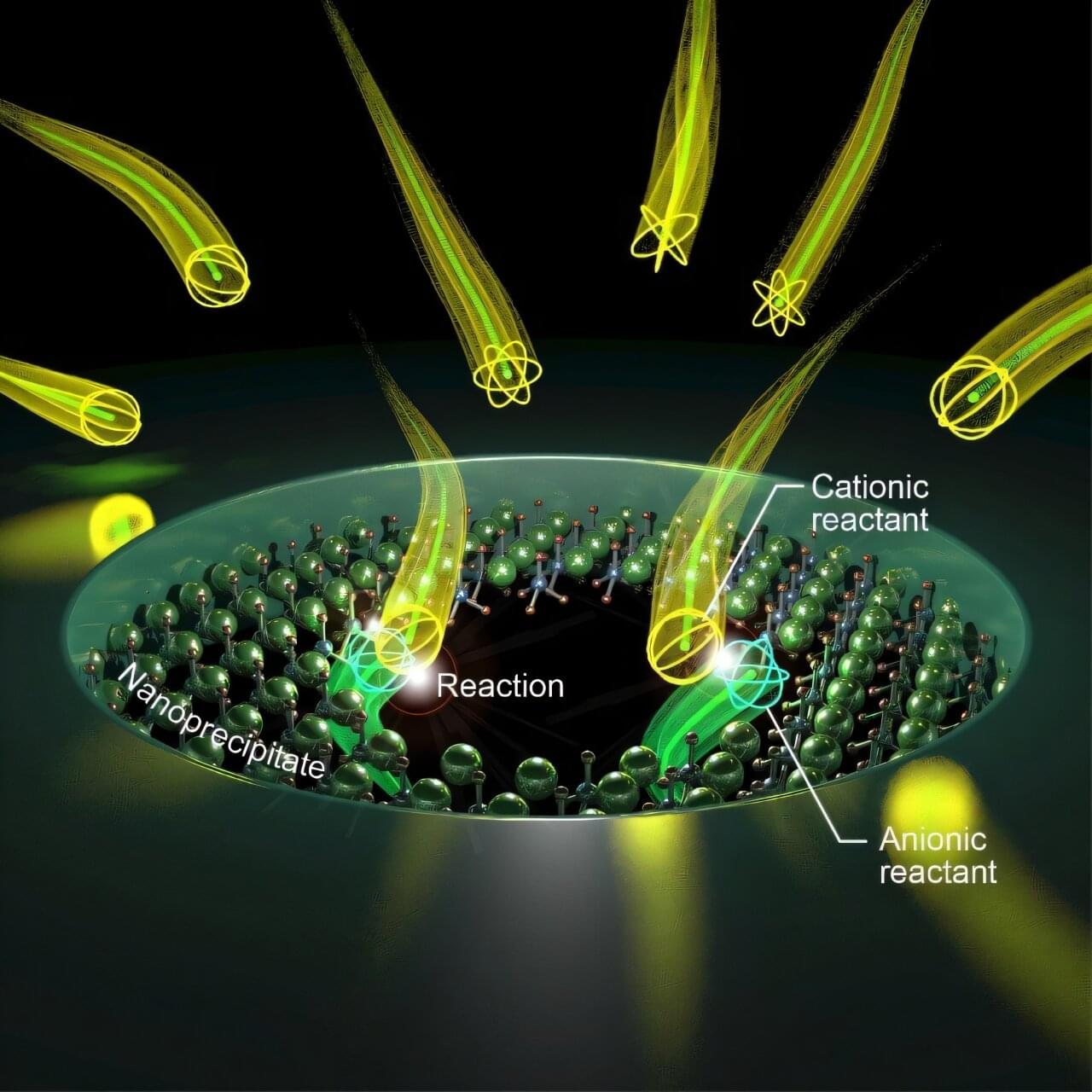
Ion channels are narrow passageways that play a pivotal role in many biological processes. To model how ions move through these tight spaces, pores need to be fabricated at very small length scales. The narrowest regions of ion channels can be just a few angstroms wide, about the size of individual atoms, making reproducible and precise fabrication a major challenge in modern nanotechnology.
In a study published in Nature Communications, researchers at The University of Osaka have addressed this challenge by using a miniature electrochemical reactor to create ultra-small pores approaching subnanometer dimensions.
In biological cells, ions flow in and out through channels in cell membranes. This ion flow is the basis for generating electrical signals, such as nerve impulses that trigger muscle contraction. The channels themselves are made of proteins and can have angstrom-wide narrow regions. Conformational changes of these proteins in response to external stimuli open and close the channels.