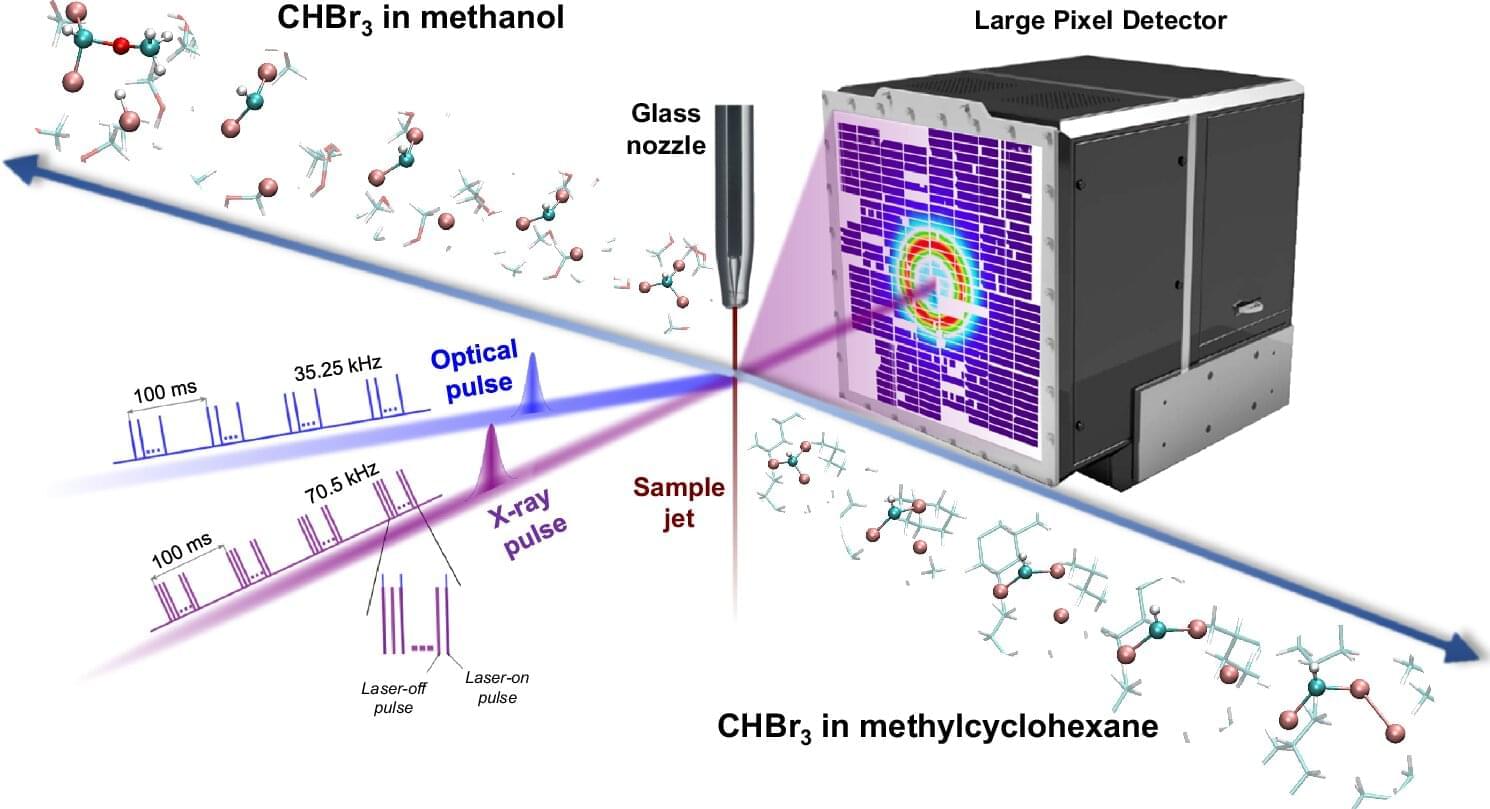
The halomethane compound bromoform (CHBr3) has devastating effects on the ozone layer. In the upper layers of the atmosphere, bromoform reacts with UV radiation, releasing bromine molecules which destroy ozone molecules. This reaction, however, has long puzzled scientists; the molecules involved seem to wander relative to each other in a way that energetically does not make sense. Scientists at European XFEL have now revealed structural evidence for this roaming mechanism for the first time, establishing it as a universal characteristic of photochemical reactions.
The study, published in Nature Communications, provides key insights into the field of atmospheric photochemistry and how halomethane compounds such as bromoform impact the ozone layer.
The ozone layer envelops Earth some 15–30 km above the planet’s surface. Ozone gas absorbs ultraviolet light as it enters the atmosphere, thereby protecting life on Earth from the effects of the harmful radiation. Ozone, however, reacts readily with other compounds also found in the stratosphere, leading to ozone depletion, and ultimately the creation of the ozone hole.