Almost half a century ago, a remarkable molecule called metallocene took center stage in chemistry, earning Geoffrey Wilkinson and Ernst Otto Fischer the Nobel Prize. These organic compounds, made of a transition metal “sandwiched” between two flat, ring-shaped organic layers, have since become an integral part of new-age polymers, materials, and pharmaceuticals.
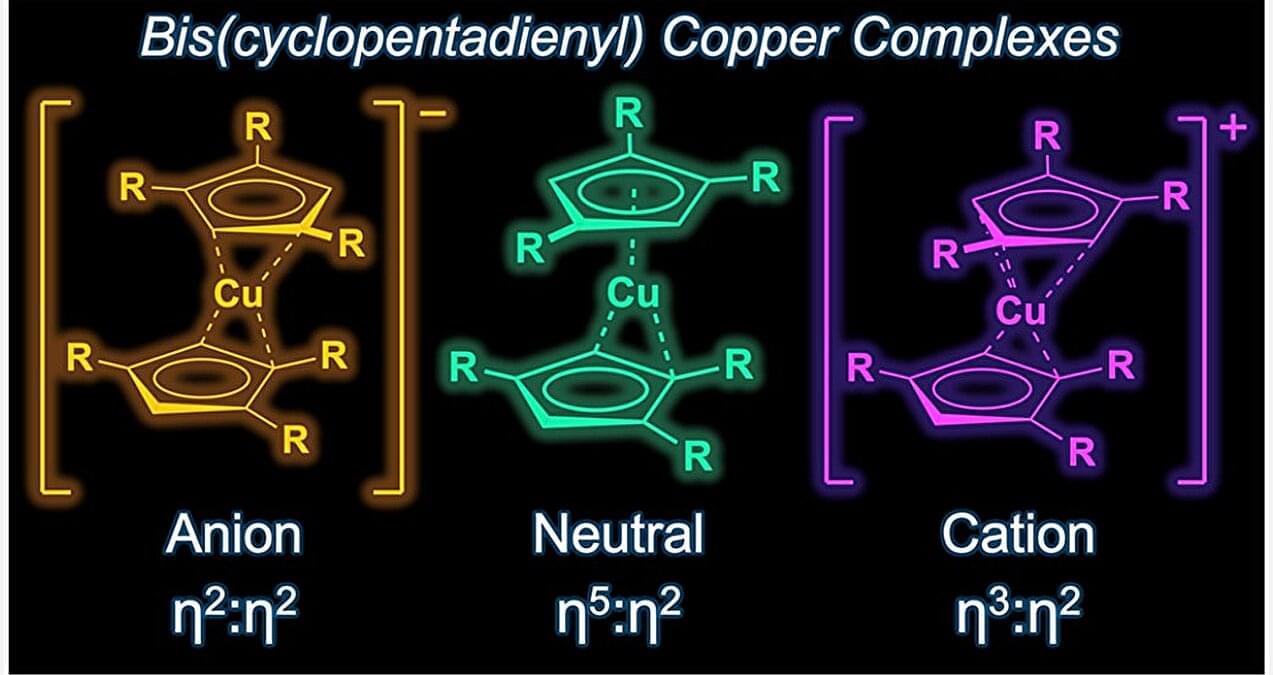
In their recent work published in the Journal of the American Chemical Society, a team from University of California brought metallocene back into the limelight with the synthesis of cuprocenes—the first stable version of neutral copper metallocene with the chemical formula Cpttt 2 Cu where Cpttt stands for C5H2tBu3 or bis(tri-tert-butylcyclopentadienyl) ligand. This new complex of copper has blue-green crystals and is stable at room temperature, away from light.
They also produced two new forms of cuprocene: a colorless, negatively charged version via reduction, and a purple, positively charged version via oxidation.