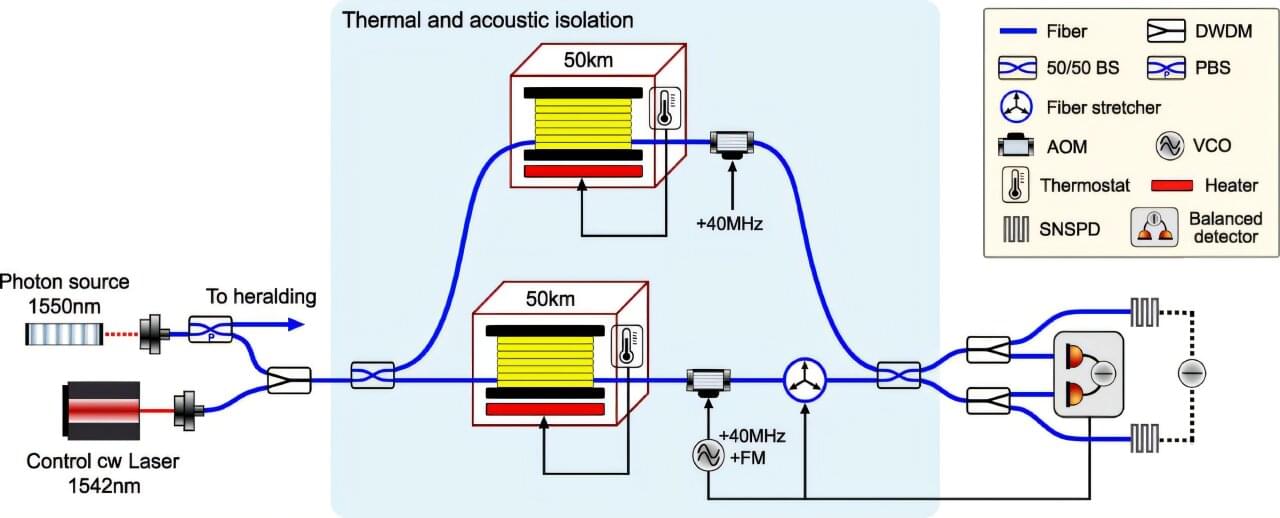
Assistant Professor Haocun Yu is something of a scientific diplomat. In a recent Physical Review Letters publication, she and her colleagues show how a tabletop experiment can bring together two bedrock physics theories that have never been fully reconciled.
More than a century ago, Albert Einstein gave us the theory of general relativity, describing gravity in relation to space and time on a large scale. Within a decade, physicists were developing a deeper knowledge of quantum mechanics, the laws that govern the subatomic world, including atoms, photons and other microscopic systems.
“Quantum mechanics and general relativity are two of the most successful theories in physics, but they describe nature in very different ways,” Yu explained.