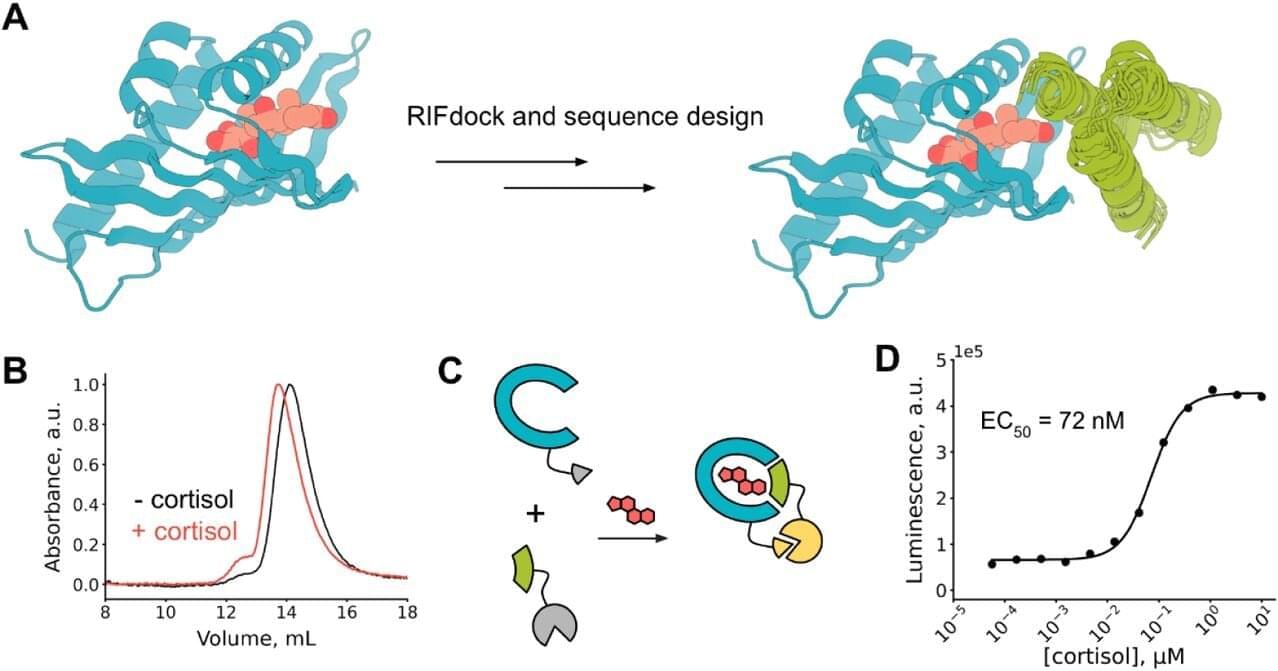
Professor Gyu Rie Lee of the Department of Biological Sciences successfully designed artificial proteins that selectively recognize specific compounds using AI through joint research with Professor David Baker. The research, published in the journal Nature Communications, is characterized by using AI to design proteins that recognize specific compounds from scratch (de novo) and implementing them as functional biosensors.
While the conventional approach mainly involved searching for natural proteins or modifying some of their functions, this research is highly significant in that it “custom-built” proteins with desired functions through AI-based design and even completed experimental verification.
In particular, the research team successfully designed a protein that selectively recognizes the stress hormone cortisol and implemented an AI-designed biosensor based on it. This is evaluated as a case that extends beyond protein design to actual measurable sensor technology, solving the long-standing challenge of small-molecule recognition in the field of protein design.