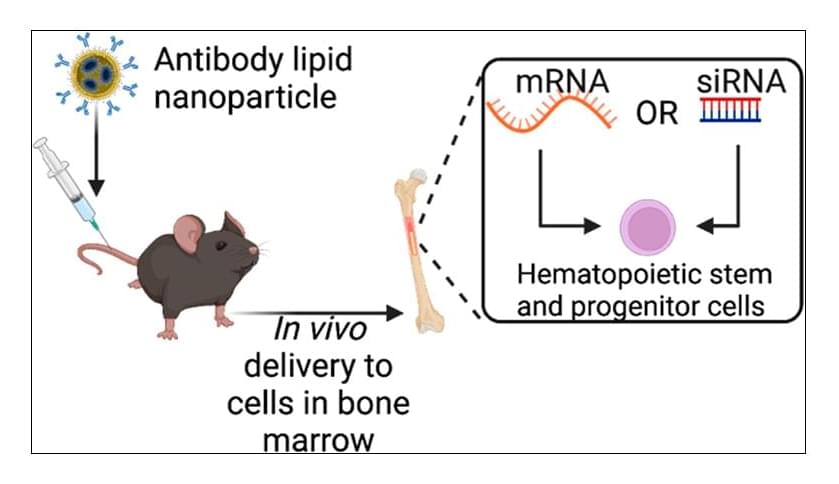
A nicely concise paper on antibody-linked lipid nanoparticles which target hematopoietic stem and progenitor cells in vivo, important yet tricky cell types to transduce for hematological gene therapy.
Ex vivo autologous hematopoietic stem cell (HSC) gene therapy has provided new therapies for the treatment of hematological disorders. However, these therapies have several limitations owing to the manufacturing complexities and toxicity resulting from required conditioning regimens. Here, we developed a c-kit (CD117) antibody-targeted lipid nanoparticle (LNP) that, following a single intravenous injection, can deliver RNA (both siRNA and mRNA) to HSCs in vivo in rodents. This targeted delivery system does not require stem cell harvest, culture, or mobilization of HSCs to facilitate delivery. We also show that delivery of Cre recombinase mRNA at a dose of 1 mg kg–1 can facilitate gene editing to almost all (∼90%) hematopoietic stem and progenitor cells (HSPCs) in vivo, and edited cells retain their stemness and functionality to generate high levels of edited mature immune cells.