of NIR light,” said Yao. “At those wavelengths, light can penetrate deeper into a tissue, and because there is less natural background fluorescence to filter out, we can take longer exposures and capture clearer images.”
Verkhusha and his lab used a process called directed molecular evolution to engineer their proteins, using photoreceptors normally found in bacteria as the basis for the structure. “The state-of-the-art NIR FPs were engineered from bacterial phytochrome photoreceptors (BphPs),” the team noted. “Applying rational design, we developed 17 kDa cyanobacteriochrome-based near-infrared (NIR-I) fluorescent protein, miRFP718nano.”