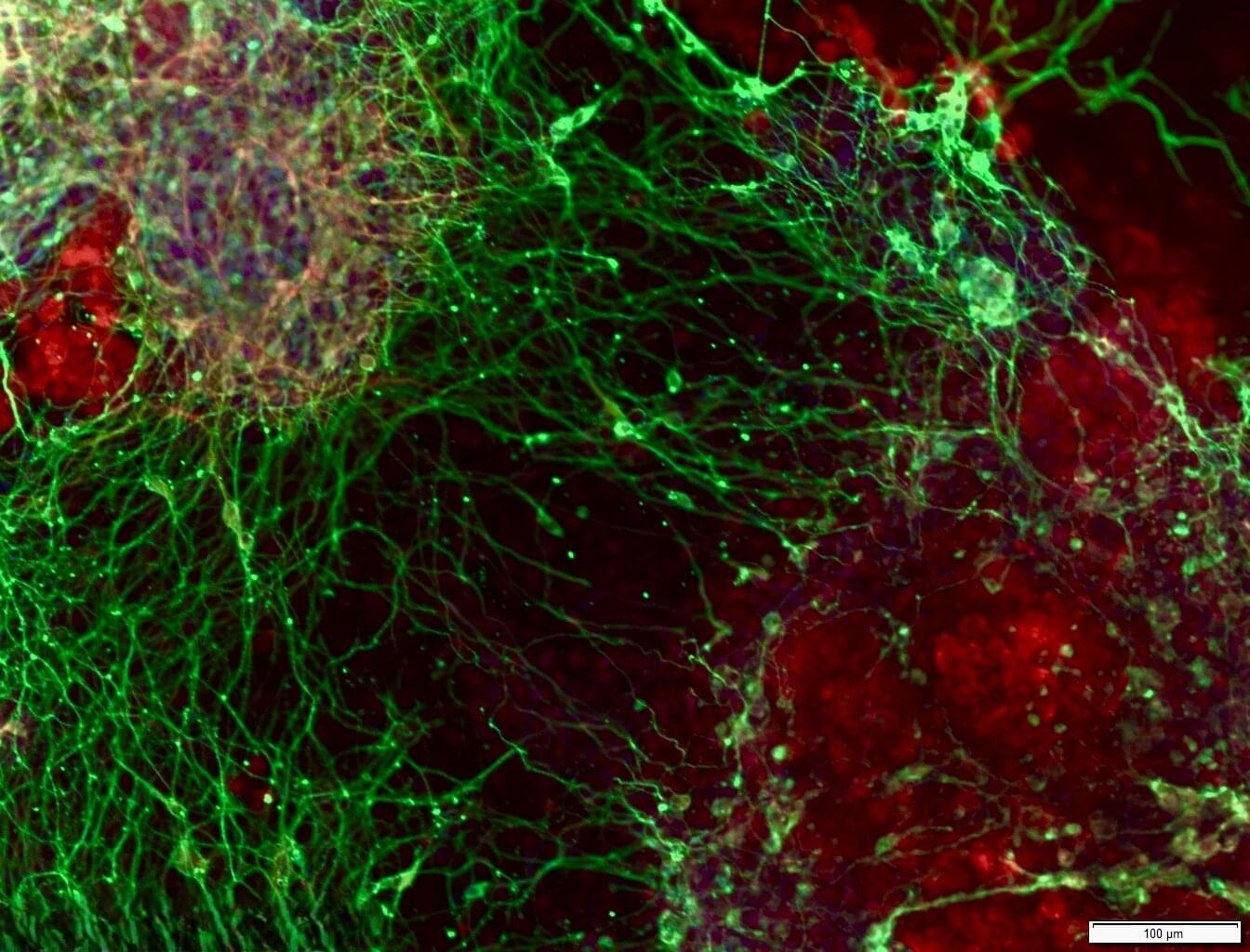
Which genes are required for turning embryonic stem cells into brain cells, and what happens when this process goes wrong? In a new study published today in Nature Neuroscience, researchers led by Prof. Sagiv Shifman from The Institute of Life Sciences at The Hebrew University of Jerusalem, in collaboration with Prof. Binnaz Yalcin from INSERM, France, used genome-wide CRISPR knockout screens to identify genes that are needed for early brain development.
The study set out to answer a straightforward question: which genes are required for the proper development of brain cells?
Using CRISPR-based gene-editing methods, the researchers systematically and individually “switched off” roughly 20,000 genes to study their role in brain development. They performed the screen in embryonic stem cells while the cells changed into brain cells. By disrupting genes one by one, the team could see which genes are required for this transition to proceed normally.