This is a ~57 minute talk titled “The Bioelectric Interface to the Collective Intelligence of Morphogenesis: development, regeneration, cancer, and beyond” which I gave at a UCSF seminar for an audience of graduate students and post-docs in Biophysics, Bioinformatics, and Chemical Biology. I covered the role of bioelectricity as cognitive glue underlying high-level adaptive plasticity in living tissue, recent progress in exploiting that interface, and new developments in research platforms for this field.
Category: biotech/medical – Page 117
Brave New Biology: Intelligence Trumps DNA — with Dr. Michael Levin and Dr. John Vervaeke
Dr. Michael Levin is a professor in the Department of Biology at Tufts University and an associate faculty member at the Wyss Institute at Harvard. He directs the Allen Discovery Center at Tufts, where his team integrates biophysics, computational modeling, and behavioral science to study how cellular collectives make decisions during embryogenesis, regeneration, and cancer.
Levin’s research centers on diverse forms of intelligence and unconventional embodied minds, bridging conceptual theory, experimental biology, and translational work aimed at regenerative medicine. His lab also pioneers efforts in artificial intelligence and the bioengineering of novel living machines.
Read more about Dr. Michael Levin’s work: https://drmichaellevin.org/
X: https://twitter.com/drmichaellevin.
YouTube: @drmichaellevin
John Vervaeke’s YouTube channel: @johnvervaeke
📖 Let’s take our stories back. Check out our latest book in the Tales for Now and Ever series, Rapunzel and the Evil Witch: https://rapunzelbook.com/
Join Fr. Stephen De Young in his Jubilees and the Nephilim course, now streaming live on The Symbolic World: https://www.thesymbolicworld.com/cour… 00:00 — Coming up 01:14 — Intro music 01:40 — Introduction 02:23 — What Michael does 06:19 — Example experiments 07:51 — Memories outside the brain 12:46 — Terminology: memory 13:59 — Communicate to biological cells 15:54 — Limitations? 17:39 — Platonic patterns 34:06 — Incarnation and constraints 39:26 — Causes 49:28 — New beings in new spaces 52:25 — What the Enlightenment dismissed 55:32 — Molecular medicine 57:36 — Subtle bodies 01:00:45 — Ethics 01:03:37 — Medical and meaning applications 01:11:42 — Frightening 01:14:31 — Against the status quo 01:19:03 — Should we dabble in this technology? 💻 Website and blog: http://www.thesymbolicworld.com 🔗 Linktree: https://linktr.ee/jonathanpageau 🔒 BECOME A PATRON: https://thesymbolicworld.com/subscribe Our website designers: https://www.resonancehq.io/ My intro was arranged and recorded by Matthew Wilkinson: https://matthewwilkinson.net/
Current & Future Role of Anktiva (N-803): Dr. Patrick Soon-Shiong
In this episode of the Oncology Brothers podcast, we engaged in a thought-provoking discussion with Dr. Patrick Soon-Shiong, a pioneer in oncology and the innovator behind the approval of nab-paclitaxel (Abraxane). We delved into the exciting potential of Anktiva (N-803), an IL-15 receptor super agonist designed to expand and activate natural killer (NK) cells and CD8 T cells, with the hope of revolutionizing cancer treatment.
Dr. Soon-Shiong shared insights into the mechanism of action of Anktiva, its current approvals in non-muscle invasive bladder cancer, and extended approval in non-small cell lung cancer in Saudi Arabia, and the promising clinical trial data that suggests a significant increase in overall survival for patients. The conversation also touched on the importance of restoring lymphocyte counts and the implications for treating various tumor types.
Join us as we explore the future of immunotherapy, the challenges of regulatory approval, and the potential for Anktiva to change the landscape of cancer treatment.
Key Topics:
• Mechanism of action of Anktiva.
• Current approvals and clinical trial data.
• The role of lymphocyte counts in cancer treatment.
• Future directions for immunotherapy.
Follow us on social media:
• X/Twitter: / oncbrothers.
• Instagram: / oncbrothers.
• Website: https://oncbrothers.com/
Don’t forget to like, subscribe, and hit the notification bell for more discussions on the latest in oncology!
#Oncology #CancerTreatment #Immunotherapy #IL15Agonist #OncologyBrothers

Building blocks of life discovered in Bennu asteroid rewrite origin story
Amino acids, the building blocks necessary for life, were previously found in samples of 4.6-billion-year-old rocks from an asteroid called Bennu, delivered to Earth in 2023 by NASA’s OSIRIS-REx mission. How those amino acids—the molecules that create proteins and peptides in DNA—formed in space was a mystery, but new research led by Penn State scientists shows they could have originated in an icy-cold, radioactive environment at the dawn of Earth’s solar system.
According to the researchers, who published new findings in the Proceedings of the National Academy of Sciences, some amino acids in the asteroid Bennu samples likely formed in a different way than was previously thought, in the harsh conditions of the early solar system.
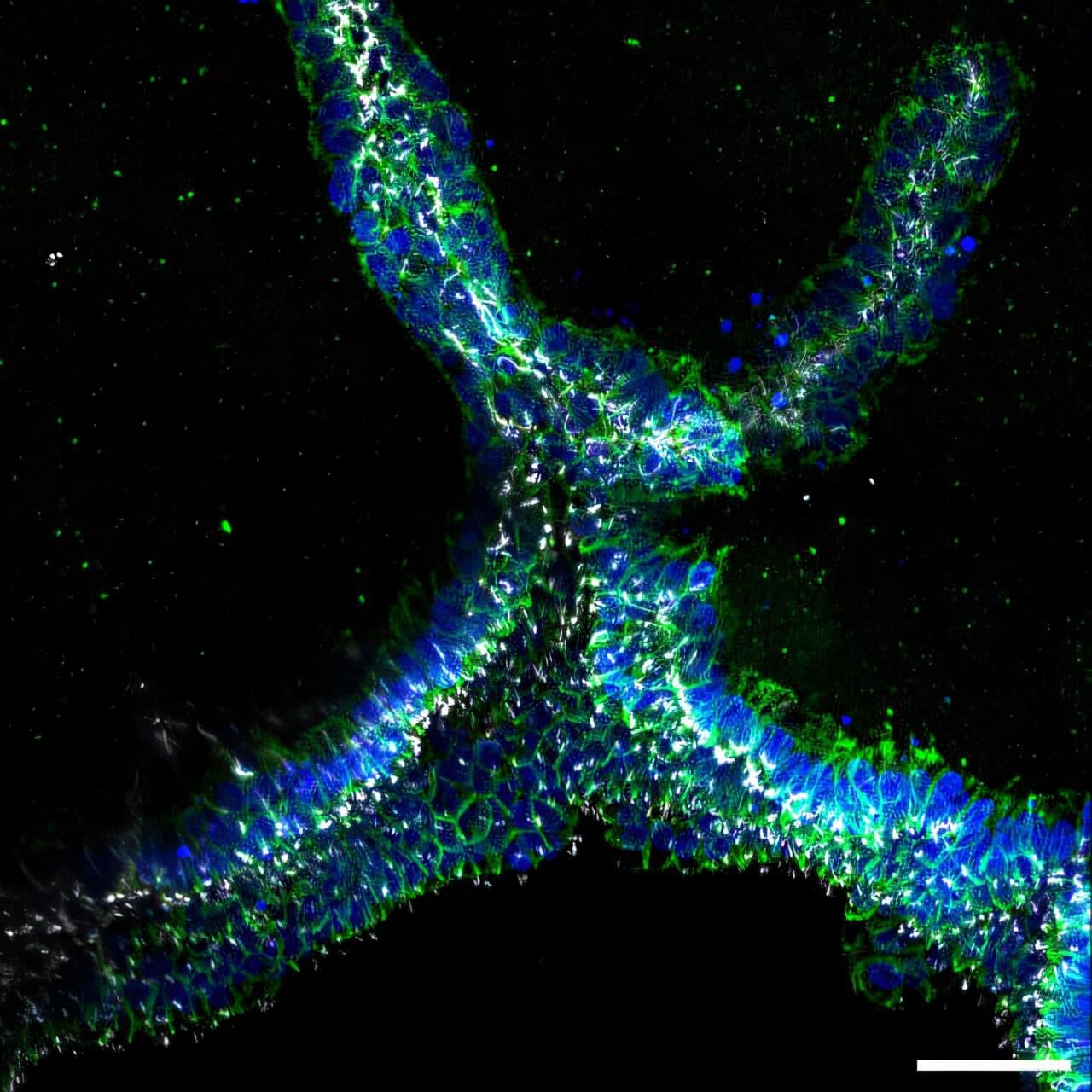
Bioengineers build branched, perfusable kidney collecting ducts using 3D bioprinting
The human kidney filters about a cup of blood every minute, removing waste, excess fluid, and toxins from it, while also regulating blood pressure, balancing important electrolytes, activating Vitamin D, and helping the body produce red blood cells. This broad range of functions is achieved in part via the kidney’s complex organization. In its outer region, more than a million microscopic units, known as nephrons, filter blood, reabsorb necessary nutrients, and secrete waste in the form of urine.
To direct urine produced by this enormous number of blood-filtering units to a single ureter, the kidney establishes a highly branched three-dimensional, tree-like system of “collecting ducts” during its development. In addition to directing urine flow to the ureter and ultimately out of the kidney, collecting ducts reabsorb water that the body needs to retain, and maintain, the body’s balance of salts and acidity at healthy levels.
Finding ways to recreate this system of collecting ducts is the focus of researchers and bioengineers who are interested in understanding how duct defects cause certain kidney diseases, underdeveloped kidneys, or even the complete absence of a kidney. Being able to fabricate the kidney’s plumbing system from the bottom up would be a giant step toward tissue replacement therapies for many patients waiting for a kidney donation: In the U.S. alone, 90,000 patients are on the kidney transplant waiting list. However, rebuilding this highly branched fluid-transporting ductal system is a formidable challenge and not possible yet.
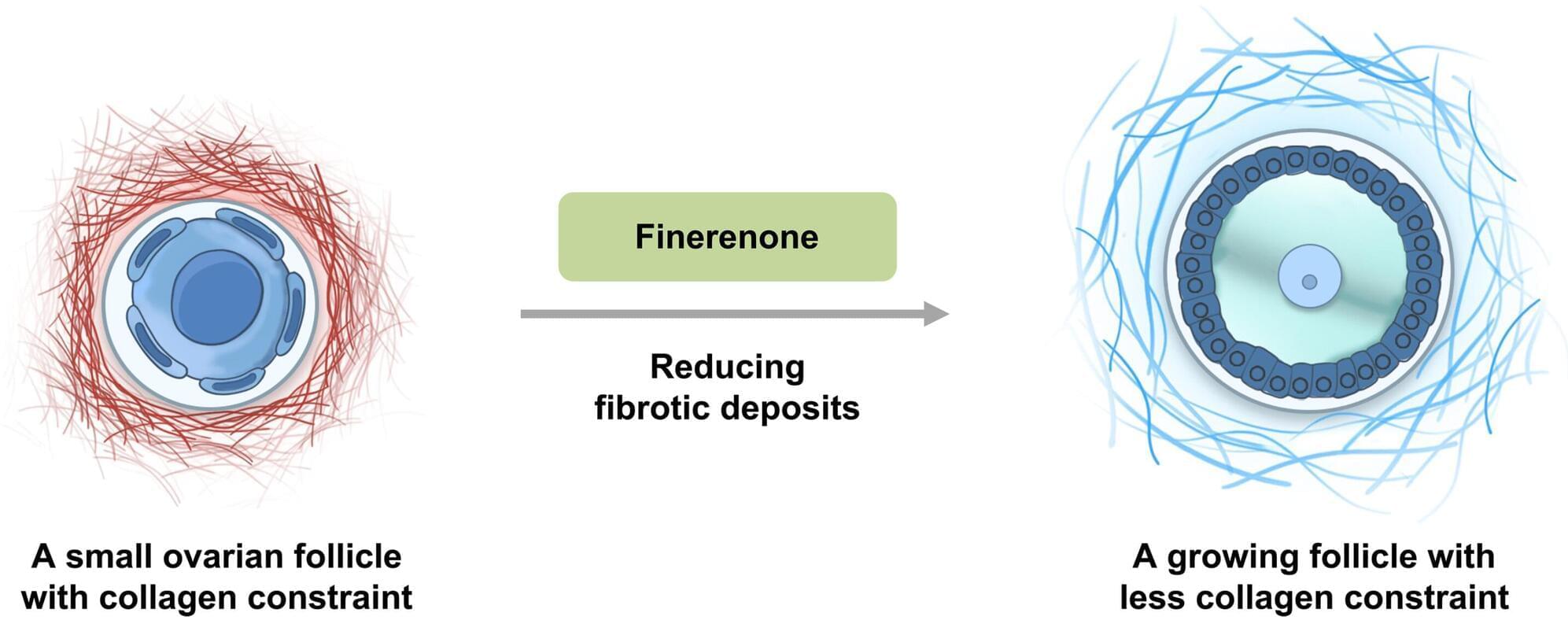
A kidney drug may help restore fertility in premature ovarian insufficiency
A common kidney medication could be the key to treating a type of infertility that affects up to 3% of women under 40, according to a study published in Science.
Premature ovarian insufficiency (POI) is a condition where the ovaries stop functioning properly, leading to low estrogen levels and follicles that often fail to develop or respond to fertility treatments. This leaves the eggs in a dormant state. Current treatments are limited to managing symptoms such as hot flashes and night sweats.
For years, scientists focused on the eggs themselves, but researchers led by Tianren Wang and Kui Liu from the University of Hong Kong suspected the problem could be the environment the eggs live in (the ovarian stroma).
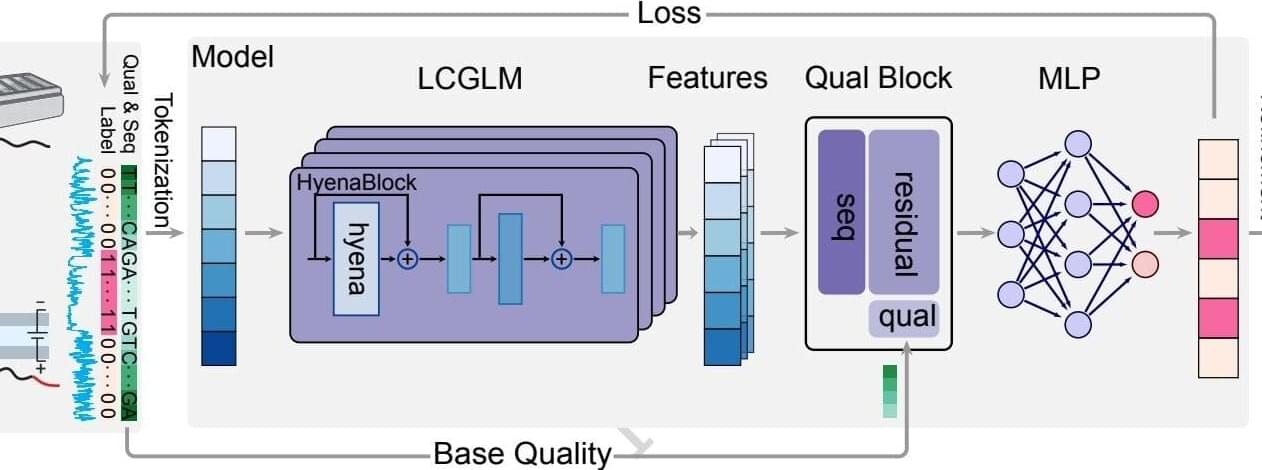
DeepChopper model improves RNA sequencing research by mitigating chimera artifacts
Scientists in the laboratory of Rendong Yang, Ph.D., associate professor of Urology, have developed a new large language model that can interpret transcriptomic data in cancer cell lines more accurately than conventional approaches, as detailed in a recent study published in Nature Communications.
Long-read RNA sequencing technologies have transformed transcriptomics research by detecting complex RNA splicing and gene fusion events that have often been missed by conventional short-read RNA-sequencing methods.
Among these technologies includes nanopore direct RNA sequencing (dRNA-seq), which can sequence full-length RNA molecules directly and produce more accurate analyses of RNA biology. However, previous work suggests this approach may generate chimera artifacts—in which multiple RNA sequences incorrectly join to form a single RNA sequence—and limit the reliability and utility of the data.
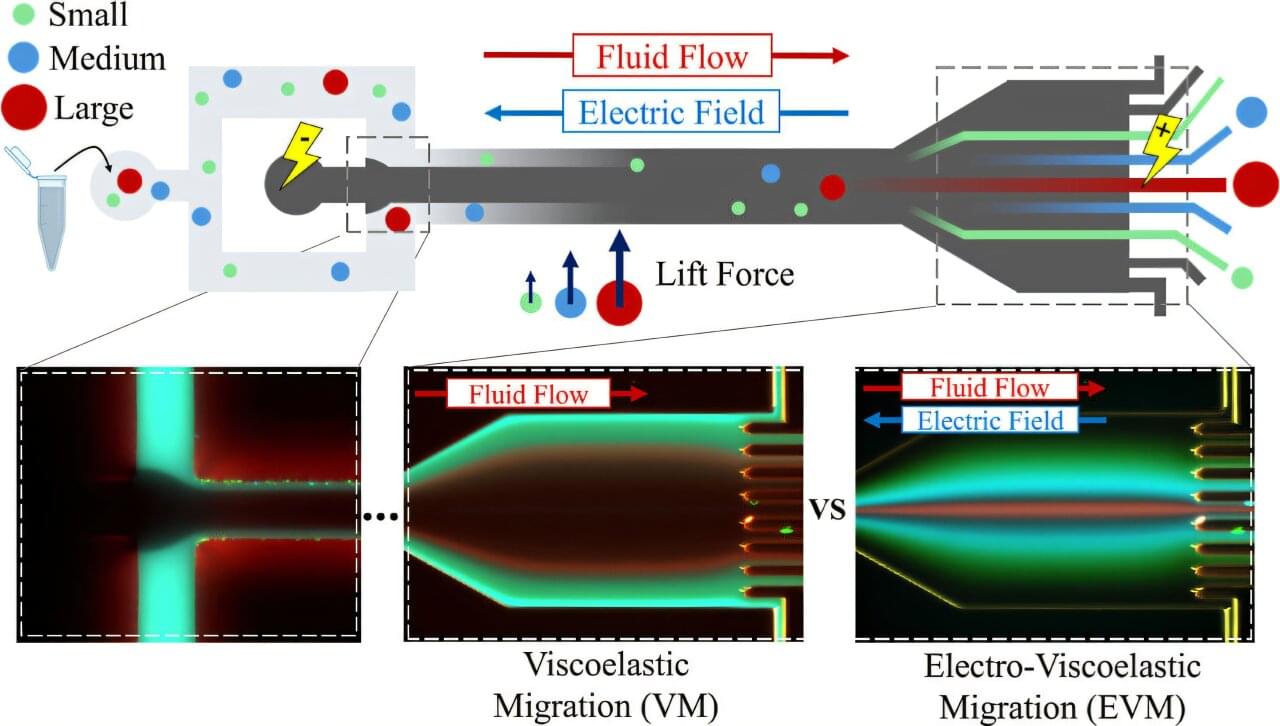
Microfluidic method boosts control and separation of tiny particles—a promising tool for medical research
In nanoscale particle research, precise control and separation have long been a bottleneck in biotechnology. Researchers at the University of Oulu have now developed a new method that improves particle separation and purification. The promising technique could be applied, for example, in cancer research.
Separating nanosized particles remains a persistent challenge in biotechnology. Once particle size drops below a few hundred nanometers, their behavior becomes dominated by diffusion—the random walk of particles. This weakens the forces used to guide them, causing separation accuracy to collapse.
A microfluidics research group led by Professor Caglar Elbuken at the University of Oulu has developed a new solution to the problem. The method significantly improves the separation and purification of both small synthetic particles and nanoscale vesicles secreted by living cells.