Over one billion people worldwide are over 60, and the population is projected to more than double by 2050. But as more people live into their 60s, 70s, and 80s, health care systems across the globe may face new challenges as they attempt to manage associated increases in age-related disease.
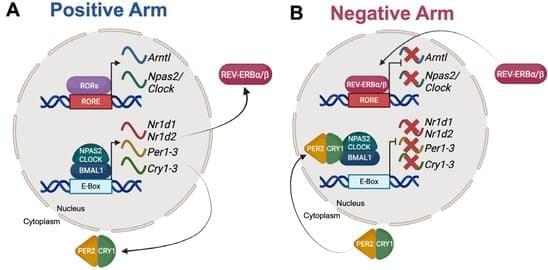
Metabolic biologist Andreas Stahl and preeminent longevity researcher Irina Conboy argue that the graying of the global population underscores the need to understand aging as a biological process, and how it might be slowed or reversed. Longevity therapeutics, however, are expensive to develop, and the lack of rapid, reliable tools to study human aging can make it difficult to test these next-generation therapies. While animal models can provide important data, there are often many caveats when applying those findings to human biology during trials.
“Over $130 billion is spent on drug development each year in the United States, but over 90% end up failing in clinical trials,” explained Stahl, the Ruth Okey Professor in the Department of Metabolic Biology and Nutrition (MBN) and a member of the California Institute for Quantitative Biosciences at UC Berkeley (QB3-Berkeley). “Pharmaceutical developers and regulators such as the US Food and Drug Administration are increasingly realizing that we need to change our drug development pipeline and make it more relevant to human biology.”