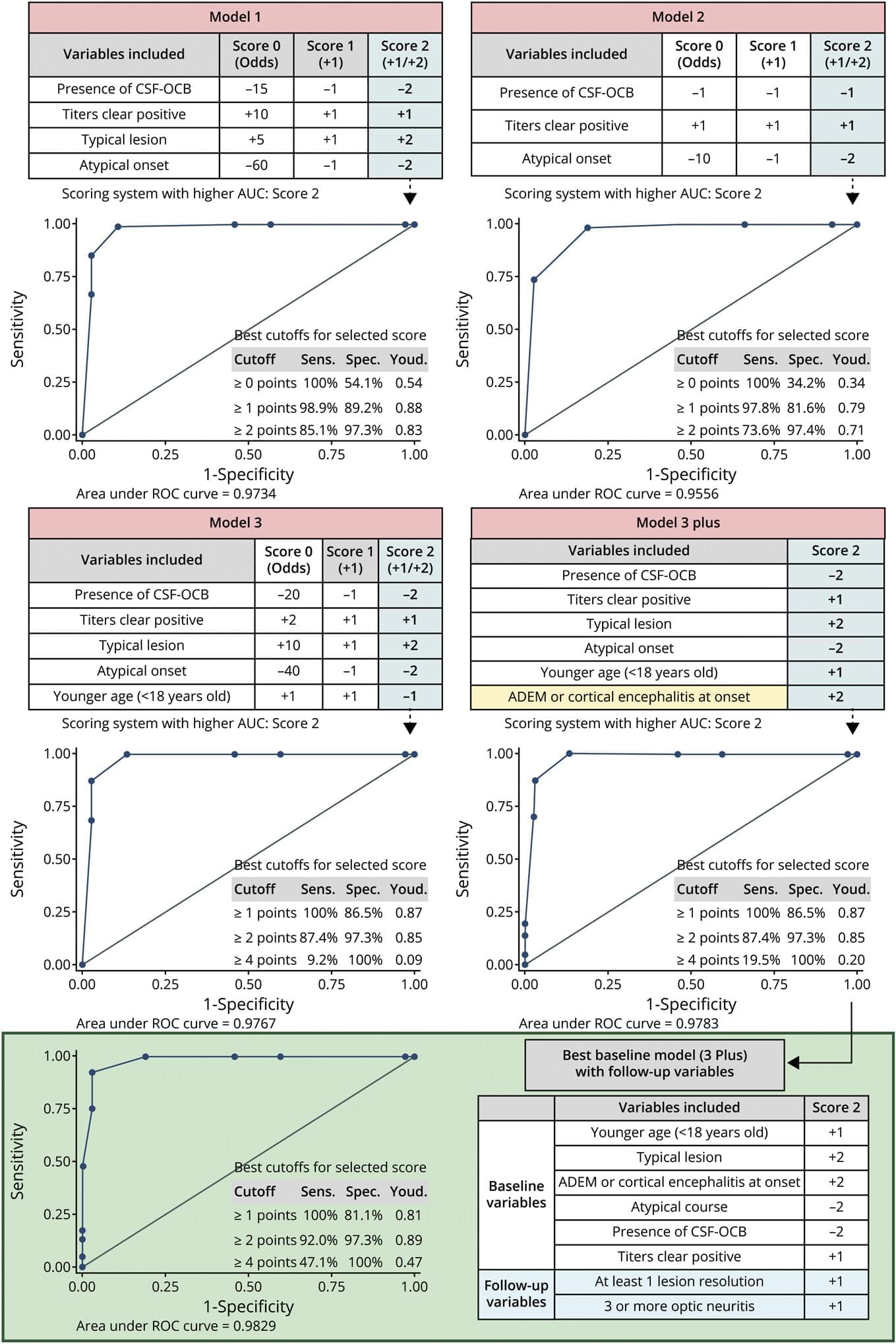
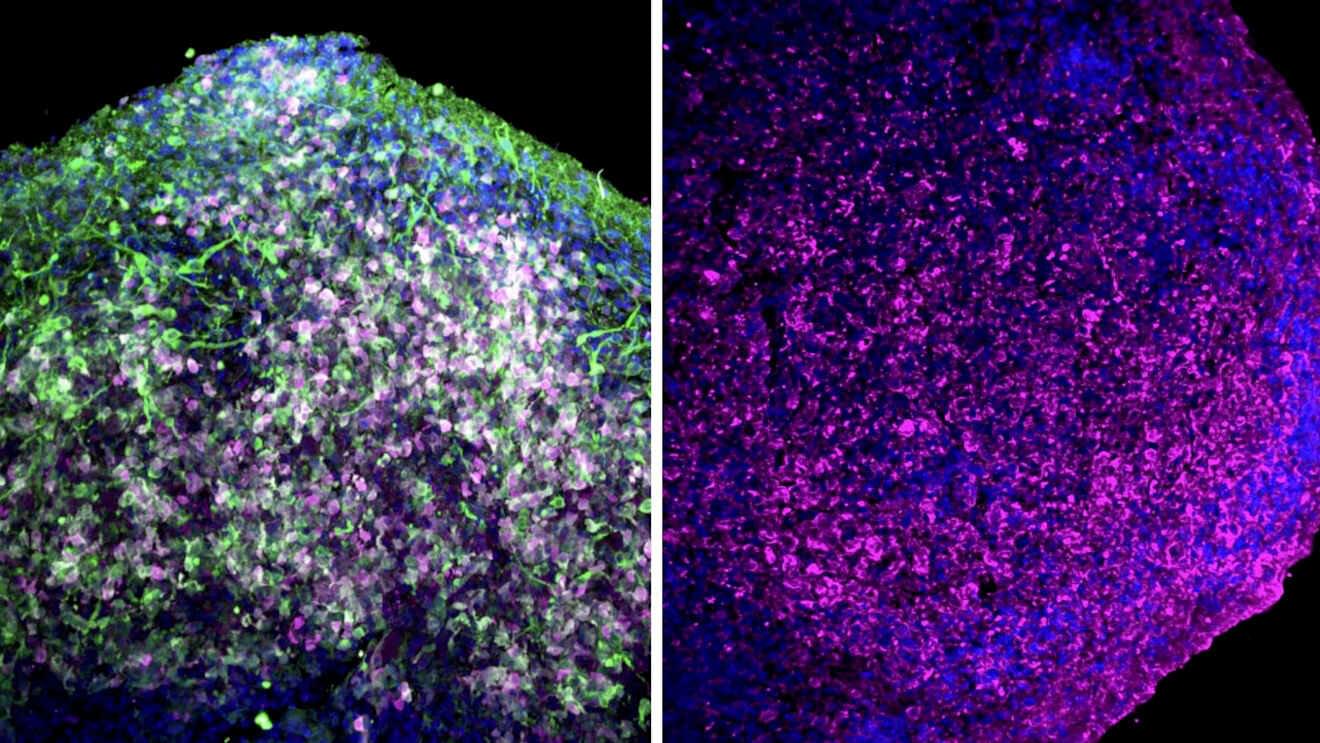
Organoids are miniature, simplified versions of an organ. Over the past two decades, scientists have developed them for the gut, lung, liver, mammary gland, brain, and more. Now, researchers at Yale School of Medicine (YSM) have organoid-ized the pineal gland, a small structure in the brain that regulates sleep patterns through its production of the hormone melatonin.
In a study published in Cell Stem Cell, the researchers demonstrate how pineal gland organoids can be used to study sleep dysfunction in conditions like Angelman syndrome, autism, and depression.
“In a number of neuropsychiatric conditions, severe sleep problems are a major symptom,” says In-Hyun Park, Ph.D., associate professor of genetics at YSM and senior author of the study. “With pineal gland organoids, we may be able to uncover the causes of those sleep disturbances and possibly identify treatments.”