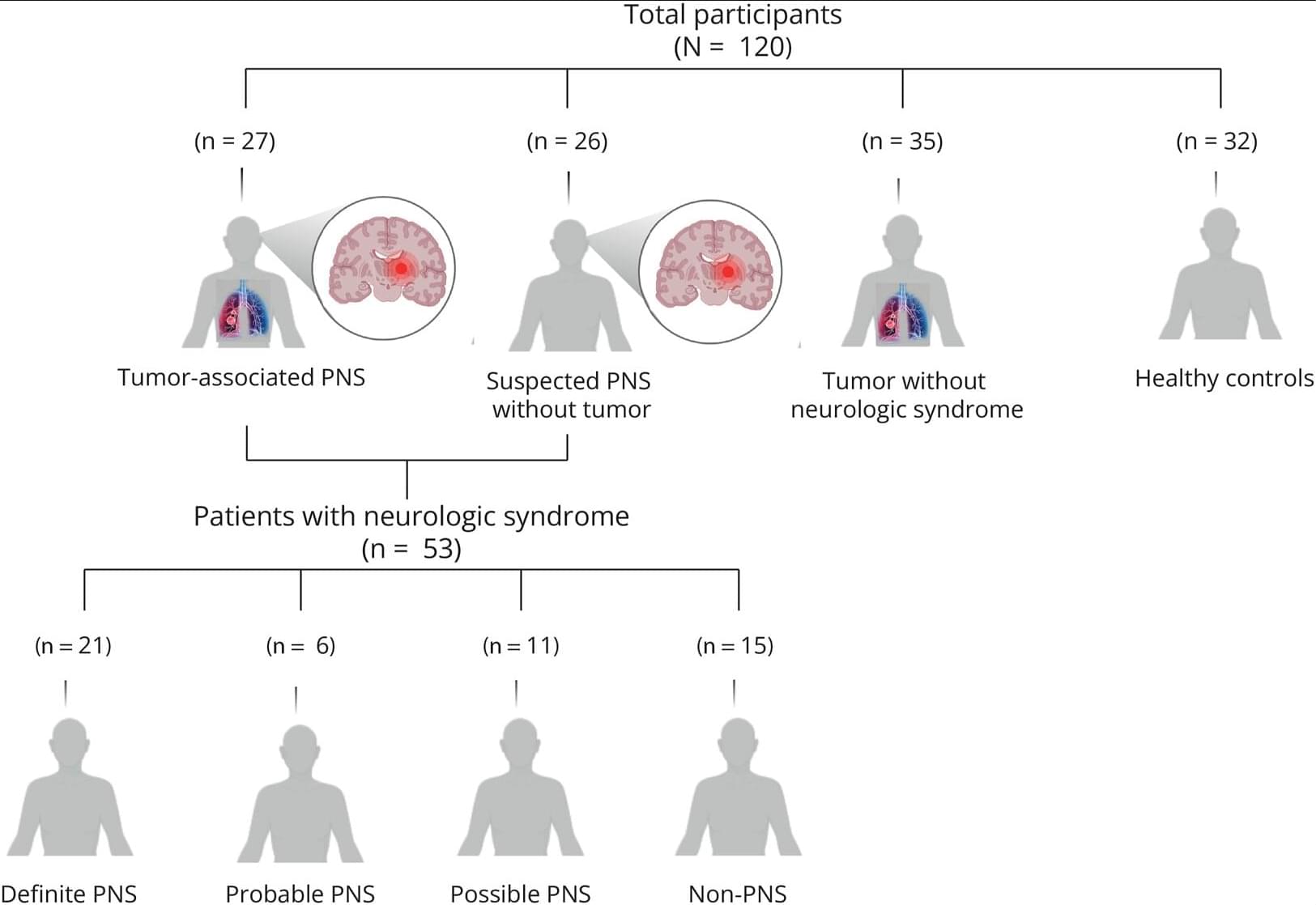
Class IV evidence that higher circulating blood levels of extracellular vesicles can distinguish between tumor-associated paraneoplastic neurologic syndrome (PNS) from suspected PNS without tumor.
Background and Objectives.
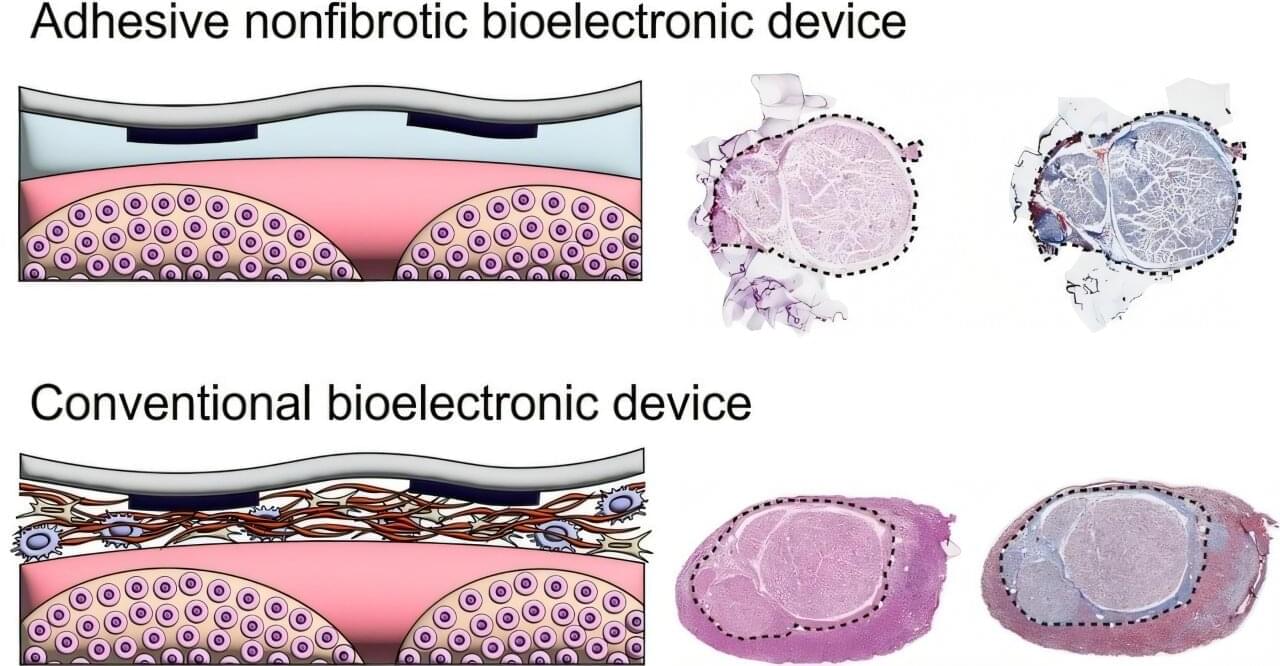
Peripheral nerves—the network connecting the brain, spinal cord, and central nervous system to the rest of the body—transmit sensory information, control muscle movements, and regulate automatic bodily functions. Bioelectronic devices implanted on these nerves offer remarkable potential for the treatment and rehabilitation of neurological and systemic diseases.
However, because the body perceives these implants as foreign objects, they often trigger the formation of dense fibrotic tissue at bioelectronic device–tissue interfaces, which can significantly compromise device performance and longevity.
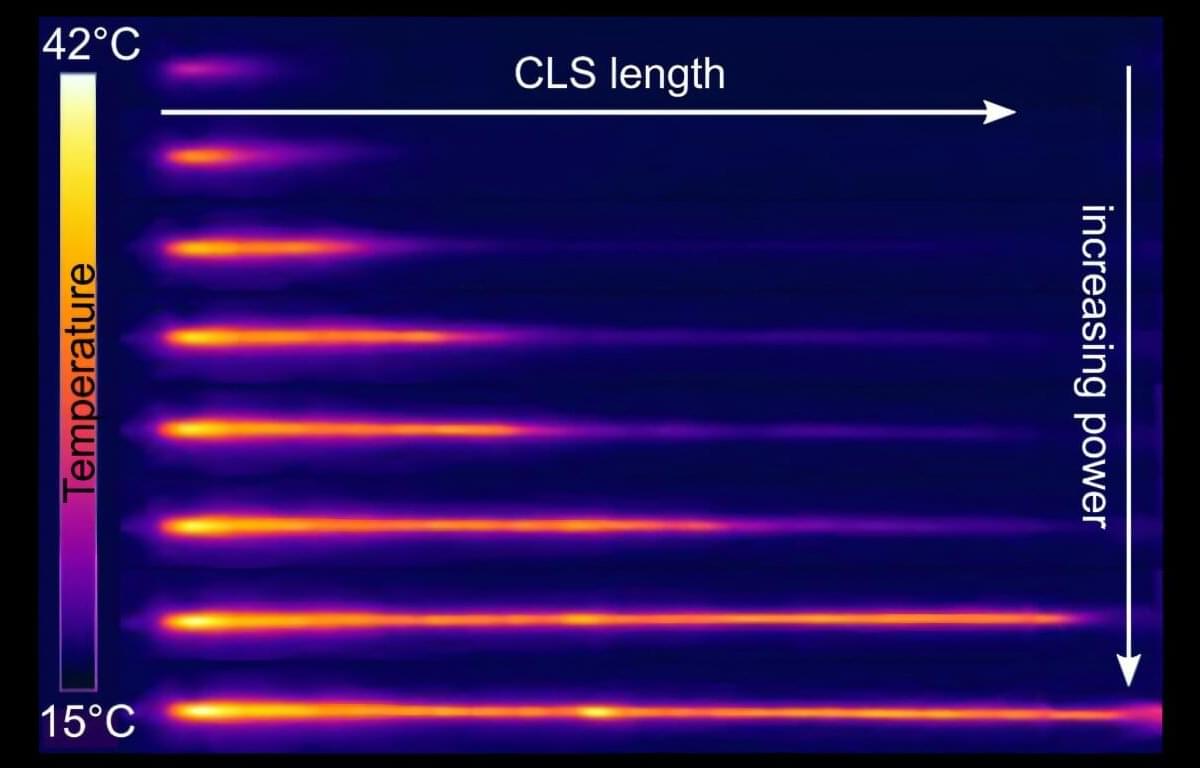
Thulium fiber lasers, operating at a wavelength of 2 micrometers, are valued for applications in medicine, materials processing, and defense. Their longer wavelength makes stray light less damaging compared to the more common ytterbium lasers at 1 micrometer.
Yet, despite this advantage, thulium lasers have been stuck at around 1 kilowatt of output power for more than a decade, limited by nonlinear effects and heat buildup. One promising route to break this barrier is inband pumping—switching from diode pumping at 793 nm to laser pumping at 1.9 µm. This approach improves efficiency and reduces heat, but it introduces new challenges for fiber components, especially the cladding light stripper (CLS).


Researchers peered through microscopes, hooked up electrodes, and built entire careers around one cell type: neurons. These electrically active cells were clearly the brain’s protagonists, zipping signals through our heads at lightning speed to create thoughts, memories, and movements. Everything else—especially the star-shaped cells called astrocytes that outnumber neurons—was dismissed as mere scaffolding. Glial cells, they were called: “glue.”
Inbal Goshen, a memory researcher at Hebrew University of Jerusalem, remembers feeling like an outsider when she started investigating astrocytes in the early 2010s. “Oh, that’s the weird one who works on astrocytes,” she imagined colleagues whispering at conferences. The skepticism was palpable. Yet new molecular tools had finally given her a way to peek into these mysterious cells, and what she found was too intriguing to ignore.
Unlike neurons, astrocytes don’t fire electrical signals. They were “electrically silent,” which is why they’d been ignored. But they were whispering in another language entirely: calcium. Using advanced imaging, researchers discovered that astrocytes communicate through slow, rhythmic waves of calcium signals—more like a gentle tide than neuron’s lightning strike. And their reach is astonishing: a single human astrocyte can touch up to two million synapses, the junctions where neurons meet. Their bushy tendrils fill every crevice of the brain, each cell nestling against neurons and blood vessels, creating an intimate, three-way relationship.
Memory research revealed another layer. Goshen’s team watched astrocytes in mice navigating toward water rewards. As the animals approached familiar prize locations, astrocyte activity slowly ramped up—but showed no response in new environments. The cells were encoding spatial memories, not just supporting them. Other labs found that astrocytes help stabilize and recall fear memories, their slow calcium signals perfectly suited to bridge the gap between learning something and remembering it days later. As neuroscientist Jun Nagai describes it, “Think of them as the brain’s long-exposure camera: they capture the trace of meaningful events that might otherwise fade too fast.”
Astrocytes make up one-quarter of the brain, but researchers are only now realizing their true value.
A rare and life-threatening kidney disease in children finally has an effective therapy, thanks in large part to pioneering research and clinical leadership from University of Iowa Health Care Stead Family Children’s Hospital.
The disease, known as C3 glomerulopathy (C3G), is an ultra-rare condition that primarily affects children and young adults. Only around 5,000 Americans have C3G, which causes progressive kidney damage, with more than half of patients reaching end-stage kidney failure within a decade of diagnosis.
Unlike previous treatments for C3G that aimed to alleviate the damaging inflammatory process of the disease, the new, first-of-its-kind drug directly targets the root cause of C3G dysfunction in the body’s complement system, a part of the immune response.

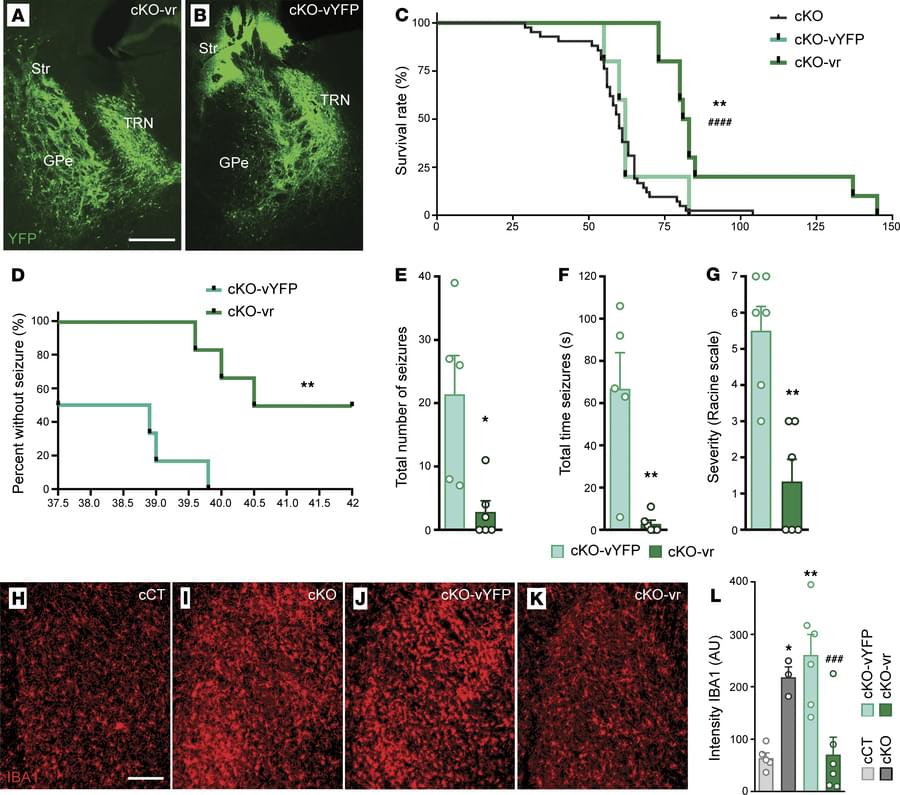
1 Institut de Neurociències, and.
2Department of Cell Biology, Physiology and Immunology, Universitat Autònoma de Barcelona, Bellaterra, Barcelona Spain.
3Institute of Neuroscience, CSIC-UMH, Alicante, Spain.
4Neurodegenerative Diseases Research Group, Vall d’Hebron Research Institute-Network Center for Biomedical Research in Neurodegenerative Diseases (CIBERNED), Barcelona, Spain.

The living cell, once thought to be a precise molecular factory, is turning out to be more like an improvising jazz ensemble. The old dogma—one gene, one protein, one function—has collapsed. Molecular biologist Ewa Grzybowska argues that recent discoveries show that proteins can switch folds, shift shapes, or even remain gloriously unstructured, improvising their roles as they go. Genes are not blueprints but texts, open to continuous interpretation by cells. Life, it turns out, is not built like a machine but is instead fluid, improvisational, and brimming with creative possibility.
1. The old paradigms in biology
When Watson and Crick decoded DNA in 1953 and the mechanism of protein-making was discovered, we obtained an extraordinary tool to explain the inner workings of life. The basic principle of making one protein from one DNA template (gene) with the assistance of one messenger RNA (mRNA) and several well-defined amino-acid-transporting RNAs (tRNAs) was so successful that it has been enshrined in millions of textbooks and not questioned for a long time.