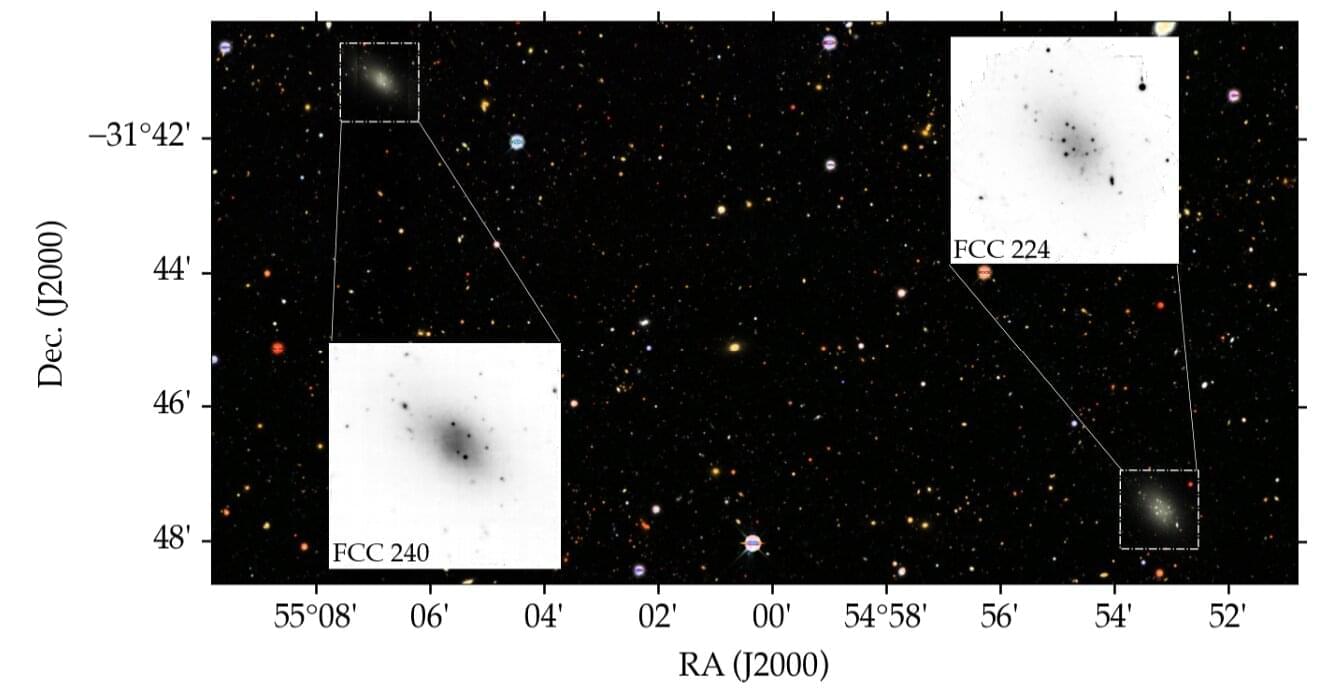
Astronomers have identified a possible new example of one of the universe’s strangest galaxy types: galaxies that appear to contain little or no dark matter. The newly studied pair, FCC 224 and FCC 240, on the outskirts of the Fornax Cluster, share several unusual traits with the only known pair of controversial dark-matter-deficient galaxies. The findings were uploaded to the arXiv preprint server on May 22.
Ultra-diffuse galaxies are faint systems that are roughly the size of the Milky Way but have much less mass, containing far fewer stars. They have sparked debate for more than 10 years, mainly because they have been observed with two contrasting levels of dark matter content.
On one end, the dark-matter-rich ultra-diffuse galaxies are reasonably well understood: These are thought to be “failed galaxies” quenched early, never building much stellar mass but holding on to many globular clusters. The opposite extreme is far stranger. A small number of ultra-diffuse galaxies appear to contain little or no dark matter at all, and the globular clusters they host are unusually bright.