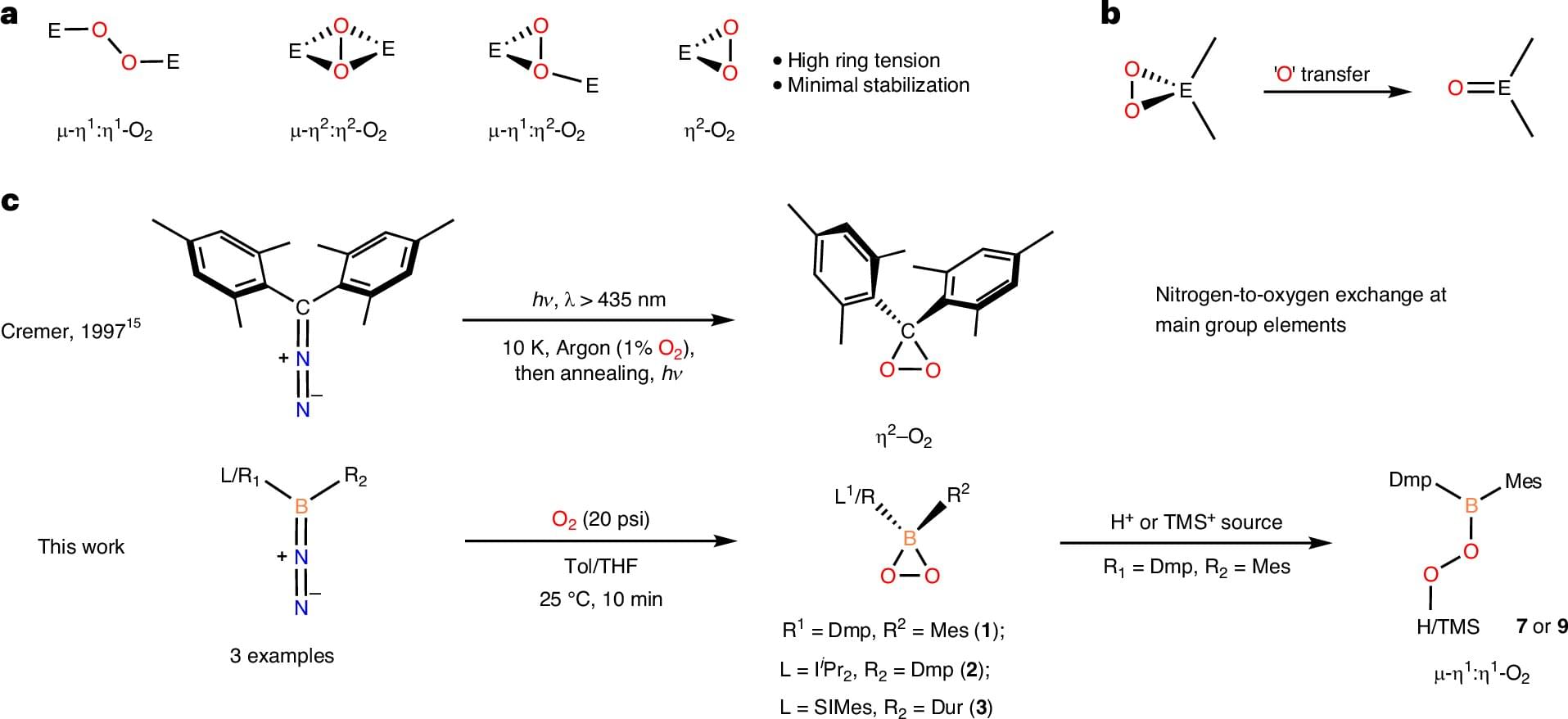
Oxygen is a cornerstone of chemistry, largely because it is so good at building the organic molecules that make up our world. Some oxygen-based compounds called peroxides are famous for being highly reactive—they act like oxygen delivery trucks, transferring atoms to other molecules. This process is essential for everything from creating new medicines to industrial manufacturing.
In a study published in Nature Chemistry, researchers from the labs of MIT professors Christopher C. Cummins and Robert J. Gilliard, Jr. have revealed a brand-new type of peroxide containing boron. This molecule, called a dioxaborirane, represents a major advance in a field where such structures were long-proposed, but considered too unstable to actually isolate.