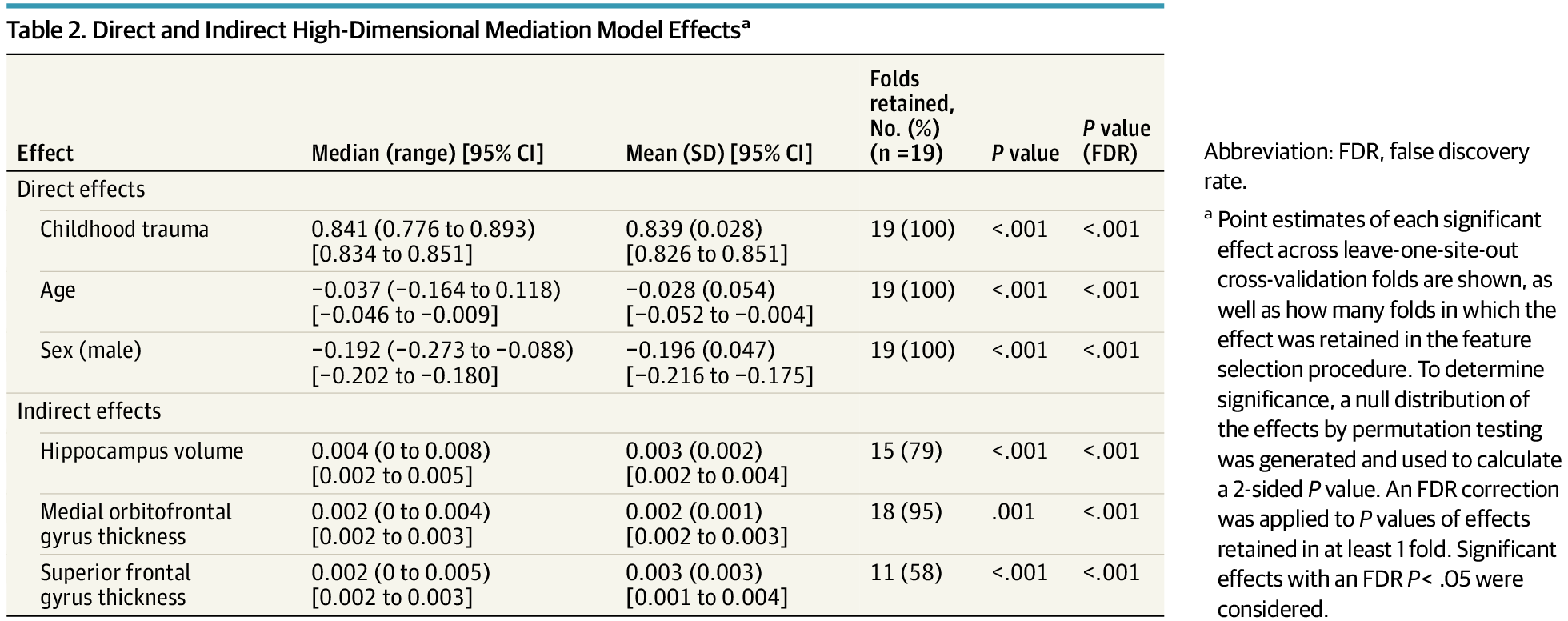
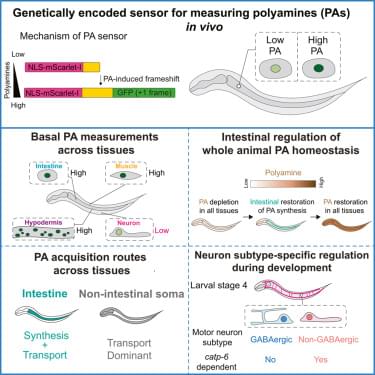
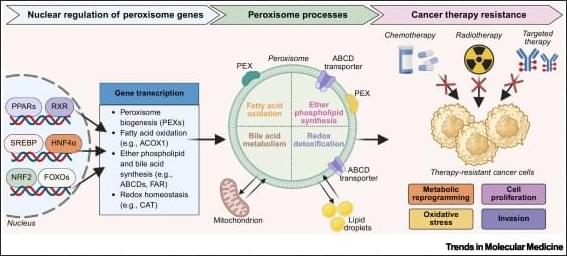
Neuroscientists are uncovering how communication between different parts of the brain creates the experience of being “you.”
📺 Watch the full episode here: https://www.pbs.org/wgbh/nova/video/y…
💙 Help us bring the universe closer to home. Your donation ensures that NOVA’s deep-space discoveries and quantum physics explorations remain free and inspiring for the next generation of scientists. Support fact-based storytelling today. https://bit.ly/48UnsrD
📰 Stay up to date on the latest science discoveries, full episodes, articles, videos, and more by signing up for NOVA’s newsletter here: https://to.pbs.org/4brCC7Y
▶️ Stream NOVA documentaries \& more with PBS Documentaries on Prime Video: https://amzn.to/4hXBlsu.
🛍️ Shop official NOVA 50th anniversary merch: https://nova-shop.org/