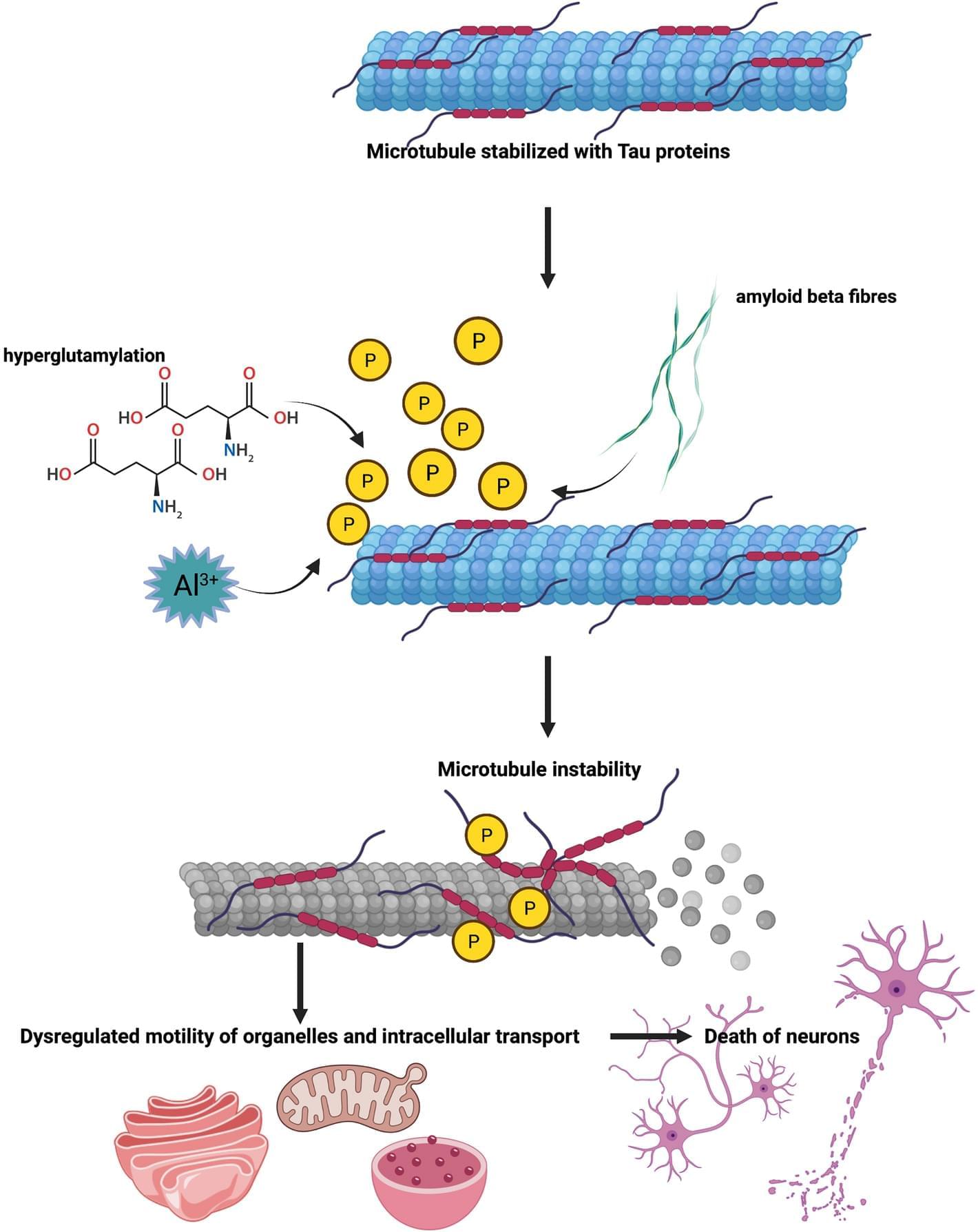
A wealthy family fighting its own disease boosted research on a little-studied brain protein, progranulin. Can it spur new dementia treatments?
Bluefield investigators, and eventually drug companies, saw something compelling about FTD-GRN, the form of the condition Alice had. In other genetic neurodegenerative disorders, such as familial Alzheimer’s and Huntington disease, mutations spark the production of toxic proteins, generating complex cascades of pathology. But the culprit mutations driving FTD-GRN block progranulin production, leaving carriers with less than half as much of the protein as noncarriers. Many dementia researchers came to describe FTD-GRN as a “low-hanging fruit” among neurodegenerative diseases, using words such as “intuitive” and “tractable” to characterize its biology. The solution seemed obvious: A treatment just needed to raise progranulin levels in the brain.
Fueled in part by that confidence, six clinical trials have been launched to test progranulin-boosting therapies in FTD-GRN. Companies also hope the anti-inflammatory properties of a progranulin-boosting agent could help in Parkinson’s disease, Alzheimer’s, amyotrophic lateral sclerosis (ALS), and FTD caused by other mutations or without a known genetic cause.
All has not gone according to plan, however. In October 2025, a landmark phase 3 clinical trial of a progranulin-boosting drug in people with FTD-GRN did not keep their disease from progressing. In February, a small trial of a gene therapy delivering a healthy copy of GRN to the brain was halted, also for lack of effect.