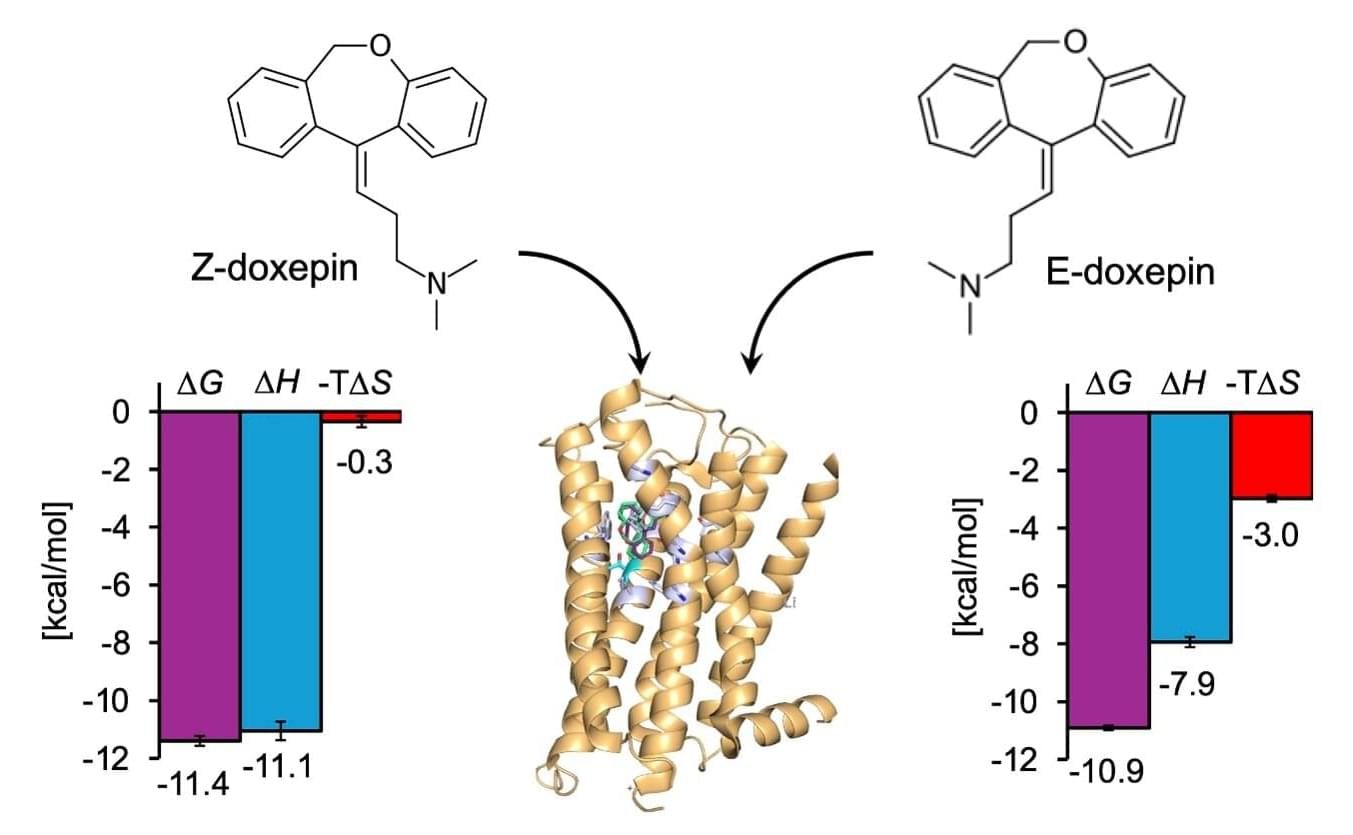
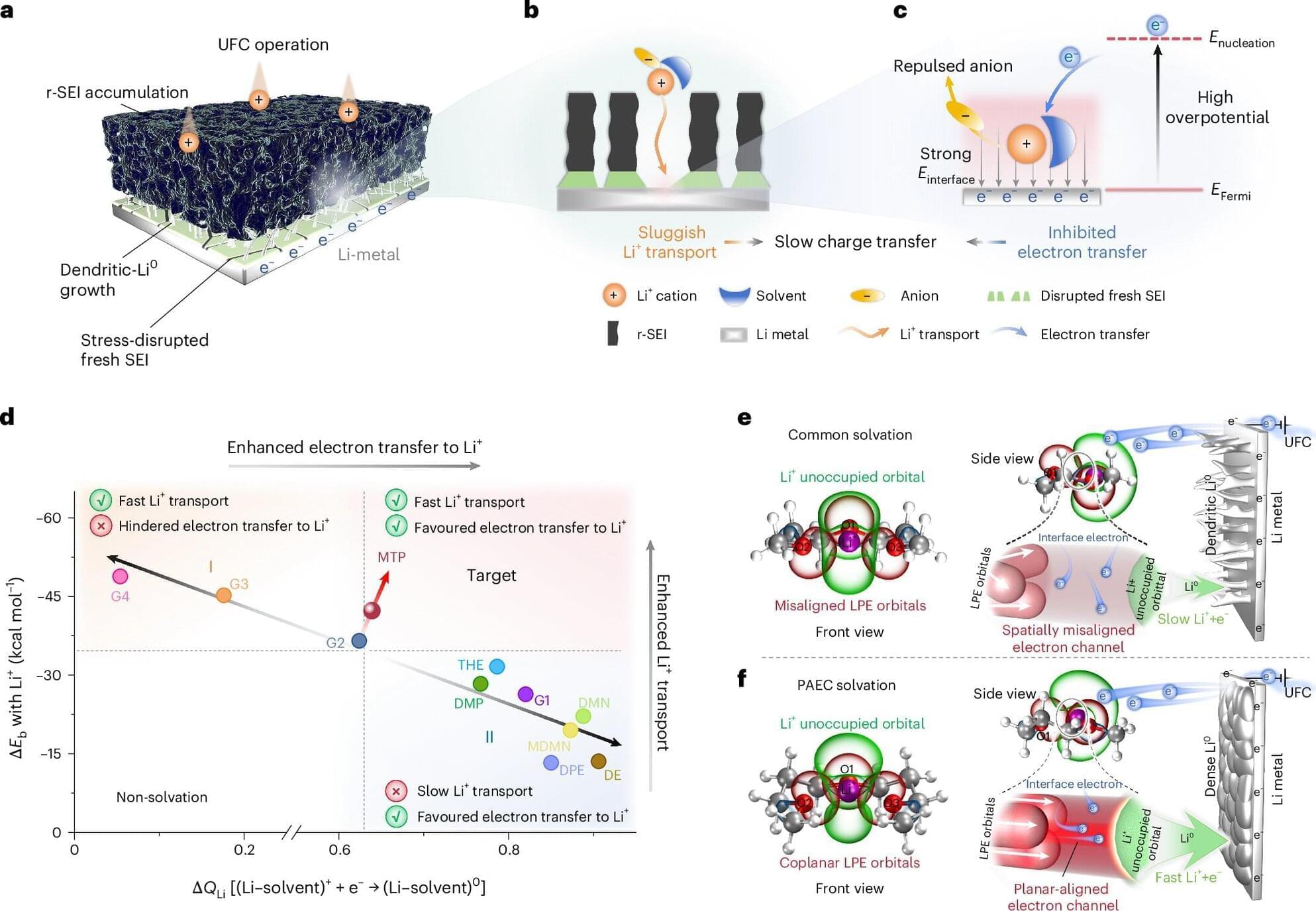
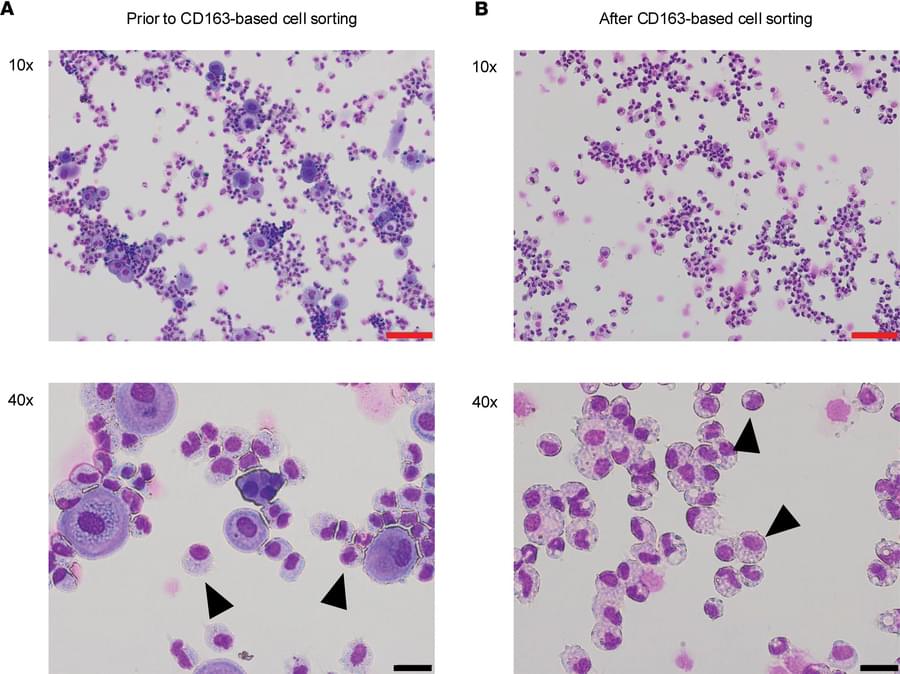
Concordia researchers have developed a new 3D-printing technique that uses sound waves to directly print tiny structures onto soft polymers like silicone with far greater precision than before. The approach, called proximal sound printing, opens new possibilities for manufacturing microscale devices used in health care, environmental monitoring and advanced sensors. It is described in the journal Microsystems & Nanoengineering.
The technique relies on focused ultrasound to trigger chemical reactions that solidify liquid polymers exactly where printing is needed. Unlike conventional methods that rely on heat or light, sound-based 3D-printing works with key materials used in microfluidic devices, lab-on-a-chip systems and soft electronics that are hard to print at small scales.
This work builds on the research team’s earlier breakthrough in direct sound printing, which first showed that ultrasound could be used to cure polymers on demand. While that earlier method demonstrated the concept, it struggled with limited resolution and consistency. The new proximal approach places the sound source much closer to the printing surface, allowing far tighter control.